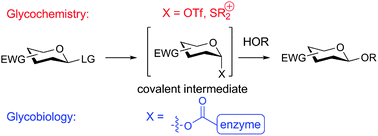
The reactivity of sugar donors and the stability of covalent intermediates formed in both chemical and biological systems is an active subject of study in both glycochemistry and glycobiology. Knowledge of the structure of these intermediates is vital for understanding reactivity and stereoselectivity in glycosidic bond formation, and in glycosidic bond destruction in the case of enzymatic hydrolysis. For chemical reactions, tuning of the electron-withdrawing power of the carbohydrate side chains allows for stabilization of covalent anomeric triflates thereby enabling chemo-, regio- and stereoselective glycosylations. Retaining glycosidase-mediated hydrolysis reactions in turn often involve a covalent intermediate. The existence of such covalent intermediates was convincingly demonstrated at the beginning of this century by making use of modified glycosyl substrates tuned such that stable adducts are formed efficiently but the ensuing hydrolysis is slowed down. Recently this concept has also been used in the design of glycosidase activity-based probes. This review describes recent investigations on different carbohydrate decoration patterns to influence both chemical and biological reactivity and selectivity.