Open Access Article
Open Access ArticleA lab-on-a-chip for preconcentration of bacteria and nucleic acid extraction
M. Hügle†
 ab,
G. Dame†b,
O. Behrmannab,
R. Rietzela,
D. Karthe
ab,
G. Dame†b,
O. Behrmannab,
R. Rietzela,
D. Karthe c,
F. T. Hufertb and
G. A. Urbana
c,
F. T. Hufertb and
G. A. Urbana
aLaboratory for Sensors, Department of Microsystems Engineering (IMTEK), University of Freiburg, Georges-Köhler-Allee 103, Freiburg, Germany
bInstitute of Microbiology and Virology, Brandenburg Medical School Theodor Fontane, Neuruppin, Germany
cGerman-Mongolian Institute of Resources and Technology, Nalaikh, Mongolia
First published on 1st June 2018
Abstract
To improve detection sensitivity, molecular diagnostics require preconcentration of low concentrated samples followed by rapid nucleic acid extraction. This is usually achieved by multiple centrifugation, lysis and purification steps, for instance, using chemical reagents, spin columns or magnetic beads. These require extensive infrastructure as well as time consuming manual handling steps and are thus not suitable for point of care testing (POCT). To overcome these challenges, we developed a microfluidic chip combining free-flow electrophoretic (FFE) preconcentration (1 ml down to 5 μl) and thermoelectric lysis of bacteria as well as purification of nucleic acids by gel-electrophoresis. The integration of these techniques in a single chip is unique and enables fast, easy and space-saving sample pretreatment without the need for laboratory facilities, making it ideal for the integration into small POCT devices. A preconcentration efficiency of nearly 100% and a lysis/gel-electrophoresis efficiency of about 65% were achieved for the detection of E. coli. The genetic material was analyzed by RT-qPCR targeting the superfolder Green Fluorescent Protein (sfGFP) transcripts to quantify mRNA recovery and qPCR to determine DNA background.
Introduction
The rapid detection of pathogens by nucleic acid amplification techniques (NAT) such as qPCR or isothermal amplification methods is of importance in diverse fields of the life sciences including medicine, veterinary medicine, food and water monitoring as well as the environmental sciences. For samples containing low amounts of the target organism(s), such as body fluids or drinking water,1 a preconcentration step is often required for successful detection. Consequently, the sample preparation for rapid detection of pathogens by NAT almost always requires a combined approach of sample preconcentration and nucleic acid extraction. This is usually done in four major steps. First, a preconcentration step is performed to reduce the risk of false-negative results in samples with low concentrations of pathogens. In a second step, the pathogens are lysed resulting in the release of their genetic material. Finally, the nucleic acids are purified and detected by amplification of pathogen-specific target sequences. This approach is much faster than conventional methods relying on the cultivation of pathogens, but requires large and thus immobile laboratory facilities as well as trained professionals for handling. To overcome these problems, innovative systems are needed which are not only faster but also smaller and easier to handle and operate. Over the last two decades, considerable efforts have been undertaken to automate and integrate certain laboratory processes into miniaturized, chip-based devices.2 For preconcentration of bacteria from liquid samples, different approaches have been pursued. Currently used