Open Access Article
Open Access ArticlePolyphenols as NLRP3 inflammasome modulators in cardiometabolic diseases: a review of in vivo studies
Marisol
Villalva
 *ab,
Juan José
Martínez-García
*ab,
Juan José
Martínez-García
 cd,
Laura
Jaime
cd,
Laura
Jaime
 b,
Susana
Santoyo
b,
Susana
Santoyo
 b,
Pablo
Pelegrín
b,
Pablo
Pelegrín
 cd and
Jara
Pérez-Jiménez
cd and
Jara
Pérez-Jiménez
 ae
ae
aDepartment of Metabolism and Nutrition, Institute of Food Science, Technology and Nutrition (ICTAN-CSIC), Madrid, Spain. E-mail: marisol.villalva@uam.es
bInstitute of Food Science Research (CIAL). Universidad Autónoma de Madrid (CEI UAM+CSIC), 28049, Madrid, Spain
cMolecular Inflammation Group, University Clinical Hospital Virgen de la Arrixaca, Biomedical Research Institute of Murcia (IMIB-Pascual Parrilla), Murcia, Spain
dDepartment of Biochemistry and Molecular Biology B and Immunology, Faculty of Medicine, University of Murcia, Murcia, Spain
eCIBER of Diabetes and Associated Metabolic Disease (CIBERDEM), ISCIII, Madrid, Spain
First published on 3rd October 2023
Abstract
The nucleotide-binding domain and leucine-rich repeat containing receptors (NLRs) are components of the innate immune system, important in coordinating the inflammatory response. Among them, NLRP3 can form inflammasomes, multiprotein complexes activating the inflammatory caspase-1 and leading, through a cell death-mediated signaling cascade, to the release of several proinflammatory cytokines. Dietary polyphenols, plant secondary metabolites, have been reported to exhibit anti-inflammatory properties, although studies have focused most on their effect on the expression of the final circulating cytokines rather than on the upstream signals activating the NLRP3 inflammasome. The present review explores current knowledge on the potential of dietary polyphenols to regulate the whole NLRP3 inflammasome pathway, in the context of cardiometabolic pathologies (obesity, cardiovascular diseases, type 2 diabetes and non-alcoholic fatty liver disease), based on in vivo studies. A clear tendency towards a decrease in the expression of the whole NLRP3 inflammasome signaling pathway when several animal models were supplemented with polyphenols was observed, commonly showing a dose–response effect; these modifications were concomitant with clinical improvements in the pathologies. Nevertheless, the diversity of doses used, the disparity in polyphenol structures tested and, particularly, the scarce clinical trials and exploration of mechanisms of action show the need to develop further research on the topic.
1. NLRP3 inflammasome: a key driver in cardiometabolic diseases
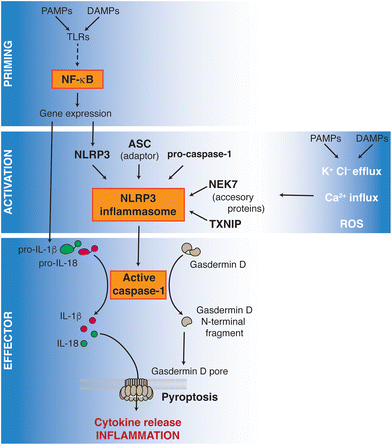
The nucleotide-binding domain and leucine-rich repeat containing receptors (NLRs) are a family of pattern recognition receptors of the innate immune system, important in coordinating the inflammatory response triggered by danger and cell damage signals, as well as by pathogens. Among the NLRs, the activation of nucleotide-binding oligomerization domain (NOD) 1 (aka. NLRC1), NOD2 (aka. NLRC2) and NLRP3 has been described during the immune response to endogenous host-related signals.1 NLRP3, as well as other NLRs (such as NLRP1, NLRP6 and NLRC4), signals by forming inflammasome, amultiprotein complex activating the inflammatory caspase-1.2The canonical activation of the NLRP3 inflammasome requires a two-step signaling process, as summarized in Fig. 1: the first step is characterized by an initial priming inducing NLRP3 expression. This first priming step is commonly achieved by the activation of toll-like receptors (TLRs) by either pathogen- or damage-associated molecular patterns (PAMPs or DAMPs); examples of them are lipopolysaccharide (LPS), from the wall of Gram-negative bacteria, or uric acid, respectively. The second activation step is characterized by the sensing of cellular stress signals including K+ and Cl− efflux, Ca2+ influx, lysosomal disruption, metabolic alterations, mitochondrial stress, or radical oxygen species (ROS) production.3,4 This second activation step induces active NLRP3 oligomers that are stabilized by their interaction with negatively charged lipids and/or accessory proteins such as never in mitosis gene a-related kinase 7 (NEK7) or thioredoxin-interacting protein (TXNIP), the negative regulator of the antioxidant thioredoxin system.5 TXNIP is released from thioredoxin in an ROS-sensitive manner and then binds to NLRP3 downstream of the cellular K+ efflux, probably binding to the active NLRP3 oligomer.6 These NLRP3 active oligomers present with the N-terminus pyrin domain exposed and able to recruit the ASC adaptor protein (apoptotic speck-like protein with a caspase activation and recruitment domain). ASC subsequently homo-oligomerizes, forming filaments that culminate in the formation of a large inflammasome aggregate. These ASC filaments recruit and activate the effector protease caspase-1 and this protease subsequently cleaves and maturates the pro-inflammatory cytokines interleukin (IL)-1β and IL-18. Caspase-1 also cleaves the protein gasdermin D (GSDMD), which develops oligomers that form pores in the plasma membrane allowing the cellular release of IL-1β and IL-18, leading to the initiation of the immunogenic type of cell death termed pyroptosis.7 It should be highlighted that NLRP3 activation has multiple additional regulatory elements, such as Src homology-2 domain containing protein tyrosine phosphatase-2 (SHP2).8
Targeting NLRP3 activation is emerging as a reasonable approach for the treatment of different chronic inflammatory, metabolic (including cardiometabolic), and degenerative diseases, where elevated plasma levels of IL-1β and IL-18 are reported.9,10 In particular, in the metabolic context the NLRP3 inflammasome is involved at two levels. First, NLRP3 participates in the molecular mechanism of immune regulation by the diet, as this receptor was firstly described as a critical mechanism of intestinal inflammation in the dextran sulfate sodium (DSS) colitis model.11 Second, NLRP3 is involved in the trained activation of myeloid cells induced by the western diet, contributing to the chronic low-grade inflammation associated with this type of diet. At the same time, the increase of serum cholesterol levels caused by a high-fat diet (HFD) is known to activate the NLRP3 inflammasome.12,13 In addition, other dietary components promote metabolic defects via NLRP3 inflammasome activation. For example, crystallization of uric acid from the diet is a direct inducer of NLRP3 activation and inflammation, increasing also glucose intolerance and insulin resistance, and resulting in hepatic steatosis.14 In this context, research has shown that some natural compounds are able to downregulate the NLRP3 inflammasome activation present in cardiometabolic alterations. In particular, there is an increasing interest in the search for dietary constituents involved in NLRP3 modulation.15–17
2. Dietary polyphenols as cardiometabolic modulators
Polyphenols constitute a diverse group of bioactive plant secondary metabolites encompassing several families, such as flavonoids, phenolic acids, lignans, stilbenes and other polyphenols.18 Dietary sources of polyphenols comprise all food groups of vegetal origin, such as fruits, vegetables, whole grains, nuts, and legumes, as well as derived beverages (wine or juices) and specific food items, e.g., coffee, tea or cocoa.19During the last decades, research on polyphenols has evolved from technology or sensory aspects to health benefits. In this context, it is relevant that, as for any other dietary components, in order to exert their potential beneficial effect, polyphenols must be bioaccessible and bioavailable after ingestion. Thus, it has been established that polyphenols undergo a series of biotransformations during their passage through the intestinal tract to finally reach the systemic circulation.20 It is well known that just a small proportion (5–10%) of the ingested polyphenols may be absorbed into the small intestine. After absorption, the resultant metabolites reach the liver through the portal circulation to finally be distributed to the organs and excreted in the urine. Other non-absorbable polyphenols can reach the colon where they are metabolized by the gut microbiota, allowing the formation of new metabolites, which may have local effects related to colon health but also lead to metabolic outcomes after absorption.21 Due to the low bioavailability of dietary polyphenols, some nanodelivery systems have been explored for improving their solubility and biocompatibility, particularly in the context of the NLRP3 inflammasome.22–25
‘Cardiometabolic alterations’ is a category of health conditions that encompasses a wide spectrum of cardiovascular and metabolic diseases, such as obesity, cardiovascular diseases (CVDs), type 2 diabetes (T2D) and non-alcoholic fatty liver disease (NAFLD), all of them with increasing prevalence in populations following western-style dietary and physical habits.26 Cumulative evidence from in vitro, preclinical, observational and intervention studies sustains a beneficial role of polyphenols in the risk reduction of cardiometabolic alterations27,28 or as adjuvant for inhibiting disease progression.29 Moreover, there is promising evidence on the potential of polyphenols for modulating other pathologies such as certain kinds of cancer30 or cognitive function.31 Nevertheless, research on polyphenols and health outcomes has also pointed to the existence of inter-individual variability in the results due to different reasons,27 as well as to different results when exploring individual foods or polyphenol-rich diets.32
Different mechanisms have been described for polyphenols’ action. Beyond the initial consideration of polyphenols just as radical scavengers, nowadays it is known that their mechanisms of action are much more complex, involving a bidirectional interaction with microbiota21 as well as several molecular activities.27 Indeed, their ability to modulate oxidative stress would be mostly derived from their potential to modulate some cytosolic proteins like Keap1 (Kelch like ECH associated protein 1) and kinase activities, such as protein kinase C, as an indicator of the multilevel regulation of nuclear factor erythroid 2-related factor 2 (Nrf2) mediated transcription.33–36 Also, polyphenols are able to regulate signaling pathways implicated in chronic inflammation, endothelial dysfunction, altered insulin signaling, adipose tissue remodeling or mitochondrial dysfunction.37–40 Other studies, related to gene expression analysis, have demonstrated the influence of polyphenols on the modulation of genes involved in cell adhesion, monocyte transendothelial migration, regulation of the actin cytoskeleton or nuclear factor kappa B (NF-κB) signalling.41–44 An additional mechanism of action of polyphenols for modulating cardiometabolic diseases, as shown by research performed in the last decade, would be related to their ability to modulate the NLRP3 inflammasome, eventually leading to clinical effects in the most common cardiometabolic pathologies. This will be the focus of this review.
