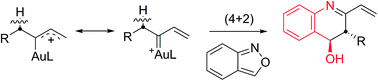Bhanudas Dattatray Mokar, Prakash D. Jadhav, Y. B. Pandit and Rai-Shung Liu
Chem. Sci., 2018,9, 4488-4492
DOI:
10.1039/C8SC00986D,
Edge Article
This work reports new (4 + 2)-annulations of α-alkyl vinylgold carbenes with benzisoxazoles to afford 3,4-dihydroquinoline derivatives with high anti-stereoselectivity. The annulations are operable with carbenes in both acyclic and cyclic forms. This reaction sequence involves an initial formation of imines from α-alkylgold carbenes and benzisoxazoles, followed by a novel carbonyl-enamine reaction to yield 3,4-dihydroquinoline derivatives. This system presents the first alkyl C–H reactivity of α-alkyl gold carbenes with an external substrate.