techniques include dielectrophoresis,3,4 antibody modified surfaces,5,6 acoustophoresis,7 micropillar sieving,8 the use of magnetic beads9,10 or electrophoretic transport.11 For the extraction of nucleic acids, usually specific protocols or kits depending on the biological sample and medium are used. The use of magnetic beads,12,13 membrane based extraction2,14 or paperfluidics15 are some examples. In previous works, we have presented on-chip lysis and RNA extraction from bacteria16,17 as well as FFE preconcentration of viable bacteria from liquid samples.18,19 Here, we present a lab-on-a-chip system based on these results. The chip includes on-chip FFE preconcentration and thermoelectric lysis of bacteria as well as separation of nucleic acids by on-chip gel-electrophoresis. The combination of these techniques in a single chip is unique and allows for fast, easy and space-saving sample pretreatment without the need for laboratory facilities, making it ideal for the integration into small POCT devices. Due to the exclusion of larger nucleic acid fractions (e.g. gDNA or plasmids) by on-chip gel-electrophoresis, the developed chip is especially suited for highly sensitive RNA based pathogen detection protocols. By simplifying the difficult and error prone extraction of pathogen-specific RNA, the chip enables transcriptome analysis at the point-of-care/need. As RNA transcripts are typically present in much higher copy numbers20,21 than the singular copy of gDNA in each cell, targeting RNA can lead to powerful increases in detection sensitivity. This is especially important for the detection of clinically significant low-level infections such as bacteremia or infections of cerebrospinal fluid.22 To demonstrate the functionalities of the chip, fluorescent E. coli spiked into LB medium were concentrated from an initial volume of 1 ml down to 5 μl. Preconcentration was directly followed by thermoelectric lysis of the bacteria and gel-electrophoresis of the released genetic material. The extracted nucleic acids were then analyzed by RT-qPCR to detect sfGFP23 mRNA as a marker and qPCR to determine the residual DNA background.Materials and methods
Bacterial culture
XL1-Blue competent E. coli transformed with a expression vector (pMH-33, 5.359 bp) containing the sfGFP gene were used for all experiments. Cells were grown in 5 ml LB-Luria media with 100 μg ml−1 ampicillin in a 15 ml sterile plastic tube. After the prepared medium was inoculated with a single colony of the E. coli strain, the tube was placed in an incubator at 37 °C and shaken at 200 rpm. Eighteen hours later, the culture was diluted in 50 ml LB-media with 100 μg ml−1 ampicillin in a sterilized 100 ml culture flask. The culture was placed in the incubator at 37 °C and shaken at 200 rpm. until the optical density at 600 nm (OD600) had increased from an initial value of 0.1 to a final value of 0.7. Arabinose was then added at a final concentration of 0.2% (w/v) to induce sfGFP expression and the culture flask was incubated for an additional three hours. The bacteria were then immediately used for the experiments.Lab-on-a-chip
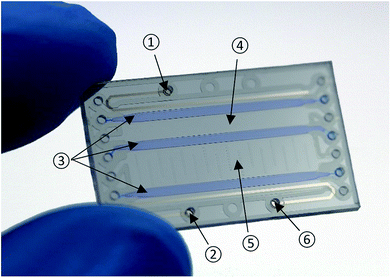
The developed chip (Fig. 1) is made from Pyrex glass substrates and four layers of a dry-film photoresist (Ordyl SY300, Elga Europe). It combines free-flow electrophoretic preconcentration, thermoelectric lysis as well as on-chip gel-electrophoresis of nucleic acids.Layout