3. Current in vivo evidence on polyphenols as NLRP3 inflammasome modulators in cardiometabolic diseases
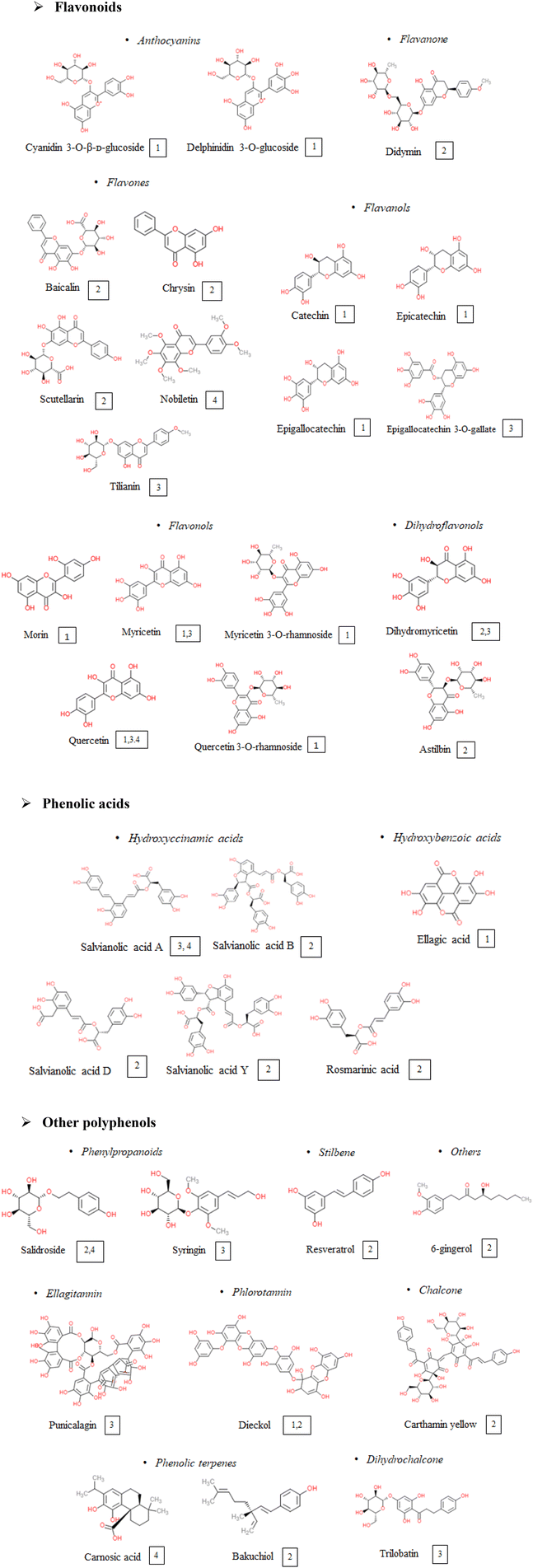
An updated bibliography search on the potential of polyphenols to modulate the NLRP3 inflammasome in the context of cardiometabolic pathologies was carried out. Study selection was based on in vivo (preclinical or clinical) studies, performed either with pure phenolic compounds or with polyphenol-rich extracts or foods. The selected studies explored the effects of about 40 polyphenols, belonging to different polyphenol classes; some of them were tested for several of the selected pathologies, while others were explored only for one of them, as shown in Fig. 2.3.1 Obesity
It has been widely established that an intrinsic characteristic of obesity, besides an excess of fat deposition, is the presence of a state of chronic, low-grade inflammation where multiple molecular pathways are involved, including the NLRP3 inflammasome.45 Six studies46–51 explored the specific role of polyphenols as NLRP3 inflammasome modulators in an obesity context (Table 1). They were all preclinical studies, five in mice and one in rats.50 Obesity was induced by an HFD except for one study, which used a high-fat high-sucrose diet (HFHSD).51 Only one study tested a pure polyphenol, morin,50 while the other ones were focused on extracts from the seaweed Ecklonia cava,46 red raspberry,48 or the tropical fruit jaboticaba,51 and on whole materials from grape seed47 or red raspberry.49 The study duration ranged from 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 to 14
50 to 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 51 weeks. There was a heterogeneity in dose reporting but, interestingly, a study with red raspberry stated that the dose provided was the equivalent in humans to a daily consumption of 150 g of this fruit.49 Finally, two studies evaluated synergistic effects of polyphenols with lactic acid bacteria47 or with polyunsaturated fatty acids.50
51 weeks. There was a heterogeneity in dose reporting but, interestingly, a study with red raspberry stated that the dose provided was the equivalent in humans to a daily consumption of 150 g of this fruit.49 Finally, two studies evaluated synergistic effects of polyphenols with lactic acid bacteria47 or with polyunsaturated fatty acids.50
| Experimental model | Diet and dosage | Polyphenols | Effect | Ref. |
|---|---|---|---|---|
a All mentioned preclinical experiments were performed with male animals.
b Polyphenol content of Ecklonia cava extract can be found at Eo et al., 201746 (https://doi.org/10.1021/acs.jafc.7b00357).
c Flavonoids content of Chardonnay GSL reported by Cho et al., 2018![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 47 (https://doi.org/10.1021/acs.jafc.8b03720).
d LAB is a freeze-dried mixture of Leuconostoc mesenteroides 4 (1 × 1010 CFU) and Lactobacillus kefiri DH5 (1 × 109 CFU). Abbreviations: BMDM, primary bone marrow cells differentiated to macrophages; bw, body weight; Casp1, caspase-1; GS, grape seeds; HFD, high-fat diet; HFHSD, high-fat high-sucrose diet; IL, interleukin; AB, lactic acid bacteria; NLRP3, nucleotide-binding, leucine-rich and pyrin domain-3 protein; PUFAs, polyunsaturated fatty acids; RB, red raspberry extract; RD, raspberry diet; Trim30a, tripartite motif-containing 30A. 47 (https://doi.org/10.1021/acs.jafc.8b03720).
d LAB is a freeze-dried mixture of Leuconostoc mesenteroides 4 (1 × 1010 CFU) and Lactobacillus kefiri DH5 (1 × 109 CFU). Abbreviations: BMDM, primary bone marrow cells differentiated to macrophages; bw, body weight; Casp1, caspase-1; GS, grape seeds; HFD, high-fat diet; HFHSD, high-fat high-sucrose diet; IL, interleukin; AB, lactic acid bacteria; NLRP3, nucleotide-binding, leucine-rich and pyrin domain-3 protein; PUFAs, polyunsaturated fatty acids; RB, red raspberry extract; RD, raspberry diet; Trim30a, tripartite motif-containing 30A.
|
||||
| - C57BL/6J mice | - Control diet | Dieckol and its derivativesb (phlorotannin) | ↓ Caspase-1, IL-1β and IL-18 (kidneys) | 46 |
| - HFD | ||||
| - HFD + Ecklonia cava extract (100–500 mg per kg bw) | ||||
| Duration: 10 weeks | ||||
| - C57BL/6J mice | - Standard diet | Flavanolsc: | ↓ Trim30a (epididymal adipose tissue) | 47 |
| - High-fat diet (HFD) microcrystalline cellulose | - Catechin | |||
| - HFD + grape seed flour (GS) (5%–10%) | - Epicatechin | |||
| - HFD + lactic acid bacteria (LAB)d (10 mL per kg bw) | - Epigallocatechin | |||
| - HFD + GS + LAB | ||||
| Duration: 9 weeks | ||||
| - C57BL/6J mice | - Control diet | Total polyphenols | ↓ Nlrp3, Casp1, Il1b, Il18 (liver) | 48 |
| - Raspberry extract diet (RD) (5% w/w) | ↓ Caspase-1 p20, IL-1β and IL-18 (liver) | |||
| - HFD | ||||
| - HFD + RD | ||||
| Duration: 12 weeks | ||||
| - C57BL/6J mice | - Low-fat diet | Quercetin (flavonol), myricetin (flavonol), ellagic acid (hydroxybenzoic acid), (+)-catechin (flavanol), (−)-epicatechin (flavanol), cyanidin 3-O-β-D-glucoside (anthocyanin) and cyanidin glycoside derivatives (anthocyanin) | ↓ Nlrp3, Il1b (BMDM) | 49 |
| Ex vivo: primary bone marrow cells differentiated to macrophages (BMDM) | - HFD | |||
| - HFD + red raspberry extract (RB) from whole fruit (0.4% w/w) | ||||
| - HFD + RB from seeds (0.1% w/w) | ||||
| - HFD + RB from pulp (0.3% w/w) | ||||
| Duration: 16 weeks | ||||
| - Wistar rats | - Standard diet | Morin (flavonol) | ↓ Caspase-1, IL-1β and IL-18 (liver) | 50 |
| - HFD, high sucrose | ||||
| - HFD + morin (5 mg per kg bw) | ||||
| - HFD + polyunsaturated fatty acids (PUFAs) (1 mL per kg bw) | ||||
| - HFD + morin + PUFAs | ||||
| Duration: 8 weeks | ||||
| - C57BL/6J mice | - HF-high sucrose diet (HFHSD) | Ellagic acid and its derivatives (hydroxybenzoic acid), delphinidin-3-O-glucoside (anthocyanin), cyanidin 3-O-glucoside (anthocyanin), myricetin 3-O-rhamnoside (flavonol), quercetin 3-O-rhamnoside (flavonol) | ↓ Caspase-1, IL-1β and IL-18 (intestine) | 51 |
| - HFHSD + phenolic-rich jaboticaba extract (50–100 mg gallic acid equivalents per kg bw) | ||||
| Duration: 14 weeks | ||||
There was a tendency to find a regulatory effect of the tested polyphenols in NLRP3 inflammasome activation. In particular, the protein levels of caspase-1, IL-1β and IL-18, present in the NLRP3 cascade signal, were significantly decreased, as compared with the obese group, in the kidneys,46 liver50 and intestine51 (based on protein levels in the first case and mRNA expression in the other two). Nevertheless, the levels of NLRP3 measured as protein in the kidneys46 and mRNA expression in the intestine51 remained unaltered as compared with the obese group. However, an increased expression of inflammasome components should not be used as proof of inflammasome activation.
A particular approach was followed in a study that performed an ex vivo experiment with primary bone marrow cells differentiated into bone-marrow derived macrophages (BMDM), finding that red raspberry and its pulp not only decreased the activation of NLRP3 inflammasome, but was also able to reverse IL-1β epigenetic adipogenesis activation,49 which is particularly relevant in the context of obesity.
