Interfacial memristors in Al–LaNiO3 heterostructures†
Abstract
Memristive devices are promising circuit elements that enable novel computational approaches which go beyond the von-Neumann paradigms. Here by tuning the chemistry at the Al–LaNiO3 (LNO) interface, a metal–metal junction, we engineer good switching behavior with good electroresistance (ON–OFF resistance ratios of 100), and repeatable multiple resistance states. The active material responsible for such a behavior is a self-formed sandwich of an AlxOy layer at the interface obtained by grabbing oxygen by Al from LNO. Using aberration corrected electron microscopy and transport measurements, it is confirmed that the memristive hysteresis occurs due to the electric field driven O2− (or 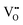 ) cycling between LNO (reservoir) and the interlayer, which drives the redox reactions forming and dissolving Al nanoclusters in the AlxOy matrix. This work provides clear insights into and details on precise oxygen control at such interfaces and can be useful for newer opportunities in oxitronics.
) cycling between LNO (reservoir) and the interlayer, which drives the redox reactions forming and dissolving Al nanoclusters in the AlxOy matrix. This work provides clear insights into and details on precise oxygen control at such interfaces and can be useful for newer opportunities in oxitronics.



 Please wait while we load your content...
Please wait while we load your content...