The chip comprises four chambers separated by three 1 mm wide polyacrylamide gels. Control of wetted areas inside the chip is achieved by patterned hydrodynamic pressure barriers (“phaseguides”).24 The total dimensions of the chip are 16 mm × 1.12 mm × 27 mm (width × height × depth). All chambers have a height of 120 μm and a depth of 20 mm. For preconcentration, lysis and electrophoresis two platinum electrodes at a distance of 10 mm are located in the outermost chambers. To reduce the risk of bubble trapping, these chambers have a width of only 1 mm resulting in a volume of 2.4 μl. The elution chamber is twice as wide (4 mm) as the sample chamber and has a volume of 10 μl.Fabrication process
Two 4 inch Pyrex glass wafers with a thickness of 500 μm are used as the top and bottom substrates of the chip. In a first step, the electrodes consisting of two layers (50 nm titanium and 120 nm platinum) are patterned on the bottom wafer using a standard lift-off process. Then, four layers (30 μm each) of a dry-film photoresist are laminated onto it, followed by an exposure and development step that patterns the phaseguides and the chamber walls. After the inlet holes have been drilled into the top wafer, the wafers are thermally bonded and individual chips are released by dicing. Further details of the fabrication process may be found in Vulto et al.25Device operation
The chip has three main functions that are performed consecutively. Prior to an experiment, a 5% polyacrylamide gel (125 μl 40% 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
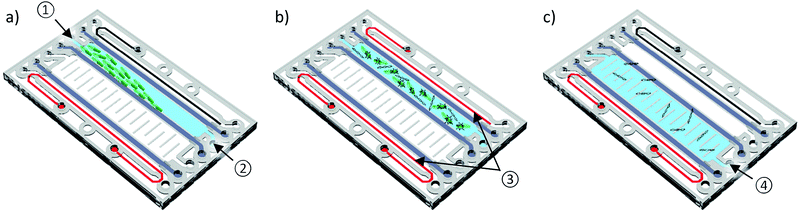
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 acrylamide/bisacrylamide solution, 867.5 μl 1× TBE, 2 μl TEMED and 5.5 μl 10% ammonium persulfate) is dispensed into the provided chambers. After 15 min in a moisturized nitrogen atmosphere, the polymerization of the gel is complete and the chip is ready for use. Fig. 2 illustrates the general process flow:
1 acrylamide/bisacrylamide solution, 867.5 μl 1× TBE, 2 μl TEMED and 5.5 μl 10% ammonium persulfate) is dispensed into the provided chambers. After 15 min in a moisturized nitrogen atmosphere, the polymerization of the gel is complete and the chip is ready for use. Fig. 2 illustrates the general process flow:
(a) Free-flow electrophoretic preconcentration: the bacteria sample is pumped continuously through the sample chamber whilst a DC voltage (100 V) is applied between the cathode (black) and anode (red). Due to the perpendicular electrical field, the negatively charged bacteria are deflected towards the anode resulting in trapping at the middle gel barrier. To avoid bubble formation, the electrode chambers are rinsed with fresh 1× TBE at a flow rate of 200 μl min−1 during the complete process. Detailed information about the on-chip FFE-preconcentration can be found in the work of Podszun et al.19
(b) Lysis: a sinusoidal AC voltage (20 kHz/230 Vpp) is applied to cause lysis of the concentrated bacteria leading to a release of their genetic material. It is performed until the formation of heat induced gas bubbles is observed. Parameters for lysis are established by Vulto et al.16 The devices used for lysis are a function generator (33120A, Hewlett-Packard) and a voltage amplifier (A600, FLC Electronics).
(c) Gel-electrophoresis: finally, the released nucleic acids are transferred through the middle gel into the elution chamber, which is filled with fresh 1× TBE, by gel-electrophoresis at a DC voltage of 100 V. Larger cell debris is retained in the sample chamber.
All samples and extracts are immediately stored at −20 °C. Each experiment is repeated three times.