Finally, it should be mentioned that the described modifications in the NLRP3 inflammasome were concomitant with some improvement in clinical markers, such as improved glucose tolerance, insulin sensitivity and hepatic insulin signaling after raspberry supplementation,48 and the HOMA-IR (homeostatic model assessment for insulin resistance), or body weight, plasma insulin and plasma cholesterol in the study with grape seed flour and kefir.47
3.2 Cardiovascular diseases (CVDs)
Activation of the NLRP3 inflammasome has been shown to be involved in certain key events in CVDs, such as atherosclerosis, myocardial infarction or cardiac remodeling.52 CVDs are the cardiometabolic alterations for which more in vivo studies have explored how polyphenols could modulate the NLRP3 inflammasome, with a total of 18 studies on the topic (17 pre-clinical and 1 clinical), whose main findings are summarized in Table 2.53–70 Regarding the pre-clinical trials, the two most commonly selected animal models corresponded to the middle cerebral artery occlusion/reperfusion model (MCAO/R) in rats56,59,61,64–67 and the myocardial ischemia/reperfusion model (MI/R) in rats or mice.53,55,58,69,70 Other different reported models included: an atherosclerotic study with the apolipoprotein E-deficient (ApoE−/−) mouse model,60 a ventricular arrhythmia model with New Zealand white rabbits,62 an induced cardiac tissue damage (using an anthracycline antineoplastic drug) in male Sprague Dawley (SD) rats,57 a classic permanent coronary occlusion without reperfusion in male SD rats55 and a hepatic ischemia/reperfusion model in mice.68| Preclinical/clinical model | Diet and dosage | Polyphenols | Effect | Ref. |
|---|---|---|---|---|
| a All mentioned preclinical experiments were performed with male animals. Abbreviations: AMI, acute myocardial infarction; ApoE−/−, apolipoprotein E-deficient; AS, atherosclerosis; ASC, apoptotic speck-like protein; bw, body weight; CXCR3; chemokine receptor 3; DIC, cardiotoxicity induced; HEF, heart failure; HI/R, hepatic ischemia/reperfusion; IL, interleukin; IL1R, interleukin 1 receptor type 1; LC, liposomal chrysin; MCAO/R, middle cerebral artery occlusion/reperfusion; MI/R, myocardial ischemia/reperfusion; NF-κB, nuclear factor kappa B; NLRP3, nucleotide-binding, leucine-rich and pyrin domain-3 protein; PC, procyanidins; p-NF-κB, phosphorylated nuclear factor kappa B; Pycard, gene encoding for ASC; RCE, Rhodiola crenulate extract; SD, Sprague Dawley rats; SL, salidroside; TLR, toll-like receptor; TNF-α, tumor necrosis factor alfa. | ||||
| - Sprague Dawley (SD) rats. Myocardial ischemia/reperfusion (MI/R) model | - Control | Total flavones | ↓ NLRP3, caspase-1, IL-1β, IL-6, TNF-α (serum) | 53 |
| - MI/R | ↓ ASC, NLRP3 (myocardial tissue) | |||
| - MI/R + Abelmoschus manihot (L.) Medic extract (40–80 mg per kg bw) 4 h after MIR | ||||
| - C57BL/6J mice | - Control | Resveratrol (stilbene) | ↓ ASC, NLRP3, caspase-1 p20 (myocardial tissue) | 55 |
| MI/R model | - MI/R | ↓ Il1b, Il6, Tnfa (myocardial tissue) | ||
| - Resveratrol (320 mg per kg bw) | ||||
| - MI/R + resveratrol | ||||
| 7 days before MI/R induction | ||||
| - SD rats | - Control diet | Astilbin (dihydroflavonol) | ↓ NLRP3 (brain tissue) | 56 |
| Middle cerebral artery occlusion/reperfusion (MCAO/R) model | - MCAO/R | ↓ IL-1β, IL-18 (serum) | ||
| - MCAO/R + astilbin (40–60–80) mg per kg bw | ||||
| 3 days supplementation before MCAO/R | ||||
| - SD rats | - Control | Dihydromyricetin (dihydroflavonol) | ↓ Pro-caspase-1/caspase-1 (myocardial tissue). | 57 |
| Cardiac tissue damage induced by doxorubicin | - Cardiotoxicity induced (DIC) | ↓ IL-1β, IL-18 (plasma) | ||
| - DIC + dihydromyricetin (100–200 mg per kg bw) | ||||
| 6 weeks supplementation | ||||
| - SD rats | - Control | Scutellarin (flavone) | ↓ NLRP3, caspase-1, IL-1β, IL-18, TNF-α (serum) | 58 |
| MIR/R model | - MI/R | ↓ NLRP3 (myocardial tissue) | ||
| - MI/R + scutellarin (5–10–20 mg per kg bw) | ||||
| 15 min before vascular ligation | ||||
| - SD rats | - Control | Procyanidins (flavanol) | ↓ TLR4-p38, caspase-1, IL-1β (brain tissue) | 59 |
| MCAO/R model | - Procyanidins (PC) (80 mg per kg bw) | |||
| - MCAO/R | ||||
| - MCAO/R + PC (20–40–80 mg per kg bw) | ||||
| 1 h before MCAO | ||||
| - Apolipoprotein E-deficient (ApoE−/−) mice. Atherosclerosis model | - Control | Baicalin (flavone) | ↓ NLRP3, caspase-1, IL-1β, IL-18 (aorta) | 60 |
| - Atherosclerosis (AS) | ↓ Nlrp3, Casp1 (aorta) | |||
| - AS + baicalin (20–50–100 mg per kg bw) | ||||
| 8 weeks supplementation | ||||
| - SD rats | - Control | Carthamin yellow (chalcone) | ↓ p-NF-κB, NLRP3, caspase-1, IL-1β (brain cortex) | 61 |
| MCAO/R model | - MCAO/R | ↓ IL-1β, IL-6 and TNF-α (serum) | ||
| - MCAO/R + carthamin yellow (20–40 mg per kg bw) | ||||
| 2 weeks supplementation before MCAO | ||||
| - New Zealand white rabbits | - Control | Salidroside (phenylpropanoid) | ↓ Il1b, Il6, Tnfa, Il8 (left ventricle) | 62 |
| Ventricular arrhythmia model | - Heart failure (HEF) | |||
| - HEF + Rhodiola crenulate extract (RCE) (125–150–500 mg per kg bw) | ||||
| - HEF + salidroside (SL) (9.5 mg per kg bw) | ||||
| - Control + RCE (250 mg per kg bw) | ||||
| - Control + SL | ||||
| 4 weeks supplementation | ||||
| - SD rats | - Control | Resveratrol (stilbene) | ↓ Pycard, Nlrp3, Casp1 (myocardial tissue) | 63 |
| Myocardial infarction model | - Control + resveratrol (50 mg per kg bw) | ↓ IL-6, TNF-α (serum) | ||
| - Acute myocardial infarction (AMI) | ||||
| - AMI + resveratrol | ||||
| 45 days prior AMI induction | ||||
| - SD rats | - Control | 6-Gingerol (other polyphenols) | ↓ NLRP3, caspase-1, IL-1β, IL-18 (brain tissue) | 64 |
| MCAO/R model | - MCAO/R | |||
| - MCAO/R + 6-gingerol (3.0–6.0 mg per kg bw) | ||||
| 30 min before MCAO | ||||
| - SD rats | - Control | Salvianolic acid B, D, Y and rosmarinic acid (hydroxycinnamic acid) | ↓ NLRP3, ASC, caspase-1, IL-1β (brain cortex) | 65 |
| MCAO/R model | - MCAO/R | ↓ Pycard, Nlrp3, Casp1, Il1b (brain cortex) | ||
| - Salvianolic acids mix (10 mg per kg bw) | ||||
| - MCAO/R + salvianolic acids mix | ||||
| 7 days supplementation after MCAO/R | ||||
| - C57BL/6 mice | - Control | Bakuchiol (phenolic terpene) | ↓ ASC, NLRP3, cleaved-caspase-1 (brain tissue) | 66 |
| MCAO/R model | - MCAO/R | |||
| - MCAO/R + bakuchiol (2.5–5 mg per kg bw) | ||||
| 5 days supplementation before MCAO | ||||
| - SD rats | - Control | Baicalin (flavone) | ↓ ASC, NLRP3, cleaved-caspase-1, IL-1β, IL-18 (brain tissue) | 67 |
| MCAO/R model | - MCAO/R | |||
| - MCAO/R + baicalin (100–200 mg per kg bw) | ||||
| 3 days supplementation before MCAO | ||||
| - C57BL/6 mice | - Control | Chrysin (flavone) | ↓ ASC, NLRP3, caspase-1, IL-1β, IL-18, IL-6, TNF-α (liver) | 68 |
| Hepatic ischemia/reperfusion model (HI/R) | - HI/R | |||
| - Liposomal chrysin (LC) (25 mg per kg bw) | ||||
| - HI/R + LC | ||||
| 16.5 h, 8.5 h, 0.5 h before HIR | ||||
| - SD rats | - Control | Flavonoids and diterpenes | ↓ Caspase-1, IL-1β, IL-18, IL-6 (left ventricle) | 69 |
| MI/R model | - MI/R | |||
| - MI/R + Herba Siegesbeckiae extract (1–2–4 g per kg bw) | ||||
| 7 days pre-treatment before MIR | ||||
| - C57/BL6 mice | - Control | Didymin (flavanone) | ↓ ASC, NLRP3, caspase-1, IL-1β, IL-18, TNF-α (myocardial tissue) | 70 |
| MI/R model | - MI/R | ↓ Il1b, Il18, Tnfa (myocardial tissue) | ||
| - MI/R + didymin (1–2–4 mg per kg bw) | ||||
| 7 days pre-treatment before MIR | ||||
| - Clinical trial | - Control (gin) | Total polyphenols | ↓ TLR4, TLR6, CASP1 (serum) (wine and gin) | 54 |
| Open, acute, randomized, cross-over trial | - Aged wine (928 mg gallic acid equivalents per day) | ↓ TLR2, CXCR3, NLRP3, IL1R (serum) (wine) | ||
| Men at high cardiovascular risk, aged 55–80 | ||||
Different classes of polyphenol were tested, highlighting the use of flavonoid compounds, including: flavonols like astilbin;56 dihydroflavonols like dihydromyricetin;57 procyanidins (polymeric flavanols);59 flavones like scutellarin,58 baicalin,60 and chrysin;68 and flavanones like didymin.70 Other categories of polyphenols included: resveratrol, a stilbene;55,63 phenolic acids like salvianolic acids B, D, Y and rosmarinic acid;65 bakuchiol, a phenolic terpene;66 carthamin yellow, a chalcone,61 and other phenols like 6-gingerol.64 Moreover, extracts obtained from Abelmoschus manihot (L.) Medic and Herba Siegesbeckiae (both containing flavonoids compound) were tested,69 as well as a Rhodiola crenulate extract containing salidroside, a phenylpropanoid.62 The doses of supplementation used in the preclinical studies ranged from 1.0–320 mg kg−1 for pure compounds or a mix of them,55,70 and 0.125–4.0 g kg−1 when extracts were used.62,69 The duration of the studies was different according to the model evaluated; for example, in those in which ischemia/reperfusion were induced the duration ranged from 15 min (ref. 58) to 7 days55,69,70 prior to the induction process. Also, one of the MI/R models involved administration 4 h after reperfusion induction.53 More extensive studies cover a duration of 4–8 weeks.
