Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Oxidative decarboxylative radical trifluoromethylthiolation of alkyl carboxylic acids with silver(I) trifluoromethanethiolate and selectfluor†
Bin He,
Zhiwei Xiao,
Hao Wu,
Yong Guo,
Qing-Yun Chen* and
Chao Liu*
Key Laboratory of Organofluorine Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, 345 Lingling Road, Shanghai 200032, China. E-mail: chaoliu@sioc.ac.cn
First published on 3rd January 2017
Abstract
A straightforward silver-mediated oxidative decarboxylative radical trifluoromethylthiolation reaction of aliphatic carboxylic acid is described. This reaction operates under mild conditions and allows the synthesis of various valuable alkyltrifluoromethylthioethers from abundant alkyl carboxylic acids and convenient nucleophilic AgSCF3 reagent. It provides a practical and efficient approach for the preparation of alkyltrifluoromethylthioethers.
Introduction
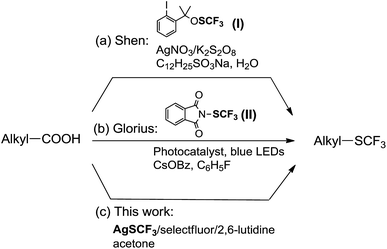
The selective introduction of fluorine and fluorine-containing groups into organic molecules can dramatically alter their physical, chemical and biological properties.1 As a star group in organofluorine chemistry in recent years, the trifluoromethylthio group (CF3S) has attracted much attention because of its electron-withdrawing nature, unique high lipophilicity and metabolic stability. Compounds containing CF3S are frequently found in pharmaceuticals and agrochemicals.2 Many methods are now available to efficiently synthesize various organic substrates containing CF3S on the basis of electrophilic, nucleophilic, radical, and oxidative direct trifluoromethylthiolation.3 Among them, more popular strategy of choice for the introduction of the CF3S group into organic substrates is the direct formation of the C–SCF3 bond. Compared with numerous methods for the efficient formation of C(sp2)–SCF3 and C(sp)–SCF3 bonds, relatively limited methods are currently available for the general and site-specific formation of various C(sp3)–SCF3 bonds. The resulting alkyltrifluoromethylthioethers are mainly accessible by the nucleophilic, electrophilic, or radical trifluoromethylthiolation of the corresponding alkanes or olefins. For examples, a direct dehydroxytrifluoromethylthiolation of alkyl alcohols using AgSCF3 and n-Bu4NI has been reported to efficiently form C(sp3)–SCF3 bonds.4 A direct nucleophilic trifluoromethylthiolation of various alkyl chlorides, bromides and tosylates with AgSCF3 was described in the presence of nBu4NI or a combination of nBu4NI/nBu4NBr.5 Several electrophilic trifluoromethylthiolation reagents have been reported for effective trifluoromethylthiolation of organometallic nucleophilic or direct α-SCF3 of carbonyl compounds.6 Recently, the direct trifluoromethylthiolation of unactivated C(sp3)–H by AgSCF3/K2S2O8 was described via a radical pathway.7Aliphatic carboxylic acids are cheap and abundant raw materials, and have been widely used as desirable starting materials for the preparation of many important and valuable compounds with different functional groups. The fundamental transformation process involves decarboxylation with the release of traceless by-product CO2 to generate alkyl radicals, and their subsequent reactions with a variety of reagents lead to efficient formation of many functionalized compounds. This important transformation process has been applied in several elegant decarboxylative fluorinations.8 Shen and co-workers described the silver-catalyzed oxidative decarboxylative trifluoromethylthiolation of alkyl carboxylic acids with electrophilic trifluoromethylthiolating reagent I in an aqueous emulsion (Scheme 1a).8b Very recently, Glorius group reported the visible-light-promoted decarboxylative trifluoromethylthiolation of alkyl carboxylic acids with electrophilic trifluoromethylthiolating reagent II under mild conditions (Scheme 1b).8f Although these methods are effective for the site-selective formation of C(sp3)–SCF3, they utilize relatively expensive electrophilic trifluoromethylthiolating reagents as the CF3S source, which are commonly prepared from readily available AgSCF3.