Experimental setup
Fig. 3 shows the custom chip holder milled from polyetheretherketone (PEEK). For preconcentration, a disposable poly methyl methacrylate (PMMA) connector is inserted into the chip holder. Two Luer- and four Lee (062 MINSTAC – The Lee Company) connectors as well as two spring probe pins are used for interfacing. Prior to an experiment, the sample is filled inside a 1 ml syringe (B|BRAUN – Omnifix®F) and the chip is mounted into the fixture. By means of a syringe pump, the sample is pumped through the chip into another 1 ml syringe, which acts as waste reservoir. | ||
| Fig. 3 CNC milled custom chip holder. ① Syringe ② pressure clamp ③ disposable PMMA connector ④ lee connector ⑤ luer connector ⑥ electrical port ⑦ O-ring ⑧ chip. | ||
RT-qPCR and qPCR
Eluted mRNA (758 bp) and plasmid DNA obtained from fluorescent E. coli were quantified using RT-qPCR and qPCR. Fluorescence readout and thermal cycling were performed using the LightCycler®1.5 (Roche). The supplied data analysis software suite was used to determine the cycle threshold (Ct). All error bars indicate standard deviation.Protocol for RT-qPCR
Reverse transcription of the extracted mRNA and the qPCR-reaction are carried out in one tube using Thermus thermophilus (Tth) polymerase (BIORON GmbH). The total reaction volume of 20 μl containing 5× RT-PCR buffer (50 mM bicine/KOH, 115 mM K-acetate, 40% glycerol (v/v)), dNTP mix (0.2 mM), MnCl2 (4 mM), forward primer (0.5 mM/5′-CAAAGATGACGGGAC CTACA-3′), reverse primer (0.5 mM/5′-CCAGTTTGTGTCCGA GAATG-3′), TaqMan probe (0.3 mM/FAM-TTCAAACTTGACT TCAGCACGCGTC-BBQ), Tth polymerase (2.5 U/rx) and 1 μl sample is pipetted into LightCycler® capillaries. The size of the amplicon is 125 bp. The one step RT-qPCR cycle protocol begins with an initial denaturation for 2 min at 94 °C followed by reverse transcription (30 min at 60 °C) and 50 cycles of PCR (15 s at 94 °C; 30 s at 60 °C).Protocol for qPCR
qPCR of the extracted plasmid DNA is carried out using Tth polymerase (BIORON GmbH). The final reaction volume of the Tth polymerase mix amounts to 20 μl and consists of 10× PCR buffer (10 mM Tris–HCl, 100 mM KCl, 500 μg ml−1 BSA, 0.5% Tween 20 and 1.5 mM MgCl2), dNTP mix (0.2 mM), forward primer (0.5 mM), reverse primer (0.5 mM), TaqMan probe (0.3 mM) and Tth polymerase (2 U/rx). After an initial denaturation step (2 min at 94 °C) 50 cycles of PCR (15 s at 94 °C, 30 s at 60 °C) are performed.Results and discussion
To demonstrate the operation of the system, fluorescent E. coli were preconcentrated from an initial volume of 1 ml down to 5 μl. Preconcentration was followed by thermoelectric lysis and on-chip gel-electrophoresis of released nucleic acids to remove larger fractions (e.g. gDNA or plasmids) and residual cell debris.Induction controls
To verify the induction of sfGFP in the presence of arabinose, direct (RT)-qPCR of samples with and without added arabinose was performed omitting any preconcentration (Table 1).| RT-qPCR | qPCR | |
|---|---|---|
| E. coli (+ara.) | 15.05 ± 0.33 | 20.16 ± 0.49 |
| E. coli (−ara.) | 19.09 ± 0.83 | 20.78 ± 0.73 |
The Ct-values of the qPCR are constant for both experimental conditions, indicating a stable concentration of plasmid DNA. Interestingly, the RT-qPCR Ct-value of the uninduced (E. coli-ara.) culture is in the same range (slightly higher Ct-value) as the one obtained by qPCR, indicating that the RT-qPCR signal is predominantly due to the presence of the plasmid DNA background. The Ct-values of the induced and uninduced samples show a difference of 4.04 ± 0.51, which corresponds to an about 16-fold increase in target copy number indicating successful sfGFP induction by arabinose. A Ct-difference of 5.1 ± 0.38 was observed for E. coli (+ara.) when measured by RT-qPCR and qPCR, showing that sfGFP mRNA is present at about 34-fold higher levels than the plasmid DNA after arabinose induction.