In the most common model found in the preclinical studies, MCAO/R, brain protein levels of NLRP3 were significantly decreased after supplementation with different pure polyphenols such as astilbin,56 carthamin yellow, a chalcone,61 6-gingerol,64 salvianolic acids and rosmarinic acid,65 bakuchiol66 and baicalin.67 Other upstream markers involved in the NLRP3 activation cascade were also significantly modified in some of these studies; such was the case for TLR4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 59 after procyanidin supplementation and phosphorylated (p-) NF-κB61 after a chalcone was provided. The expression of the adaptor ASC protein and the effector caspase-1 were also significantly downregulated in the brain tissue in some of these studies,59,61,64,66 and dose–response relationships67 were observed. Regarding downstream inflammasome-related signals, IL-1β and IL-18 were downregulated after polyphenol treatment, either in the serum57 or in brain tissue.59,61,64,65,67 Some studies did not find significant modifications in the assessed molecules, such as GSDMD65 or the levels of pro-IL-1β and pro-caspase-1, despite significant decreases in the cleaved active forms.59
59 after procyanidin supplementation and phosphorylated (p-) NF-κB61 after a chalcone was provided. The expression of the adaptor ASC protein and the effector caspase-1 were also significantly downregulated in the brain tissue in some of these studies,59,61,64,66 and dose–response relationships67 were observed. Regarding downstream inflammasome-related signals, IL-1β and IL-18 were downregulated after polyphenol treatment, either in the serum57 or in brain tissue.59,61,64,65,67 Some studies did not find significant modifications in the assessed molecules, such as GSDMD65 or the levels of pro-IL-1β and pro-caspase-1, despite significant decreases in the cleaved active forms.59
Regarding the studies based on the MI/R model, a tendency towards downregulation of the NLRP3 inflammasome pathway due to polyphenol supplementation was observed in the different steps of the signaling cascade. Thus, at the priming step, significant decreases in NLRP3 protein levels in myocardial tissue were observed after supplementation with didymin,70 scutellarin,59 reveratrol55 and flavones.53 At the triggering step, the levels of the adaptor ASC detected in myocardial tissue were significantly decreased in the three studies53,55,70 that measured it, while caspase-1 was decreased in two studies in the myocardial tissue55,70 and in another one in serum.58 In addition, all studies detected a decrease in IL-1β in myocardial tissue or in serum, based on protein levels,53,58,69 mRNA expression,55 or both.70 Interestingly, when several doses were compared, in some studies the effects were observed for all the tested concentrations, with no clear dose–response relationship,53 while in another study only the highest doses were effective.58
A modulation of the NLRP3 inflammasome pathway was also observed in the less common animal models for CVDs that were identified. Thus, several constituents of this signaling cascade were found to be inhibited in myocardial tissue from a model of cardiac tissue damage with doxorubicin,57 the aorta of an atherosclerosis model,60 the left ventricle of a ventricular arrythmia model,62 serum in a myocardial infarction model without reperfusion63 and liver in a hepatic ischemia/reperfusion model.68 All together, these results suggest the ability of dietary polyphenols to exert their action on NLRP3 inflammasome regulation at a multi-tissue scale, thus eventually leading to a risk reduction in CVDs onset or progression.
The reported clinical trial was performed in male subjects at high cardiovascular risk, aged 55–80.70 It comprised the acute administration of aged wine (0.5 g ethanol per kg) with 928 mg of gallic acid equivalents, using gin as a control. The postprandial response was studied for 4 h. Both aged wine and gin led to a significant decrease in TLR4, TLR6 and CASP1, indicating the effect was due to ethanol, while aged wine supplementation specifically caused a significant expression decrease of the TLR2, chemokine receptor 3 (CXCR3), NLPR3 and interleukin 1 receptor type 1 (IL1R1) genes, being thus associated with polyphenol presence.
Overall, at the same time that modifications in NLRP3 activation were observed, there were significant improvements in several clinical markers. For instance, in the MI/R model, improvements in cardiac function parameters of mice and rats, such as stroke work, infarction area, fibrosis area, ejection fraction or fractional shortening were reported in several studies,55,58 being concomitant with improvements in oxidative stress markers in myocardial tissue69 or serum.55 Some of these parameters were also improved in the less common models, such as the fibrotic area after cardiac tissue damage with doxorubicin57 or the ejection fraction in the myocardial infarction model without reperfusion.63 Also, atherosclerotic plaques were reduced following a dose–response relationship after baicalin supplementation in the ApoE−/− mouse model.60 In the case of the MCAO/R approach, significant improvements were observed in specific markers of this pathological model, such as the neurological deficit score,59 infarct rate,61 brain water content,64 or infarction volume,65–67 together with an improvement in serum endogenous antioxidants.61 Notably, dose–response relationships were observed for several of the tested polyphenols.59,61,66,67 No clinical modification was observed in the clinical trial, although that was hard to expect, since it was an acute study based on a single supplementation.
3.3 Type 2 diabetes (T2D)
A decade ago, it was reported that monocyte-derived macrophages (MDMs) from T2D patients had significantly increased mRNA and protein expression of NLRP3 as compared with healthy controls, as well as that the stimulation of MDMs with several molecules caused upregulated IL-1β and IL-18 maturation and secretion, as well as caspase-1 cleavage.71 In this context, a total of 14 in vivo studies dealing with NLRP3 inflammasome modulation in T2D by polyphenols were identified, as shown in Table 3.72–85 All of them were preclinical studies, with streptozotocin (STZ)-induced T2D as the most common one. In particular, 9 of the identified studies used this approach, with diabetic nephropathy72,74,75,82 being the most studied diabetic complication, but with other studies focused on diabetic retinopathy,77 diabetic cardiomyopathy,78,85 diabetic neuropathic pain,80 and atherosclerosis in a diabetic context.84 One study used Zucker diabetic rats as a model, additionally inducing atherosclerosis with an HFD.76 Indeed, an HFD alone was used to cause T2D in two studies in mice,79,83 while one study in rats73 induced T2D through a high-fructose diet. The last study was focused on gestational diabetes.81 Most studies were focused on flavonoids, belonging to different families: flavonols such as quercetin72,73,77 or myricetin;75 dihydroflavanols such as dihydromyricetin;85 flavanols such as epigallocatehin 3-O-gallate,79,82 or procyanidins;81 and flavones such as tilianin.78 Additionally, the phenolic acid salvianolic acid A,77,84 the ellagitannin punicalagin,74 the dihydrochalcone trilobatin83 and the phenylpropanoids syringin78 and salisdroside80 were tested. The duration of the studies ranged from 6 to 16 weeks.73,77,84 The dose range was rather wide, from 0.5 mg per kg bw![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 76 to 250 mg per kg bw.85
76 to 250 mg per kg bw.85
| Experimental model | Diet and dosage | Polyphenols | Effect | Ref. |
|---|---|---|---|---|
| a All mentioned preclinical experiments were performed with male animals, except when otherwise indicated. Abbreviations: ApoE−/−, apolipoprotein E-deficient; ASC, apoptotic speck-like protein; BMDM, primary bone marrow cells differentiated to macrophages; bw, body weight; DMHF, diabetic model high fructose; DNM, diabetic nephropathy model; DRG, dorsal root ganglion; GDM, gestational diabetes mellitus; GSDMD; gasdermin D protein; HMGB1, high-mobility group box-1; HP, lectin-rich protein fraction from horsegram seeds; MyD88; myeloid differentiation primary response-88; NAFLD, non-alcoholic fatty liver disease; NF-κB, nuclear factor kappa B; N-GSDMD, N-terminal gasdermin D protein; NLRP3, nucleotide-binding, leucine-rich and pyrin domain-3 protein; Pycard, gene encoding for ASC; SD, Sprague Dawley rats; STZ, streptozotocin; Syr, syringin; Til, tilianin; TLR4, toll-like receptor-4; TNF-α, tumor necrosis factor alfa; TXNIP, thioredoxin-interacting protein; ZDF, Zucker diabetic fatty rats. | ||||
| - Sprague Dawley rats (SD). Diabetic nephropathy model (DNM) (streptozotocin induced, STZ) | - Diabetic control | Quercetin (flavonol) | ↓ ASC, NLRP3, caspase-1 (kidneys) | 72 |
| - STZ control | ↓ Nlrp3, Casp1 (kidneys) | |||
| - STZ + quercetin (25–50–100 mg per kg bw) | ↓ IL-1β, IL-18 (serum and kidneys) | |||
| - STZ + allopurinol | ||||
| Duration: 7 weeks | ||||
| - SD rats | - Control | Quercetin (flavonol) | ↓ TXNIP, NLRP3, caspase-1, IL-1β (hypothalamus) | 73 |
| Diabetic model (high fructose induced, DMHF) | - DMHF | |||
| - DMHF + quercetin (50–100 mg per kg bw) | ||||
| - DMHF + allupurinol | ||||
| Duration: 6 weeks | ||||
| - C57BL/6J mice | - Control | Punicalagin (ellagitannin) | ↓ NLRP3, caspase-1, GSDMD, IL-1β (kidneys) | 74 |
| DNM (STZ induced) | - STZ (high-fat diet) | |||
| - STZ + punicalagin (20 mg per kg bw) | ||||
| Duration: 8 weeks | ||||
| - Wistar rats | - Control | Myricetin (flavonol) | ↓ NLRP3 (kidneys), IL-1β (plasma) | 75 |
| DNM (STZ induced) | - STZ control (with high-fat diet) | ↓ Pycard, Casp1 (kidneys) | ||
| - STZ + myricetin (Myr) (20 mg per kg bw) | ||||
| - STZ + horsegram protein (HP) (100 mg per kg bw) | ||||
- STZ + Myr + HP (ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
||||
| - STZ + metformin | ||||
| Duration: 6 weeks | ||||
| - Zucker diabetic fatty rats (ZDF) | - Control | Salvianolic acid A (hydroxycinnamic acid) | ↓ NLRP3, caspase-1, IL-1β (aorta) | 76 |
| Diabetic atherosclerosis model (high-fat diet) | - ZDF (high-fat diet) | |||
| - ZDF + salvianolic acid A (0.5–1 mg per kg bw) | ||||
| - ZDF + atorvastatin | ||||
| Duration: 10 weeks | ||||
| - SD rats | - Control | Quercetin (flavonol) | ↓ IL-1 β, IL-18, IL-6, TNF-α (retina) | 77 |
| Diabetic retinopathy (STZ induced) | - STZ | ↓ HMGB1, NLRP3, Hmgb1, Nlpr3 (retina) | ||
| - STZ + quercetin (150 mg per kg bw) | ↓ TLR4, NF-κB (retina) | |||
| - STZ + quercetin + zinc protoporphyrin | ||||
| Duration: 16 weeks | ||||
| - SD rats | - Control | Syringin (phenylpropanoid) | ↓ TLR4, MyD88, NF-κB (Syr + Til) (left ventricles) | 78 |
| Diabetic cardiomyopathy (STZ induced) | - Control + syringin (Syr) (50 mg per kg bw) | Tilianin (flavone) | ↓ NLPR3, IL-1β, TNF-α (Syr, Til, Syr + Til) (left ventricles) | |
| - Control + tilianin (Til) (60 mg per kg bw) | ↓ IL-6 (Til, Syr + Til) (left ventricles) | |||
| - Control + Syr + Til | ||||
| - Diabetic (high-fat diet + STZ) | ||||
| - Diabetic + Syr/Til/Syr + Til | ||||
| Duration: 14 weeks | ||||
| - C57BL/6 mice | - Control | Epigallocatechin 3-O-gallate (flavanol) | ↓ Caspase-1, IL-1β, IL-18 (BMDM) | 79 |
| Ex vivo: primary bone marrow cells differentiated to macrophages (BMDM) | - Diabetic (high-fat diet) | |||
| Diabetic model (high fat induced) | - Diabetic + EGCG (50 mg per kg bw) | |||
| Duration: 12 weeks | ||||
| - SD rats | - Control | Salidroside (phenylpropanoid) | ↓ TXNIP, pro-caspase-1, pro- IL-1β, IL-18, NLRP3, caspase-1 (dorsal root ganglions) | 80 |
| Diabetic neuropathic pain (STZ induced) | - Diabetic (high-fat diet + STZ) | |||
| - Diabetic + salidroside (50–100 mg per kg bw) | ||||
| Duration: 6 weeks | ||||
| - C57BL/6 female mice | - Control | Procyanidins (flavanol) | ↓ Pro-IL-1β, ASC, NLRP3, caspase-1, IL-1β (liver) | 81 |