Recently, we developed a practical and efficient method for the direct trifluoromethylthiolation of unactivated C(sp3)–H bonds by AgSCF3/K2S2O8 under mild conditions.7b However, the regioselectivity of the reaction generally depends on the alkyl radical stability in situ formed, and several regioselective isomers are often generated in many cases. With the aim of overcoming the issue and further expanding the scope of the oxidative radical trifluoromethylthiolation methodologies, herein we report an efficient and general site-selective preparation of alkyltrifluoromethylthioethers by means of oxidative decarboxylative radical trifluoromethylthiolation of various aliphatic carboxylic acids by AgSCF3/selectfluor (Scheme 1c).
Results and discussion
Our study commenced by examing the oxidative decarboxylative trifluoromethylthiolation reaction of decanoic acid 1a with AgSCF3 (ref. 9) (1.0 equiv.) at 60 °C for 4 hours in the presence of different oxidants and solvents (Table 1). Unfortunately, no desired trifluoromethylthiolation product was observed with K2S2O8 as oxidant in CH3CN (Table 1, entry 1). To our surprise, under similar conditions, Na2S2O8 resulted in the formation of oxidative decarboxylative trifluoromethylthiolation products (Table 1, entry 2). However, after careful analysis and spectroscopic characterization, it was found that the products are not the desired product 2a, but a various di-trifluoromethylthiolation isomer mixture, which is presumably due to concurrent occurrence of both direct trifluoromethylthiolation of C(sp3)–H and the desired oxidative decarboxylative trifluoromethylthiolation of 1a (Scheme 2). In order to suppress the undesired direct trifluoromethylthiolation of C(sp3)–H, different solvents including DMSO, DMF, acetone, and ClCH2CH2Cl/CH3CN/H2O were screened. However, the reactions led to no formation of the desired products.10 When AgSCF3 was used as the limiting reagent with 5 equiv. of 1a in CH3CN in the presence of Na2S2O8, a complex di-trifluoromethylthiolation isomer mixture was still obtained (Table 1, entry 3).| Entry | AgSCF3 (equiv.) | Oxidant (equiv.) | Solvent | Yieldb (%) |
|---|---|---|---|---|
a Reaction conditions: decanoic acid (0.2 mmol, 1.0 equiv.), AgSCF3 (1.0–2.0 equiv.), oxidant (2.0–4.0 equiv.), and solvent (2.0 mL) at 60 °C for 4 hours under Ar atmosphere.b Yields were determined by 19F NMR spectroscopy with benzotrifluoride as the internal standard.c A di-trifluoromethylthiolation isomer mixture was obtained.d Acetone/CH3CN (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) was used.e Acetone/H2O (4 1 v/v) was used.e Acetone/H2O (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) was used.f 2,6-Lutidine (2.0 equiv.) was used as ligand.g CuSCF3 was used instead of AgSCF3. 1 v/v) was used.f 2,6-Lutidine (2.0 equiv.) was used as ligand.g CuSCF3 was used instead of AgSCF3. |
||||
| 1 | 1.0 | K2S2O8 (2.0) | CH3CN | 0 |
| 2 | 1.0 | Na2S2O8 (2.0) | CH3CN | 37c |
| 3 | 1.0 | (NH4)2S2O8 | CH3CN | 28c |
| 4 | 1.0 | Selectfluor (2.0) | Acetone | 31 |
| 5 | 1.0 | Selectfluor (2.0) | CH3CN | 1 |
| 6 | 1.0 | Selectfluor (2.0) | DMF | 3 |
| 7 | 1.0 | Selectfluor (2.0) | Acetone/CH3CN | 3d |
| 8 | 1.0 | Selectfluor (2.0) | Acetone/H2O | 0e |
| 9 | 1.0 | NFSI (2.0) | Acetone | 25 |
| 10 | 1.0 | FP (2.0) | Acetone | 5 |
| 11 | 2.0 | Selectfluor (2.0) | Acetone | 54 |
| 12 | 2.0 | Selectfluor (4.0) | Acetone | 65 |
| 13 | 2.0 | Selectfluor (4.0) | Acetone | 82f |
| 14 | 2.0 | Selectfluor (4.0) | Acetone | 0g |