Free-flow electrophoretic preconcentration of bacteria
The deflection of the bacteria towards the anode and successful trapping at the middle gel barrier (Fig. 2, step a) highly depends on the strength of the applied electric field. Therefore, the voltage should be as high as possible to achieve a high preconcentration efficiency. However, the higher the voltage, the more heat induced gas bubbles were formed by resistive heating, interfering with the FFE inside the chip. Previous observations showed that a maximum DC voltage of 100 V could be applied to guarantee process stability. This means that electrolytic gas was removed efficiently from the electrode chambers and no formation of heat induced gas bubbles inside the chip was observed during the whole process duration. With a fixed voltage of 100 V, the highest flow rate that achieved successful trapping of bacteria had to be determined. For this, a bacteria suspension was continuously pumped through the chip at different flow rates, whilst observing the chip under a fluorescence microscope. Fig. 4 shows the sample chamber of the chip at three different flow rates and a constant DC voltage of 100 V. The inlet is on the right side. The bright green lines are bacteria, which were previously collected at the gel front. The red line highlights the deflection of the bacteria caused by the applied electric field. At a flow rate of 10 μl min−1 and 20 μl min−1 hardly any loss of visible bacteria was observed. At 30 μl min−1 there was a substantial number of bacteria observed exiting the flow channel. Thus, a DC voltage of 100 V and a sample flow rate of 20 μl min−1 were chosen for all further experiments. To determine the preconcentration efficiency, suspensions of E. coli sfGFP (500 μl and 1000 μl) were reduced to a volume of 5 μl (sample chamber volume) inside the chip (Fig. 2, step a), resulting in a 100- or 200-fold reduction of the sample volume. After preconcentration, the outflow (waste) as well as the initial sample were analyzed by direct RT-qPCR for the presence of sfGFP mRNA as well as for the presence of viable bacteria by the plate count method. Preconcentration efficiencies were then calculated according eqn (1) and eqn (2) for the plate-count method and for RT-qPCR, respectively.
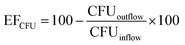 | (1) |
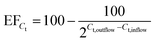 | (2) |
Table 2 shows the results for the plate-count method and Table 3 for RT-qPCR.
| Plate Count | |
|---|---|
| CFUinflow (ml−1) | 6.22 × 106 ± 6.05 × 105 |
| Inflow sample vol. (μl) | 500 |
| CFUoutflow (ml−1) | 8.19 × 103 ± 5.32 × 103 |
| Reduction in sample volume | 100-fold |
| EFCFU | 99.86% ± 0.093 |
| RT-qPCR | |
|---|---|
| Ct,inflow | 22.97 ± 1.24 |
| Inflow sample vol. (μl) | 1000 |
| Ct,outflow | None (>50 cycles) |
| Reduction in sample volume | 200-fold |
| EFCt | > 99.99% |
Both analysis methods showed that more than 99% of all detectable bacteria were successfully captured inside the chip at a flow rate of 20 μl min−1 and a DC voltage of 100 V.
Lysis
Bacteria were thermoelectrically lysed by applying a sinusoidal voltage to the coplanar electrodes (Fig. 2, step b). The applied voltage and frequency were the same as shown by Vulto et al.16 to be effective for the lysis of E. coli (230 Vpp/20 kHz). Lysis duration was limited by the formation of heat induced gas bubbles inside the microfluidic chambers. A duration of 60 s was found to be the longest possible time before unacceptable levels of bubbles occurred that pushed fluid out of the filling ports.On-chip gel-electrophoresis and detection of nucleic acids
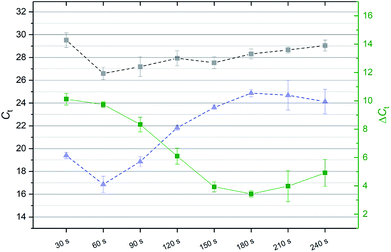