| Gestational diabetes mellitus (GDM) model | - Diabetic (high-fat high-sucrose diet) | ↓ IL-6, IL-7, TNF-α (serum) | ||
| - Diabetic + procyanidins (PA) (28 mg per kg bw) | ↓ Il6, Il7, Tnfa (liver) | |||
| - Diabetic + PA + Abs | ||||
| Duration: 7 weeks | ||||
| - SD rats | - Control | Epigallocatechin 3-O-gallate (flavanol) | ↓ IL-1β, IL-18, IL-6, TNF-α (kidneys) | 82 |
| DNM (STZ induced) | - Diabetic (high-fat diet + STZ) | |||
| - Diabetic + epigallocatechin 3-O-gallate (40–80 mg per kg bw) | ||||
| Duration: 8 weeks | ||||
| - C57BL/6J mice | - Control | Trilobatin (dihydrochalcone) | ↓ NLRP3, caspase-1, GSDMD, N-GSDMD, IL-1 β, IL-18 (liver) | 83 |
| NAFLD in a diabetic model (high fat induced) | - Diabetic (high-fat diet + STZ) | (High dose dependence) | ||
| - Diabetic + trilobatin (20–60 mg per kg bw) | ||||
| - Diabetic + metformin | ||||
| Duration: 10 weeks | ||||
| - ApoE−/− mice | - Control | Salvianolic acid A (hydroxyccinamic acid) | ↓ ASC, NLRP3, GSDMD (aortic sinus) | 84 |
| Diabetic atherosclerosis model (STZ induced) | - STZ control | |||
| - STZ + salvianolic acid A (10–20 mg per kg bw) | ||||
| Duration: 16 weeks | ||||
| - C57BL/6 mice | - Control | Dihydromyricetin (dihydroflavonol) | ↓ NLRP3, IL-1β (myocardium) | 85 |
| Diabetic cardiomyopathy model (STZ induced) | - STZ control | |||
| - STZ + dihydromyricetin (250 mg per kg bw) | ||||
| - Control + dihydromyricetin | ||||
| Duration: 12 weeks | ||||
Regarding STZ-induced T2D models, as the most common one, a clear tendency towards an inhibition of the NLRP3 signaling cascade – as evidenced by a decrease in the concentrations of its markers in different tissues – due to polyphenol supplementation was observed. Thus, NLRP3 was decreased in the kidneys,72,74,75 retina,77 cardiac tissue,78,85 aortic sinus84 and dorsal root ganglion;80 ASC levels were reduced in the kidneys,72 retina,77 and aortic sinus;84 caspase-1 in the kidneys,72,74,75 and dorsal root ganglion;80 IL-1β or IL-18 in serum,72 plasma,75 kidneys,72,74 retina,77 cardiac tissue78,85 and dorsal root ganglion;80 and other cytokines in all the mentioned tissues. The inactive forms of caspase-1 and IL-1β were also decreased in the dorsal root ganglia after polyphenol supplementation.80 In some studies, gene expression evaluation was performed at the same time as the determination of protein concentration, finding similar results.72,75,77 It is also worth mentioning that a dose–response effect was observed for quercetin effects in a diabetic nephropathy model,72 as well as the fact that synergistic effects were found between syringin and tilianin in a diabetic cardiomyopathy model.78
In the other T2D models, a similar downregulation of NLRP3 inflammasome components due to polyphenol supplementation was observed. Thus, the concentrations of the main signals of the pathway were decreased in several tissues: TXNIP in the hypothalamus;73 NLRP3 in the hypothalamus,73 aortic tissue,76 and liver;81,83 caspase-1 in the hypothalamus,73 aortic tissue,76 and liver;81,83 and IL-1β, IL-18 or TNF-α in the hypothalamus,73 aortic tissue,76 serum,81 liver80,81,83 and macrophages.79 Also, other cytokines, such as IL-6 and IL-7, were decreased in the serum of animals in a gestational diabetes model.81
It should be mentioned that four of the above studies compared the inhibitory effects of polyphenols with existing NLRP3 inhibitors – allopurinol, metformin, and atorvastatin – in a type 2 diabetes mellitus context.86 In that sense, Wang et al.,72 and Zhang et al.,73 used allopurinol with Sprague Dawley rats for a diabetic nephropathy model induced by streptozotocin, and for a diabetic model induced by a high-fructose diet, respectively. They used allopurinol as a positive control group in contrast with quercetin supplementation. Their findings suggest that quercetin responds in a similar way to allopurinol for the reduction of some protein levels like ASC, NLRP3, caspase-1, IL-1β and IL-18 measured in the kidneys, as well as levels of IL-1β and IL-18 found in the serum,72 and TXNIP, caspase-1 and IL-1β measured in the hypothalamus.73 In the same context, Lalitha et al.,75 found that levels of NLRP3 protein expression in the liver tissue of diabetic Wistar rats, as well as gene expressions of Pycard and Casp1, and IL-1β levels in blood plasma, were similarly modified by metformin and myricetin. Also, Ma et al.76 employed atorvastatin as a control positive group in Zucker diabetic fatty rats with atherosclerosis. Their results showed that salvianolic acid A decreased the expression levels of NLRP3, caspase-1 and IL-1β in the aortic tissues in a similar way to atorvastatin.
Finally, inhibition of the NLRP3 inflammasome was accompanied by several improvements in clinical markers, first in those ones directly related to glucose hemostasis, such as blood glucose or insulin, hemoglobin A1c (Hb1Ac) or fructosamines,75,83,84 but also in markers associated with diabetic complications, such as blood urea nitrogen, serum creatinine and kidney lipid profile in the case of diabetic nephropathy;72,74 thickness of the retina in retinopathy;77 echocardiogram parameters in cardiomyopathy;78 and paw withdrawal threshold and paw withdrawal latency in neuropathic pain.80 Overall, the downregulation of the NLRP3 inflammasome after supplementation with dietary polyphenols had an adjuvant effect in the multiple disturbances present in a T2D situation.
3.4 Non-alcoholic fatty liver disease (NAFLD)
Although NLRP3 inflammasome activation plays a key role in all the selected cardiometabolic alterations, it plays an unequivocal role in the case of NAFLD, since it has been suggested that activation of the TLR4/NLRP3 pathway and the subsequent NLRP3 priming is involved in the onset of NAFLD and in the progression of this pathology to the characteristic fibrosis found in nonalcoholic steatohepatitis (NASH).87,88Table 4 summarizes the main findings from the seven studies89–95 that were identified dealing with the modulation of the NLRP3 inflammasome by polyphenols regarding NAFLD. They comprised six preclinical studies and a clinical trial in NAFLD patients.95 Regarding the selected animal models, one study was performed in rats fed an HFD,89 while the other ones used the same diet either in wild type mice,90–93 in ApoE−/− mice,94 in C57BLKS/Leprdb mice93 or in MARCKS (myristoylated alanine-rich C-kinase substrate)-deficient mice.92 The studies tested phenolic compounds belonging to different categories: salisdroside (tyrosol glucoside),93 a phenylpropanoid; carnosic acid, a phenolic terpene,92 and salvianolic acid A, a phenolic acid;89 quercetin, a flavonol;91 and nobiletin, a flavone.94 The distribution in nature of these compounds is quite different. Quercetin is an ubiquitous polyphenol; carnosic acid and nobiletin are characteristic of specific foods (rosemary and orange, respectively); and the other ones are found in some medicinal plants. Also, a cranberry extract90 and an anthocyanin-rich extract95 were tested. The study duration in the animal models ranged from 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 89,92,93 to 24 weeks,94 while the clinical trial lasted 12 weeks.95 Finally, the doses used in the preclinical models ranged from 8 to 200 mg kg−1.89,94 Participants in the clinical trial received a daily dose of 320 mg of anthocyanins.95
89,92,93 to 24 weeks,94 while the clinical trial lasted 12 weeks.95 Finally, the doses used in the preclinical models ranged from 8 to 200 mg kg−1.89,94 Participants in the clinical trial received a daily dose of 320 mg of anthocyanins.95
| Preclinical/clinical model | Diet/protocol and dosage | Polyphenols | Effect | Ref. |
|---|---|---|---|---|
| a All mentioned preclinical experiments were performed with male animals. ApoE−/−, apolipoprotein E-deficient; bw, body weight; Casp1, caspase 1 gene; HFD, high-fat diet; IFN-γ, interferon gamma; IL, interleukin; Leprdb (db/db), mouse with a leptin receptor mutation; LFD, low-fat diet; MARCKS, myristoylated alanine-rich C-kinase substrate; MT, MARCKS-deficient mice; NAFLD, non-alcoholic fatty liver disease; NF-κB, nuclear factor kappa B; NLRP3, nucleotide-binding, leucine-rich and pyrin domain-3 protein; PBMCs, peripheral blood mononuclear cells; RD, regular diet; Rela: transcription factor NF-κB p65; SA, salvianolic acid A; TNF-α, tumor necrosis factor alfa; TLR4, toll-like receptor-4; TXNIP, thioredoxin-interacting protein; WT, wild-type mice. | ||||
| - Sprague Dawley rats | - Control with regular diet (RD) | Salvianolic acid A (hydroxycinnamic acid) | ↓ TXNIP, Txnip (liver) | 89 |
| High-fat diet (HFD) induced obesity model | - SD + salvianolic acid A (SA) (16 mg per kg bw) | |||
| - HFD | ||||
| - HFD + SA (8–16 mg per kg bw) | ||||
| - Duration: 8 weeks | ||||
| - C57BL/6J mice | - HFD | Anthocyanins and proanthocyanidins | ↓ Tlr4, Rela (liver) | 90 |
| HFD-induced obesity model | - HFD + cranberry extract (0.8% w/w) | ↓ Nlrp3, Txnip, Casp1 (liver) | ||
| - Duration: 21 weeks | ↓ TNF-α (liver) | |||
| - C57BL/6J mice | - Low-fat diet (LFD) | Quercetin (flavonol) | ↓ TLR4, Tlr4 (liver) | 91 |
| HFD-induced obesity model | - LFD + quercetin (0.05% w/w) | ↓ NF-κB p65 subunit (liver) | ||
| - HFD | ↓ Il16, Tnfa (liver) | |||
| - HFD + quercetin (0.05% w/w) | ||||
| - Duration: 16 weeks | ||||
| - C57BL/6J wild-type mice (WT) | - Control RD (WT and MT) | Carnosic acid (phenolic terpene) | ↓ NLRP3, caspase-1 (liver) | 92 |
| - MARCKS-deficient mice (MT) | - HFD (WT and MT) | ↓ Il1b, Il18, Tnfa, Il2, Il4, Il6, Il12, Ifng (liver) | ||
| HFD-induced obesity model | - WT + HFD + carnosic acid (CA) (15–30 mg per kg bw) | ↓ IL-1β, IL-18, TNF-α, IL-2, IL-4, IL-6, IL-12, IFN-γ (serum) | ||
| - Duration: 8 weeks | (No clear dose–response relationship) | |||
| - C57BL/6J WT | - Control RD (WT and db/db) | Salidroside (phenylpropanoid) | ↓ TXNIP, pro-caspase-1, caspase-1, pro-IL-lβ, IL-1β (liver db/db mice) | 93 |
| - C57BLKS/Leprdb (db/db) mice | - HFD (WT) | ↓ Casp1, Il1b, Tnfa (liver WT) | ||
| HFD-induced obesity model | - HFD + salidroside (100 mg per kg bw) | ↓ IL-1β (serum WT) | ||
| - db/db + salidroside (100 mg per kg bw) | ||||
| - Duration: 8 weeks | ||||
| - ApoE−/− mice | - Control RD | Nobiletin (flavone) | ↓ NLRP3 (low and medium dose) (liver) | 94 |
| HFD-induced obesity model | - HFD | ↓ IL-1β (low and high dose) (liver) | ||
| - HFD + nobiletin (50–100–200 mg per kg bw) | ||||
| - Duration: 24 weeks | ||||
| - Clinical trial | - Control group (healthy subjects, n = 69) | Anthocyanins | ↓ CASP1, IL1B, IL18 (PBMCs) | 95 |
| Randomized, double-blind, parallel study | - NAFLD patients + anthocyanins l capsule (320 mg) | ↓ IL-1β, IL-18 (plasma) | ||
| NAFLD patients and healthy subjects (25–65 years old) | - NAFLD patients + placebo (320 mg) | |||
| - Duration: 12 weeks | ||||
The effects on NLRP3 activation were mostly measured in the liver, since it was the explored target. The results, based either on protein levels or mRNA expression, clearly showed a tendency for a regulation of NLRP3 inflammasome components expression by polyphenols, at least partially restoring the disturbances caused by an HFD. This regulation first affected the priming step in NLRP3 inflammasome activation. As previously explained, the canonical pathway for NLRP3 priming is the one mediated by TLRs. In this context, it was shown that quercetin was able to revert the dysbiosis by TLR4 activation, which ultimately would have beneficial effects on NAFLD pathology, through a gut–liver axis.91 Similarly, the variations observed in Tlr4 gene expression after supplementation with a cranberry extract led the authors to the conclusion that the effects observed in several components of the NLRP3 inflammasome were partially mediated by a regulation of the TLR4/NLRP3 pathway.90 Also, a decrease in hepatic TXNIP, as evidenced by mRNA expression and protein levels, was found after diet supplementation with salvianolic acid A90 and with salisdroside.93 TXNIP liver gene expression was also inhibited after diet supplementation with cranberry extract.90 It should be mentioned that, although the most common pathway to induce the TXNIP/NLRP3 complex is derived from an ROS excess, it has been shown that this pathway may be also independently activated in the presence of high glucose and insulin levels. Thus, the inhibition of the pathway observed in these NAFLD animal models has relevant implications regarding T2D, considering the interconnection between both pathologies. Downstream in the priming process, hepatic nuclear and cytosolic measurements of the protein level of transcription factor p65 showed that its translocation to the nucleus was inhibited after supplementation with quercetin.91