Next, we were delighted to find that when selectfluor was used as oxidant, the desired oxidative decarboxylative monotrifluoromethylthiolation product 2a was obtained and no undesired di-trifluoromethylthiolation products were observed, demonstrating the successful suppression of the undesired direct trifluoromethylthiolation of C(sp3)–H (Table 1, entry 4). Notably, neither competitive oxidative decarboxylative fluorination reaction was observed, which was considered to presumably occur under the reaction conditions according to literature.8a These results demonstrate that oxidative decarboxylative trifluoromethylthiolation under the reaction conditions is the predominant process presumably because selectfluor would not activate the C(sp3)–H to give the corresponding key C(sp3)-centered radical intermediate. Among several common reaction solvents we screened, acetone was the best one and provided 31% yield of the desired product 2a (Table 1, entries 4–7). It is worth noting that water was harmful for the reaction and resulted in no formation of the desired product (Table 1, entry 8). Then various oxidants such as N-fluorobenzenesulfonimide (NFSI), and 1-fluoropyridium tetrafluoroborate (PF) were further tested, it turned out that worse results were observed (Table 1, entries 9 and 10). By changing the amount of AgSCF3/selectfluor, the yield of the desired product 2a was further improved to 65% (Table 1, entries 11 and 12). The influence of several ligands were also estimated, 2,6-lutidine gave the best yield of 82% (Table 1, entry 13), which might coordinate with active Ag species to enhance their stability during the reaction.10 Furthermore, the replacement of AgSCF3 with CuSCF3 led to no formation of the desired product, demonstrating the key role of the free silver cation for the reaction (Table 1, entry 14). All these experimental results indicate that a combination of AgSCF3 (2.0 equiv.)/selectfluor (4.0 equiv.)/2,6-lutidine (2.0 equiv.) in acetone at 60 °C for 4 hours is established as the optimal conditions for oxidative decarboxylative trifluoromethylthiolation reactions of various aliphatic carboxylic acids.
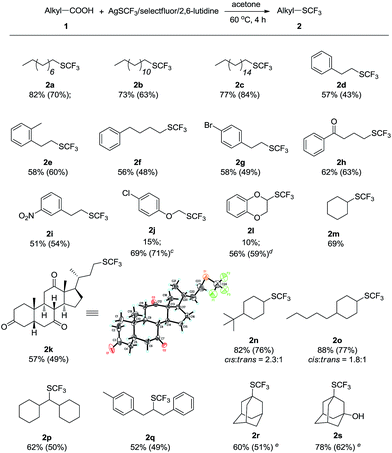
With the optimal conditions in hand, we then explored the generality of the oxidative decarboxylative trifluoromethylthiolation reactions with a variety of aliphatic carboxylic acids (Scheme 3). It was found that various aliphatic carboxylic acids including primary, secondary and tertiary carboxylic acids are all suitable substrates for the oxidative decarboxylative trifluoromethylthiolation reactions. For example, a number of primary alkyl carboxylic acids underwent smooth oxidative decarboxylative trifluoromethylthiolation to give the desired alkylthifluormethylthioethers in isolated yields of 48–84% (2a–k). Secondary aliphatic carboxylic acids could also be subjected to the reactions to provide the corresponding products in modest to good yields (2l–q). When tertiary alkyl carboxylic acids were employed as the substrates, the desired oxidative decarboxylative trifluoromethylthiolation products were generated in acceptable yields (2r, s). It should be mentioned that reduced reaction time should be applied to the aliphatic carboxylic acids 1j and 1l since significant decrease in the yields of the corresponding desired trifluoromethylthiolation products was observed under the optimal reaction conditions,10 which might be due to the instability of the corresponding products 2j and 2l under the reaction conditions. With regard to the potential of the reaction in late-stage functionalization of valuable synthetic intermediates, a structurally complicated biologically active steroid derivative was applied to the oxidative decarboxylative trifluoromethylthiolation reaction and the desired product 2k was produced smoothly in good isolated yield. Its structure was unambiguously assigned by various spectral analysis, including X-ray single-crystal analysis.