The electrophoretic migration time depends on the size of the desired nucleic acid fraction, the applied voltage as well as the composition and width of the polyacrylamide gel inside the chip. In order to determine the migration time of the nucleic acids from the sample into the elution chamber, a time series was performed. For this, a DC voltage (100 V) was applied to the electrodes (Fig. 2, step c). Eluted nucleic acids (10 μl) were removed from the elution chamber and replaced with fresh 1× TBE-buffer every 30 seconds up to a maximum of 240 seconds using a pipette. The extracts were then analyzed for the presence of sfGFP transcripts and its DNA coding region by RT-qPCR and qPCR. Fig. 5 shows the Ct-values (left axis) of the extracts from the elution chamber (30 s - 240 s) obtained by qPCR (gray, dashed) and RT-qPCR (blue, dashed). The green line depicts the difference (ΔCt, right axis) of the qPCR and RT-qPCR results. The amount of extracted DNA remained almost constant over the complete time series, whereas the amount of extracted mRNA decreased sharply after 60 s. The difference of the detected mRNA and DNA levels (green line, right axis) also decreased sharply after the 60 s fraction. As the goal was the reliable detection of the target sequence (mRNA) with a low background (DNA), a duration of 60 s was used for gel-electrophoresis for all further experiments as mRNA was detected at an about 855-fold (ΔCt = 9.74 ± 0.2) higher level than DNA.Combined operation for signal enhancement
To demonstrate the full functionality of the microfluidic chip, a bacteria suspension was diluted 200-fold and sfGFP mRNA was extracted by on-chip lysis and gel-electrophoresis with and without FFE preconcentration. First, mRNA was extracted from 5 μl of the diluted bacteria suspension by on-chip lysis and gel-electrophoresis (Fig. 2, steps b and c). Next, 1000 μl of the same diluted sample were preconcentrated by on-chip FFE within the sample chamber (5 μl) and mRNA was extracted from the collected bacteria (Fig. 2, steps a, b and c). Table 4 shows the experimental results.| Ct, no preconcentration | 24.97 ± 0.98 |
| Ct, preconcentration | 17.88 ± 1.65 |
The mRNA extracted without preconcentration was detected at Ct = 24.97 ± 0.98 and with preconcentration at Ct = 17.88 ± 1.65. The threshold-cycle difference of the two experiments (ΔCt = 7.09) corresponds to a 136-![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) fold increase in mRNA concentration after preconcentration. The overall detection of bacteria is thus enhanced 136-fold by the addition of the FFE preconcentration step. Assuming a preconcentration efficiency of >99%, a 200-fold increase in the amount of eluted mRNA is expected when comparing the mRNA amounts resulting from the experiments with and without FFE preconcentration. Considering the RT-qPCR results of the diluted (Ct = 23.14 ± 0.87) and undiluted (Ct = 15.44 ± 0.95) sample material which were dispensed into the sample chamber, the expected amount of mRNA concentration increases up to 208-fold (ΔCt = 7.70 ± 0.77). This allows for the calculation of the combined lysis/electrophoresis efficiency of the chip system as
fold increase in mRNA concentration after preconcentration. The overall detection of bacteria is thus enhanced 136-fold by the addition of the FFE preconcentration step. Assuming a preconcentration efficiency of >99%, a 200-fold increase in the amount of eluted mRNA is expected when comparing the mRNA amounts resulting from the experiments with and without FFE preconcentration. Considering the RT-qPCR results of the diluted (Ct = 23.14 ± 0.87) and undiluted (Ct = 15.44 ± 0.95) sample material which were dispensed into the sample chamber, the expected amount of mRNA concentration increases up to 208-fold (ΔCt = 7.70 ± 0.77). This allows for the calculation of the combined lysis/electrophoresis efficiency of the chip system as
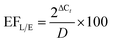 | (3) |
Conclusions and outlook
