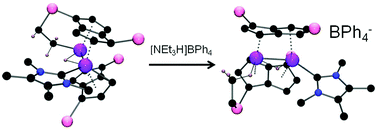
The reaction of the bis(pentalene)dititanium complex Ti2(μ:η5,η5-Pn†)2 (Pn† = C8H4(1,4-SiiPr3)2) (1) with the N-heterocyclic carbene 1,3,4,5-tetramethylimidazol-2-ylidene results in intramolecular C–H activation of an isopropyl substituent to form a tucked-in hydride (3). Whilst pyridine will also effect this cyclometallation reaction to form (5), the pyridine analogue of (3), the bases 1,2,4,5-tetramethyl-imidazole, 2,6-lutidine, DABCO or trimethylphosphine are ineffective. The reaction of (1) with 2,6-dichloro-pyridine affords crystallographically characterised (6) which is the product of oxidative addition of one of the C–Cl bonds in 2,6-dichloro-pyridine across the Ti–Ti double bond in (1). The tucked-in hydride (3) reacts with hydrogen to afford a dihydride complex (4) in which the tuck-in process has been reversed; detailed experimental and computational studies on this reaction using D2, HD or H2/D2 support a mechanism for the formation of (4) which does not involve σ-bond metathesis of H2 with the tucked-in C–H bond in (3). The reaction of (3) with tBuCCH yields the corresponding acetylide hydrido complex (7), where deuteration studies show that again the reaction does not proceed via σ-bond metathesis. Finally, treatment of (3) with HCl affords the chloro-derivative (9) [(NHC)Ti(μ-H)Ti{(μ,η5:η5)Pn†}2Cl], whereas protonation with [NEt3H]BPh4 yielded a cationic hydride (10) featuring an agostic interaction between a Ti centre and an iPr Me group.