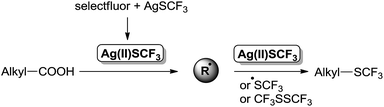
To gain some understanding on the reaction pathway, a radical inhibitor (hydroquinone) or a radical scavenger (2,2,6,6-tetramethyl-1-piperidinyloxy, TEMPO) were added to the reaction of 1a, and a sharp decrease in the yield was observed for both cases.10 These preliminary results combined with previous reports on silver-mediated oxidative decarboxylative functionalization7,8a,b indicate that the reaction proceeds through a radical pathway.10 First, AgSCF3 is oxidized by selectfluor to generated Ag(II)SCF3 intermediate, CF3S radical or CF3SSCF3. Ag(II) combines with alkyl carboxylic acids to generate the corresponding alkyl radical via decarboxylation pathway. Then, the produced alkyl radical attacks a CF3S radical, CF3SSCF3, or abstracts the CF3S ligand of Ag(II)SCF3 intermediate to provide the desired oxidative decarboxylative trifluoromethylthiolation product (Scheme 4).
Conclusions
In conclusion, we developed a practical and effective silver-meditated oxidative decarboxylative trifluoromethylthiolation reaction of aliphatic carboxylic acids using nucleophilic AgSCF3 as CF3S source and selectfluor as the oxidant. The reaction can be applied to primary, secondary and tertiary alkyl carboxylic acids under mild conditions and is tolerant of many functional groups. It represents an alternative approach for site-selective construction of C(sp3)–SCF3 bonds.Acknowledgements
The authors gratefully acknowledge the financial support from the National Basic Research Program of China (2012CB821600), the National Natural Science Foundation of China (21032006, 21302207, 21421002).Notes and references
- (a) J.-P. Bégué and D. Bonnet-Delpon, Bioorganic and Medicinal Chemistry of Fluorine, John Wiley & Sons, New Jersey, 2008 Search PubMed; (b) I. Ojima, Fluorine in Medicinal Chemistry and Chemical Biology, Wiley-Blackwell, Hong Kong, 2009 Search PubMed; (c) W. K. Hagmann, J. Med. Chem., 2008, 51, 4359 CrossRef CAS PubMed; (d) S. Purser, P. R. Moore, S. Swallow and V. Gouverneur, Chem. Soc. Rev., 2008, 37, 320 RSC; (e) K. Müller, F. Faeh and F. Diederich, Science, 2007, 317, 1881 CrossRef PubMed.
- J. Wang, M. Sánchez-Roselló, J. L. Aceña, C. D. Pozo, A. E. Sorochinsky, S. Fustero, V. A. Soloshonok and H. Liu, Chem. Rev., 2014, 114, 2432 CrossRef CAS PubMed.
- For selected reviews, see: (a) X.-H. Xu, K. Matsuzaki and N. Shibata, Chem. Rev., 2015, 115, 731 CrossRef CAS PubMed; (b) F. Toulgoat, S. Alazet and T. Billard, Eur. J. Org. Chem., 2014, 2415 CrossRef CAS; (c) X. Shao, C. Xu, L. Lu and Q. Shen, Acc. Chem. Res., 2015, 48, 1227 CrossRef CAS PubMed; (d) L. Chu and F.-L. Qing, Acc. Chem. Res., 2014, 47, 1513 CrossRef CAS PubMed; (e) G. Landelle, A. Panossian, S. Pazenok, J.-P. Vors and F. Leroux, Beilstein J. Org. Chem., 2013, 9, 2476 CrossRef PubMed; (f) T. Liang, C. Neumann and T. Ritter, Angew. Chem., Int. Ed., 2013, 52, 8214 CrossRef CAS PubMed; (g) V. Boiko, Beilstein J. Org. Chem., 2010, 6, 880 CrossRef PubMed; (h) K. Zhang, X. Xu and F. Qing, Chin. J. Org. Chem., 2015, 35, 556 CrossRef CAS; (i) H. Zheng, Y. Huang and Z. Weng, Tetrahedron Lett., 2016, 57, 1397 CrossRef CAS; (j) H. Chachignon and D. Cahard, Chin. J. Chem., 2016, 34, 445 CrossRef CAS.