We have developed an lab-on-a-chip that combines highly efficient FFE preconcentration of bacteria with nucleic acid extraction for efficient detection of bacteria from dilute aqueous samples by organism specific mRNA. To demonstrate the full functionality of the chip, 1 ml of a dilute bacteria sample was preconcentrated down to a volume of 5 μl. This step was followed by on-chip thermo-electric lysis and on-chip gel-electrophoresis of released mRNAs. All processing steps were completed in less than one hour with minimal hands-on time. Specific mRNA was then quantified in the extracts by RT-qPCR. The system showed an efficiency of nearly 100% for preconcentration and about 65% for on-chip lysis and gel-electrophoresis. Overall, a 136-fold signal improvement was achieved for the detection of bacteria by addition of the FFE preconcentration step. The unique combination of these techniques in a single chip allows for easy, fast and space-saving sample pretreatment making it ideal for the integration into small POCT devices. Furthermore, it reduces the risk of cross-contamination and the loss of sample material by additional pipetting steps.To enable field deployment capability, we have developed a compact power supply using a boost converter for high-voltage generation and high-voltage operational amplifiers for amplification of the waveforms necessary for on-chip lysis. By optimizing the mRNA/DNA-separation, the developed chip system could be a very useful device for both gene expression studies and further RNA-based measurements such as RNA-sequencing. For example, analysis of the bacterial transcriptome allows for more accurate determination of antimicrobial resistances at the point-of-care, as the sole presence of the resistance gene is often not directly related to the observed resistance phenotype(s).21,26 Furthermore, the system may be suitable for the detection of viruses from dilute clinical samples such as those arising from e.g. bronchoalveolar lavage.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors like to thank Dr Gregory Stevens for the protocol for sfGFP expression, Tobias Wieland for bacteria preparation, the German Federal Ministry of Education and Research (BMBF; Grant No. 033W010, EDIT) as well as the Ministry of Science, Research and Culture (MWFK; Grant No. 06-GeCa:H228-05/002/004, digilog-Gesundheitscampus Brandenburg) for funding this work. The pMH-33 plasmid was a generous gift from Prof. Dr Andreas Plückthun of the University of Zurich. The article processing charge was funded by the German Research Foundation (DFG) and the University of Freiburg in the funding programme Open Access Publishing.References
- D. Karthe, O. Behrmann, V. Blättel, D. Elsässer, C. Heese, M. Hügle, F. Hufert, A. Kunze, R. Niessner, J. Ho, B. Scharaw, M. Spoo, A. Tiehm, G. Urban, S. Vosseler, T. Westerhoff, G. Dame and M. Seidel, Environ. Earth Sci., 2016, 75, 1481 CrossRef.
- L. van Heirstraeten, P. Spang, C. Schwind, K. S. Drese, M. Ritzi-Lehnert, B. Nieto, M. Camps, B. Landgraf, F. Guasch, A. H. Corbera, J. Samitier, H. Goossens, S. Malhotra-Kumar and T. Roeser, Lab Chip, 2014, 14, 1519–1526 RSC.
- B. Del Moral-Zamora, J. Punter-Villagrassa, A. M. Oliva-Branas, J. M. Alvarez-Azpeitia, J. Colomer-Farrarons, J. Samitier, A. Homs-Corbera and P. L. Miribel-Catala, Electrophoresis, 2015, 36, 1130–1141 CrossRef PubMed.
- B. H. Lapizco-Encinas, R. V. Davalos, B. A. Simmons, E. B. Cummings and Y. Fintschenko, J. Microbiol. Methods, 2005, 62, 317–326 CrossRef PubMed.
- K. Tsougeni, G. Papadakis, M. Gianneli, A. Grammoustianou, V. Constantoudis, B. Dupuy, P. S. Petrou, S. E. Kakabakos, A. Tserepi, E. Gizeli and E. Gogolides, Lab Chip, 2016, 16, 120–131 RSC.
- U. Dharmasiri, M. A. Witek, A. A. Adams, J. K. Osiri, M. L. Hupert, T. S. Bianchi, D. L. Roelke and S. A. Soper, Anal. Chem., 2010, 82, 2844–2849 CrossRef PubMed.
- P. Ohlsson, M. Evander, K. Petersson, L. Mellhammar, A. Lehmusvuori, U. Karhunen, M. Soikkeli, T. Seppa, E. Tuunainen, A. Spangar, P. von Lode, K. Rantakokko-Jalava, G. Otto, S. Scheding, T. Soukka, S. Wittfooth and T. Laurell, Anal. Chem., 2016, 88, 9403–9411 CrossRef PubMed.