Following the inflammasome cascade, mRNA expression of Nrlp3 was significantly decreased in hepatic tissues after supplementation with cranberry extract90 and the same tendency was found with Casp1 after the same supplementation and also after providing salidroside.93 Similarly, carnosic acid significantly inhibited hepatic protein levels for NLRP3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 92 and caspase-1, as well as after supplementation with salidrose.92,93 The final step in the cascade is the release of IL-1β and IL-18 from their inactive forms. Thus, hepatic levels of pro-IL-1β were decreased after supplementation with salisdroside,93 while Il1b expression was significantly decreased after supplementation with salisdroside93 and carnosic acid,92 and protein levels were lower than in the HFD group after both supplementations and also after providing nobiletin.94 IL-18 was significantly decreased, as measured both by gene expression and protein levels, after carnosic acid supplementation.92 Furthermore, downstream in the NF-κB cascade, carnosic acid inhibited the release of TNF-α, IL-2, IL-4, IL-6, IL-12 and interferon gamma (IFN-γ) (all measured at the level of gene expression and protein);92 quercetin decreased the mRNA levels of Tnfa and Il6;91 and cranberry extract reduced the protein expression of TNF-α.90 It should also be mentioned that supplementation with the anthocyanin cyanidin 3-O-glcuoside in an animal model of alcoholic steatohepatitis (not fulfilling the inclusion criteria of this review, dealing with NAFLD) also observed a significant decrease in multiple markers of the NLRP3 cascade.96
92 and caspase-1, as well as after supplementation with salidrose.92,93 The final step in the cascade is the release of IL-1β and IL-18 from their inactive forms. Thus, hepatic levels of pro-IL-1β were decreased after supplementation with salisdroside,93 while Il1b expression was significantly decreased after supplementation with salisdroside93 and carnosic acid,92 and protein levels were lower than in the HFD group after both supplementations and also after providing nobiletin.94 IL-18 was significantly decreased, as measured both by gene expression and protein levels, after carnosic acid supplementation.92 Furthermore, downstream in the NF-κB cascade, carnosic acid inhibited the release of TNF-α, IL-2, IL-4, IL-6, IL-12 and interferon gamma (IFN-γ) (all measured at the level of gene expression and protein);92 quercetin decreased the mRNA levels of Tnfa and Il6;91 and cranberry extract reduced the protein expression of TNF-α.90 It should also be mentioned that supplementation with the anthocyanin cyanidin 3-O-glcuoside in an animal model of alcoholic steatohepatitis (not fulfilling the inclusion criteria of this review, dealing with NAFLD) also observed a significant decrease in multiple markers of the NLRP3 cascade.96
The clinical study95 in NAFLD patients deserves a specific comment. It was a randomized, double-blind, parallel study where subjects (n = 37 per group) daily consumed a pill with either the anthocyanin extract or the placebo. After verifying in a previous case-control study that peripheral blood mononuclear cells (PBMCs) were able to discriminate NAFLD patients by measuring NLRP3 inflammasome gene expression, thus avoiding liver biopsies, in this intervention study it was found that the supplementation originated a significant decrease in CASP1, IL1B, IL18, at the same time that significantly lower plasma levels of IL-1β and IL-18 were observed.
Some of the studies tested the compounds at different doses. The modifications in cytokine levels observed after supplementation with carnosic acid were achieved in some cases only at 30 mg kg−1 and other times with both 15 and 30 mg kg−1, without a clear dose–response dependency.92 The study with nobiletin found some unexpected results, such as the fact that NLRP3 hepatic protein levels were decreased by the low and medium dose but not by the high one, while in the case of IL-1β low and high doses were effective and not the intermediate concentration.94
All the studies reported clinical effects besides the modifications in the NLRP3 inflammasome. They comprised improvements in hepatic lipid deposition,80,81,83,84 insulin sensitivity,92,93 NAFLD score,89,90,92,94 SOD (superoxide dismutase) levels89,93 or MDA (malondialdehyde) concentration.89,93 It is particularly relevant that lipid accumulation and insulin sensitivity were improved in MARCKS-deficient mice, an animal model of liver fibrosis,92 although in some cases contradictory results were obtained, for instance regarding obesity.90,93 Moreover, besides the general beneficial consequences of decreasing the grade of inflammation, the effects observed in these studies on the NLRP3 inflammasome are relevant for NAFLD progression itself, since IL-1β is particularly involved in the pathogenesis of NAFLD and NASH. Indeed, in comparison with wild-type animals, Nlrp3−/− or Casp1−/− animals were protected from feeding-induced hepatomegaly, hepatic steatosis and liver fibrosis.97 However, in the clinical study analyzed here, despite the already mentioned variations in NLRP3 inflammasome components, the only biochemical modification that was observed was a decrease in liver ALT (alanine transaminase), without effects on plasma lipid profile or glucose.95
4. Perspectives for research
Overall, the critical assessment of scientific evidence performed in this review shows there is cumulative in vivo evidence on the ability of dietary polyphenols to modulate the NLRP3 inflammasome in the context of cardiometabolic alterations, by downregulating the expression, from upstream to downstream, of the key molecules involved in the signaling pathway. Moreover, although the precise molecular mechanisms through which polyphenols inhibit NLPR3 activation still remain unclear, three possible mechanisms have been proposed related to polyphenols’ action against NLRP3 activation in canonical and noncanonical inflammasome pathways, as follow: (1) inhibition of NF-κB priming in the priming step, (2) inhibition of NLPR3 inflammasome assembly in the activation step and (3) inhibition of NLPR3 inflammasome activation through the redox state and cellular metabolism.98 However, from the analysis of current knowledge on the topic several considerations emerge.First, regarding the tested, doses, a high disparity has been observed. For instance, in the case of CVD, the differences in the selected doses were up to 200-fold between the lowest and the highest one, and this was up to a 500-fold difference for studies focused on T2D. It is evident that this makes it difficult to reach a conclusion on which polyphenol doses would be the most effective ones. Indeed, some studies reported dose–response relationships, while others found the same effects for all the doses – this may be related to the fact that in some studies the lowest tested doses were already high. Moreover, this connects with the need to use doses in the animal studies of nutritional relevance. In this sense, when extrapolating the dose range used in the studies (about 1–250 mg per kg bw) to humans, based on the reported conversions for each animal species,99 they would correspond to a daily dose for an adult person weighing 60 kg of about 2.4–2400 mg per kg bw. Taking into account that several studies have reported a daily polyphenol intake about 1 g per day,100 this means that, for some of the selected polyphenols, the dose provided would be in a nutritional range, other ones would be equivalent to an additional supplementation and, in some cases, the tested doses of a single polyphenol may be risky, considering that the person's diet would include other dietary sources of these compounds and it is known that high polyphenol doses may have adverse effects,101 probably due to a transformation into pro-oxidant entities. For instance, one of the most studied polyphenols reported in this review was quercetin used in animal models for diabetes,72,73,77 and the doses provided were not on a nutritional scale: they would correspond to a daily intake of 240–1500 mg of quercetin in an adult person (after conversion) when, for instance, red onion, one of the most common sources of quercetin, has a quercetin content of 1 mg per 100 g.102 In contrast, for punicalagin, an ellagitannin tested in a mouse diabetic nephropathy model,74 we found that the proposed dose could be reasonable for an adult person (97 mg after conversion from an animal dose): this polyphenol is present in pomegranate juice (43.6 mg per 100 g),102 so a glass of this juice (225 mL) would be enough to reach the proposed dose. Similarly, the dose of didymin provided in a mouse model of MI/R (equivalent to 5–20 mg in an adult person) could be achieved by consuming orange juice (with a didymin content of 5.9 mg per 100 mL).102
Second, among the wide diversity of chemical entities belonging to the polyphenol family, only a minor fraction has been tested in the identified studies (about 40 different entities). Moreover, while some of them are ubiquitous compounds present in many food sources (such as flavanols or quercetin), other ones were very specific compounds, found just in some plant sources (e.g., salidroside or salvianolic acid A). Indeed, this would lead to two different approaches in the study of polyphenols as NLRP3 inflammasome modulators. One would be related to their potential to exert these actions through the intake of plant-based food diets and the other would be supplementation with specific compounds not found in common foods. Nevertheless, the role of many dietary polyphenols in the modulation of the NLRP3 inflammasome remain to be tested, as shown, for instance, by the scarce number of phenolic acids assessed in the reviewed in vivo studies. This led to the fact that most of the in vivo studies evaluate a decrease of the expression for different components of the NLRP3 inflammasome, and this could not be directly related to effective inflammasome formation and signaling. Furthermore, overall expression in complex tissues and organs could overlook the expression in particular cells, and it might be that in myeloid or other relevant cells, the NLRP3 inflammasome could be activated as normal in certain cells albeit with a general decrease of NLRP3 expression in a tissue. Additionally, it should be considered that most of the inflammasome components are also necessary for the formation of other inflammasomes (such as NLRP1, NLRC4, NLRP6, or pyrin), and therefore not only NLRP3, but also other inflammasomes could be potentially affected by polyphenols and this should be further investigated. Additional molecular studies are needed to clarify the mechanism of action of polyphenols modulating the activation (not expression) of the NLRP3 (and other) inflammasomes. In this regard, the polyphenol punicalagin does not affect NLRP3 inflammasome expression or activation in vitro but interferes with pyroptosis and IL-1β release.103 Also, the clinical benefits of polyphenol supplementation in animal models cannot be directly attributed to a decrease in the expression of NLRP3 inflammasome components without a formal demonstration of the activation of the inflammasome complex and its implication in the studied pathology.