- J.-B. Liu, X.-H. Xu, Z.-H. Chen and F.-L. Qing, Angew. Chem., Int. Ed., 2015, 54, 897 CrossRef CAS PubMed.
- C. Xu, Q. Chen and Q. Shen, Chin. J. Chem., 2016, 34, 495 CrossRef CAS.
- For representative examples, see: (a) X. Shao, X. Wang, T. Yang, L. Lu and Q. Shen, Angew. Chem., Int. Ed., 2013, 52, 3457 CrossRef CAS PubMed; (b) E. V. Vinogradova, P. Müller and S. L. Buchwald, Angew. Chem., Int. Ed., 2014, 53, 3125 CrossRef CAS PubMed; (c) X. Wang, T. Tang, X. Cheng and Q. Shen, Angew. Chem., Int. Ed., 2013, 52, 12860 CrossRef CAS PubMed; (d) C. Xu, B. Ma and Q. Shen, Angew. Chem., Int. Ed., 2014, 53, 9316 CrossRef CAS PubMed; (e) X. Shao, T. Liu, L. Lu and Q. Shen, Org. Lett., 2014, 16, 4738 CrossRef CAS PubMed; (f) F. Baert, J. Colomb and T. Billard, Angew. Chem., Int. Ed., 2012, 51, 10382 CrossRef CAS PubMed; (g) T. Bootwicha, X. Liu, R. Pluta, I. Atodiresei and M. Rueping, Angew. Chem., Int. Ed., 2013, 52, 12856 CrossRef CAS PubMed; (h) Y. D. Yang, A. Azuma, E. Tokunaga, M. Y. Amasaki, M. Shiro and N. Shibata, J. Am. Chem. Soc., 2013, 135, 8782 CrossRef CAS PubMed; (i) X. L. Zhu, J. H. Xu, D. J. Cheng, L. J. Zhao, X. Y. Liu and B. Tan, Org. Lett., 2014, 16, 2192 CrossRef CAS PubMed; (j) S. Alazet, L. Zimmer and T. Billard, J. Fluorine Chem., 2015, 171, 78 CrossRef CAS.
- (a) S. Guo, X. Zhang and P. Tang, Angew. Chem., Int. Ed., 2015, 54, 4065 CrossRef CAS PubMed; (b) H. Wu, Z. Xiao, J. Wu, Y. Guo, J.-C. Xiao, C. Liu and Q.-Y. Chen, Angew. Chem., Int. Ed., 2015, 54, 4070 CrossRef CAS PubMed.
- For recent reports, see: (a) F. Yin, Z. Wang, Z. Li and C. Li, J. Am. Chem. Soc., 2012, 134, 10401 CrossRef CAS PubMed; (b) F. Hu, X. Shao, D. Zhu, L. Lu and Q. Shen, Angew. Chem., Int. Ed., 2014, 53, 6105 CrossRef CAS PubMed; (c) M. Rueda-Becerril, O. Mahe, M. Drouin, M. B. Majewski, J. G. West, M. O. Wolf, G. M. Sammis and J. F. Paquin, J. Am. Chem. Soc., 2014, 136, 2637 CrossRef CAS PubMed; (d) S. Ventre, F. R. Petronijevic and D. W. C. MacMillan, J. Am. Chem. Soc., 2015, 137, 5654 CrossRef CAS PubMed; (e) H. Huang, K. Jia and Y. Chen, Angew. Chem., Int. Ed., 2015, 54, 1881 CrossRef CAS PubMed; (f) L. Candish, L. Pitzer, A. Gómez-Suárez and F. Glorius, Chem.–Eur. J., 2016, 22, 4753 CrossRef CAS PubMed.
- The exact molecular formula of silver(I) trifluoromethanethiolate prepared according to the literature ( G. Teverovskiy, D. S. Surry and S. L. Buchwald, Angew. Chem., Int. Ed., 2011, 50, 7312 CrossRef CAS PubMed ) is 3AgSCF3·CH3CN.7b For simplicity, we used AgSCF3 instead of 3AgSCF3·CH3CN in the text.
- See the ESI for details.†.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1484006. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra26133g |
| This journal is © The Royal Society of Chemistry 2017 |