- K.-Y. Hwang, H.-K. Lim, S.-Y. Jung, K. Namkoong, J.-H. Kim, N. Huh, C. Ko and J.-C. Park, Anal. Chem., 2008, 80, 7786–7791 CrossRef PubMed.
- A. J. Kell, G. Stewart, S. Ryan, R. Peytavi, M. Boissinot, A. Huletsky, M. G. Bergeron and B. Simard, ACS Nano, 2008, 2, 1777–1788 CrossRef PubMed.
- N. Beyor, T. S. Seo, P. Liu and R. A. Mathies, Biomed. Microdevices, 2008, 10, 909–917 CrossRef PubMed.
- A. K. Balasubramanian, K. A. Soni, A. Beskok and S. D. Pillai, Lab Chip, 2007, 7, 1315–1321 RSC.
- P.-Y. Hung, P.-S. Jiang, E.-F. Lee, S.-K. Fan and Y.-W. Lu, Microsyst. Technol., 2015, 313–320 Search PubMed.
- Y. Sun, T. L. Quyen, T. Q. Hung, W. H. Chin, A. Wolff and D. D. Bang, Lab Chip, 2015, 15, 1898–1904 RSC.
- E. A. Oblath, W. H. Henley, J. P. Alarie and J. M. Ramsey, Lab Chip, 2013, 13, 1325–1332 RSC.
- N. M. Rodriguez, W. S. Wong, L. Liu, R. Dewar and C. M. Klapperich, Lab Chip, 2016, 16, 753–763 RSC.
- P. Vulto, G. Dame, U. Maier, S. Makohliso, S. Podszun, P. Zahn and G. A. Urban, Lab Chip, 2010, 10, 610–616 RSC.
- P. Vulto, P. Kuhn and G. A. Urban, Lab Chip, 2013, 13, 2931–2936 RSC.
- D. Puchberger-Enengl, S. Podszun, H. Heinz, C. Hermann, P. Vulto and G. A. Urban, Biomicrofluidics, 2011, 5, 44111–4411110 Search PubMed.
- S. Podszun, P. Vulto, H. Heinz, S. Hakenberg, C. Hermann, T. Hankemeier and G. A. Urban, Lab Chip, 2012, 12, 451–457 RSC.
- B. Glynn, K. Lacey, J. Reilly, T. Barry, T. J. Smith and M. Maher, Res. J. Biol. Sci., 2007, 564–570 Search PubMed.
- C. Stein, O. Makarewicz, Y. Pfeifer, C. Brandt, J. C. Ramos, M. Klinger and M. W. Pletz, PLoS One, 2013, e80079 Search PubMed.
- S. A. Byron, K. R. van Keuren-Jensen, D. M. Engelthaler, J. D. Carpten and D. W. Craig, Nat. Rev. Genet., 2016, 17, 257–271 CrossRef PubMed.
- J.-D. Pédelacq, S. Cabantous, T. Tran, T. C. Terwilliger and G. S. Waldo, Nat. Biotechnol., 2006, 24, 79–88 CrossRef PubMed.
- P. Vulto, S. Podszun, P. Meyer, C. Hermann, A. Manz and G. A. Urban, Lab Chip, 2011, 11, 1596–1602 RSC.
- P. Vulto, T. Huesgen, B. Albrecht and G. A. Urban, J. Micromech. Microeng., 2009, 19, 77001 CrossRef.
- G. L. Daikos, P. Petrikkos, M. Psichogiou, C. Kosmidis, E. Vryonis, A. Skoutelis, K. Georgousi, L. S. Tzouvelekis, P. T. Tassios, C. Bamia and G. Petrikkos, Antimicrob. Agents Chemother., 2009, 53, 1868–1873 CrossRef PubMed.
Footnote |
| † Joint first author |
| This journal is © The Royal Society of Chemistry 2018 |