Another relevant point is the need to further evaluate the potential interactions (additive, antagonistic, or synergistic) that may take place between polyphenols and dietary constituents as regards the modulation of the NLRP3 inflammasome, since they are not consumed alone, but entrapped in a food matrix with many other constituents. It has been mentioned that a few studies have explored this aspect,47,50 but it has been scarcely addressed. In particular, these interactions should be explored regarding joint intakes of probiotics and polyphenols, since it is known that the interactions of these compounds with microbiota is a key step for generating the eventually bioactive metabolites,104 including their interaction with the NLRP3 inflammasome. Also, the accumulation of more scientific evidence regarding polyphenols and the NLRP3 inflammasome will allow us to establish whether certain polyphenols differentially modulate preferentially a certain step of the cascade or have a stronger effect in one of the studied cardiometabolic alterations, such a classification cannot yet be performed.
Finally, a key aspect to be further explored is the development of clinical trials aiming to evaluate the NLRP3 inflammasome, since in this review only two trials in humans were identified. It should be noted that measurements of gene expression in the NLRP3 signaling cascade, as well as measurements of direct NLRP3 inflammasome formation, may be performed in PBMCs, an approach that is much less invasive than biopsies and provides consistent results,90,91,95 being therefore a feasible alternative. Besides, one of the clinical trials that was found provided the participants with polyphenols in a capsule and another one with a very specific food, such as wine. Future studies should test different dietary sources of polyphenols, keeping the range of realistic doses that may be incorporated into a habitual diet. At the same time, since it has been suggested that polyphenol effects are achieved by a consumption of whole polyphenol-rich diets and not so much by specific foods,105 such an approach should be tested, nevertheless, a clear definition of a polyphenol-rich diet has not yet been provided.106 Also, inter-individual variability should be explored in such studies, as a commonly described characteristic related to polyphenol response.27 Overall, an extension of clinical trials in participants with the most common cardiometabolic alterations is needed to complete the validation of dietary polyphenols as modulators of the NLRP3 inflammasome, as a molecular process eventually impacting the clinical markers of those pathologies.
5. Conclusions
A relevant number of in vivo papers, mostly preclinical studies, have explored the potential of dietary polyphenols for downregulating the components of NLRP3 inflammasome expression, in the context of cardiometabolic alterations (obesity, CVD, T2D and NAFLD). In several animal models for the mentioned conditions, a clear tendency towards a decrease in the expression of the whole signalling pathway was observed, commonly showing a dose–response effect; these modifications were concomitant with clinical improvements in the pathologies. Nevertheless, the diversity of doses used, the disparity in the polyphenol structures tested, the lack of mechanistic data and, particularly, the scarce clinical trials, show the need to develop further research on the topic.Author contributions
Conceptualization: M. V. and J. P; writing – original draft preparation: J. J. M. G., P. P.; L. J., S. S., J. P. and M. V.; writing – review and editing: M. V. and J. P. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Acknowledgements
Marisol Villalva holds a postdoctoral fellow ‘Margarita Salas’ funded by the Spanish Ministry of Universities and the Autonomous University of Madrid (CA1/RSUE/2021-00588). Maylis Renard is thanked for providing support with the literature search. Jara Pérez-Jimenez and Pablo Pelegrin would like to thank the Teofilo Hernando Foundation and Royal Spanish Academy of Medicine Young Researcher Forum. Laura Jaime and Susana Santoyo would like to thank the Spanish Government (project: PID2019-110183RB-C22/AEI/10.13039/501100011033).References
- S. Tartey and T. D. Kanneganti, Differential role of the NLRP3 inflammasome in infection and tumorigenesis, Immunology, 2019, 156(4), 329–338 CrossRef CAS PubMed.
- F. Martinon, K. Burns and J. Tschopp, The Inflammasome: A molecular platform triggering activation of inflammatory caspases and processing of pro IL-β, Mol. Cell., 2002, 10(2), 417–426 CrossRef CAS PubMed.
- K. V. Swanson, M. Deng and J. P. Ting, The NLRP3 inflammasome: molecular activation and regulation to therapeutics, Nat. Rev. Immunol., 2019, 19(8), 477–489 CrossRef CAS PubMed.
- N. Kelley, D. Jeltema, Y. Duan and Y. He, The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation, Int. J. Mol. Sci., 2019, 20(13), 3328 CrossRef CAS PubMed.
- Y. He, M. Zeng, D. Yang, B. Motro and G. Núñez, NEK7 is an essential mediator of NLRP3 activation downstream of potassium efflux, Nature, 2016, 530, 354–357 CrossRef CAS PubMed.
- R. Zhou, A. Tardivel, B. Thorens, I. Choi and J. Tschopp, Thioredoxin-interacting protein links oxidative stress to inflammasome activation, Nat. Immunol., 2010, 11, 136–140 CrossRef CAS PubMed.
- M. M. Gaidt and V. Hornung, Pore formation by GSDMD is the effector mechanism of pyroptosis, EMBO J., 2016, 35(20), 2167–2169 CrossRef CAS PubMed.
- W. Guo, W. Liu, Z. Chen, Y. Gu, S. Peng, L. Shen, Y. Shen, X. Wang, G. S. Feng, Y. Sun and Q. Xu, Tyrosine phosphatase SHP2 negatively regulates NLRP3 inflammasome activation via ANT1-dependent mitochondrial homeostasis, Nat. Commun., 2017, 8(1), 2168 CrossRef PubMed.
- A. I. Suceveanu, L. Mazilu, N. Katsiki, I. Parepa, F. Voinea, A. Pantea-Stoian, M. Rizzo, F. Botea, V. Herlea, D. Serban and A. P. Suceveanu, NLRP3 inflammasome biomarker-could be the new tool for improved cardiometabolic syndrome outcome, Metabolites, 2020, 10(11), 448 CrossRef CAS PubMed.
- E. Braunwald, Biomarkers in heart failure, N. Engl. J. Med., 2008, 358, 2148–2159 CrossRef CAS PubMed.
- C. Bauer, P. Duewell, C. Mayer, H. A. Lehr, K. A. Fitzgerald, M. Dauer, J. Tschopp, S. Endres, E. Latz and M. Schnurr, Colitis induced in mice with dextran sulfate sodium (DSS) is mediated by the NLRP3 inflammasome, Gut, 2010, 59(9), 1192–1199 CrossRef CAS PubMed.
- A. Christ, P. Günther, M. A. R. Lauterbach, P. Duewell, D. Biswas, K. Pelka, C. J. Scholz, M. Oosting, K. Haendler, K. Baßler, K. Klee, J. Schulte-Schrepping, T. Ulas, S. J. C. F. M. Moorlag, V. Kumar, M. H. Park, L. A. B. Joosten, L. A. Groh, N. P. Riksen, T. Espevik, A. Schlitzer, Y. Li, M. L. Fitzgerald, M. G. Netea, J. L. Schultze and E. Latz, Western Diet Triggers NLRP3-Dependent Innate Immune Reprogramming, Cell, 2018, 172(2), 162–175 CrossRef CAS PubMed.
- P. D. Cani, J. Amar, M. A. Iglesias, M. Poggi, C. Knauf, D. Bastelica, A. M. Neyrinck, F. Fava, K. M. Tuohy, C. Chabo, A. Waget, E. Delmée, B. Cousin, T. Sulpice, B. Chamontin, J. Ferrières, J. F. Tanti, G. R. Gibson, L. Casteilla, N. M. Delzenne, M. C. Alessi and R. Burcelin, Metabolic endotoxemia initiates obesity and insulin resistance, Diabetes, 2007, 56(7), 1761–1772 CrossRef CAS PubMed.
- X. Wan, C. Xu, Y. Lin, C. Lu, D. Li, J. Sang, H. He, X. Liu, Y. Li and C. Yu, Uric acid regulates hepatic steatosis and insulin resistance through the NLRP3 inflammasome-dependent mechanism, J. Hepatol., 2016, 64, 925–932 CrossRef CAS PubMed.
- W. Guo, Y. Sun, W. Liu, X. Wu, L. Guo, P. Cai, X. Wu, X. Wu, Y. Shen, Y. Shu, Y. Gu and Q. Xu, Small molecule-driven mitophagy-mediated NLRP3 inflammasome inhibition is responsible for the prevention of colitis-associated cancer, Autophagy, 2014, 10(6), 972–985 CrossRef PubMed.
- W. Liu, W. Guo, Y. Zhu, S. Peng, W. Zheng, C. Zhang, F. Shao, Y. Zhu, N. Hang, L. Kong, X. Meng, Q. Xu and Y. Sun, Targeting Peroxiredoxin 1 by a Curcumin Analogue, AI-44, Inhibits NLRP3 Inflammasome Activation and Attenuates Lipopolysaccharide-Induced Sepsis in Mice, J. Immunol., 2018, 201(8), 2403–2413 CrossRef CAS PubMed.
- Y. Zhu, Z. Ouyang, H. Du, M. Wang, J. Wang, H. Sun, L. Kong, Q. Xu, H. Ma and Y. Sun, New opportunities and challenges of natural products research: When target identification meets single-cell multiomics, Acta Pharm. Sin. B, 2022, 12(11), 4011–4039 CrossRef CAS PubMed.
- V. Neveu, J. Perez-Jiménez, F. Vos, V. Crespy, L. du Chaffaut, L. Mennen, C. Knox, R. Eisner, J. Cruz, D. Wishart and A. Scalbert, Phenol-Explorer: an online comprehensive database on polyphenol contents in foods, Database, 2010, 2010, bap024 CrossRef CAS PubMed.
- J. Pérez-Jiménez, J. V. Neveu, F. Vos and A. Scalbert, Identification of the 100 richest dietary sources of polyphenols: an application of the Phenol-Explorer database, Eur. J. Clin. Nutr., 2010, 64(3), S112–S120 CrossRef PubMed.
- A. Rodriguez-Mateos, D. Vauzour, C. G. Krueger, D. Shanmuganayagam, J. Reed, L. Calani, P. Mena, D. Del Rio and A. Crozier, Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: an update, Arch. Toxicol., 2014, 88(10), 1803–1853 CrossRef CAS PubMed.
- T. A. F. Corrêa, M. M. Rogero, N. M. A. Hassimotto and F. M. Lajolo, The two-way polyphenols-microbiota interactions and their effects on obesity and related metabolic eiseases, Front. Nutr., 2019, 6, 188 CrossRef PubMed.
- Y. F. Lin, Y. H. Lee, Y. H. Hsu, Y. J. Chen, Y. F. Lin, F. Y. Cheng and H. W. Chiu, Resveratrol-loaded nanoparticles conjugated with kidney injury molecule-1 as a drug delivery system for potential use in chronic kidney disease, Nanomedicine, 2017, 12(22), 2741–2756 CrossRef CAS PubMed.
- D. Beconcini, F. Felice, Y. Zambito, A. Fabiano, A. M. Piras, M. H. Macedo, B. Sarmentoq and R. Di Stefano, Anti-inflammatory effect of cherry extract loaded in polymeric nanoparticles: Relevance of particle internalization inv endothelial cells, Pharmaceutics, 2019, 11(10), 500 CrossRef CAS PubMed.
- S. Marinho, M. Illanes, J. Ávila-Román, V. Motilva and E. Talero, Antiinflammatory effects of rosmarinic acid-loaded nanovesicles in acute colitis through modulation of NLRP3 inflammasome, Biomolecules, 2021, 11(2), 162 CrossRef CAS PubMed.
- L. Sun, J. Ouyang, Z. Zeng, C. Zeng, Y. Ma, F. Zeng and S. Wu, Targeted and activatable nanosystem for fluorescent and optoacoustic imaging of immunemediated inflammatory diseases and therapy via inhibiting NF-κB/NLRP3 pathways, Bioact. Mater., 2021, 10, 79–92 CrossRef PubMed.
- G. Hu, J. Ding and D. H. Ryan, Trends in obesity prevalence and cardiometabolic risk factor control in US adults with diabetes, 1999–2020, Obesity, 2023, 31(3), 841–851 CrossRef PubMed.
- T. Ruskovska, V. Maksimova and D. Milenkovic, Polyphenols in human nutrition: from the in vitro antioxidant capacity to the beneficial effects on cardiometabolic health and related inter-individual variability - an overview and perspective, Br. J. Nutr., 2020, 123(3), 241–254 CrossRef CAS PubMed.
- T. Kiyimba, P. Yiga, M. Bamuwamye, P. Ogwok, B. Van der Schueren and C. Matthys, Efficacy of Dietary Polyphenols from Whole Foods and Purified Food Polyphenol Extracts in Optimizing Cardiometabolic Health: A Meta-Analysis of Randomized Controlled Trials, Adv. Nutr., 2023, 14(2), 270–282 CrossRef PubMed.
- H. A. Hausenblas, J. A. Schoulda and J. M. Smoliga, Resveratrol treatment as an adjunct to pharmacological management in type 2 diabetes mellitus-systematic review and meta-analysis, Mol. Nutr. Food Res., 2015, 59(1), 147–159 CrossRef CAS PubMed.
- G. Grosso, J. Godos, R. Lamuela-Raventos, S. Ray, A. Micek, A. Pajak, S. Sciacca, N. D'Orazio, D. Del Rio and F. Galvano, A comprehensive meta-analysis on dietary flavonoid and lignan intake and cancer risk: Level of evidence and limitations, Mol. Nutr. Food Res., 2017, 61, 1600930 CrossRef PubMed.
- M. J. Rodrigo-Gonzalo, S. González-Manzano, R. Mendez-Sánchez, C. Santos-Buelga and J. I. Recio-Rodríguez, Effect of polyphenolic complements on cognitive function in the elderly: a systematic review, Antioxidants, 2022, 11(8), 1549 CrossRef CAS PubMed.
- R. Giacco, G. Costabile, G. Fatati, L. Frittitta, M. I. Maiorino, G. Marelli, M. Parillo, D. Pistis, C. Tubili, C. Vetrani and M. Vitale, Effects of polyphenols on cardio-metabolic risk factors and risk of type 2 diabetes. A joint position statement of the Diabetes and Nutrition Study Group of the Italian Society of Diabetology (SID), the Italian Association of Dietetics and Clinical Nutrition (ADI) and the Italian Association of Medical Diabetologists (AMD), Nutr., Metab. Cardiovasc. Dis., 2020, 30(3), 355–367 CrossRef CAS PubMed.
- S. Qin and D. X. Hou, Multiple regulations of Keap1/Nrf2 system by dietary phytochemicals, Mol. Nutr. Food Res., 2016, 60(8), 1731–1755 CrossRef CAS PubMed.
- J. Das, R. Ramani and M. O. Suraju, Polyphenol compounds and PKC signaling, Biochim. Biophys. Acta, 2016, 1860(10), 2107–2121 CrossRef CAS PubMed.
- T. Farkhondeh, S. L. Folgado, A. M. Pourbagher-Shahri, M. Ashrafizadeh and S. Samarghandian, The therapeutic effect of resveratrol: Focusing on the Nrf2 signaling pathway, Biomed. Pharmacother., 2020, 127, 110234 CrossRef CAS PubMed.
- M. Ashrafizadeh, Z. Ahmadi, R. Mohammadinejad, T. Farkhondeh and S. Samarghandian, Curcumin Activates the Nrf2 Pathway and Induces Cellular Protection Against Oxidative Injury, Curr. Mol. Med., 2020, 20(2), 116–133 CAS.
- I. Krga, D. Milenkovic, C. Morand and L. E. Monfoulet, An update on the role of nutrigenomic modulations in mediating the cardiovascular protective effect of fruit polyphenols, Food Funct., 2016, 7, 3656–3676 RSC.
- H. S. Kim, M. J. Quon and J. A. Kim, New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate, Redox Biol., 2014, 2, 187–195 CrossRef CAS PubMed.
- K. Goszcz, G. G. Duthie, D. Stewart, S. J. Leslie and I. L. Megson, Bioactive polyphenols and cardiovascular disease: chemical antagonists, pharmacological agents or xenobiotics that drive an adaptive response?, Br. J. Pharmacol., 2017, 174, 1209–1225 CrossRef CAS PubMed.
- A. Kerimi and G. Williamson, At the interface of antioxidant signalling and cellular function: key polyphenol effects, Mol. Nutr. Food Res., 2016, 60, 1770–1788 CrossRef CAS PubMed.
- L. E. Monfoulet, S. Mercier, D. Bayle, R. Tamaian, N. Barber-Chamoux, C. Morand and D. Milenkovic, Curcumin modulates endothelial permeability and monocyte transendothelial migration by affecting endothelial cell dynamics, Free Radical Biol. Med., 2017, 112, 109–120 CrossRef CAS PubMed.
- I. Krga, R. Tamaian, S. Mercier, C. Boby, L. E. Monfoulet, M. Glibetic, C. Morand and D. Milenkovic, Anthocyanins and their gut metabolites attenuate monocyte adhesion and transendothelial migration through nutrigenomic mechanisms regulating endothelial cell permeability, Free Radical Biol. Med., 2018, 124, 364–379 CrossRef CAS PubMed.
- D. Alvarez-Cilleros, S. Ramos, L. Goya and M. A. Martín, Colonic metabolites from flavanols stimulate nitric oxide production in human endothelial cells and protect against oxidative stress-induced toxicity and endothelial dysfunction, Food Chem. Toxicol., 2018, 115, 88–97 CrossRef CAS PubMed.
- D. Milenkovic, W. V. Berghe, C. Morand, S. Claude, A. van de Sandt, S. Gorressen, L. E. Monfoulet, C. S. Chirumamilla, K. Declerck, K. S. V. Szic, M. Lahtela-Kakkonen, C. Gerhauser, M. W. Merx and M. Kelm, A systems biology network analysis of nutri(epi)genomic changes in endothelial cells exposed to epicatechin metabolites, Sci. Rep., 2018, 8, 15487 CrossRef PubMed.
- A. Engin, The pathogenesis of obesity-associated adipose tissue inflammation, Adv. Exp. Med. Biol., 2017, 960, 221–245 CrossRef CAS PubMed.
- H. Eo, J. E. Park, Y. J. Jeon and Y. Lim, Ameliorative effect of Ecklonia cava polyphenol extract on renal inflammation associated with aberrant energy metabolism and oxidative stress in high fat diet-induced obese Mice, J. Agric. Food Chem., 2017, 65(19), 3811–3818 CrossRef CAS PubMed.
- Y. J. Cho, H. G. Lee, K. H. Seo, W. Yokoyama and H. Kim, Antiobesity effect of prebiotic polyphenol-rich grape seed flour supplemented with probiotic kefir-eerived lactic acid bacteria, J. Agric. Food Chem., 2018, 66(47), 12498–12511 CrossRef CAS PubMed.
- M. J. Zhu, Y. Kang, Y. Xue, X. Liang, M. P. G. García, D. Rodgers, D. R. Kagel and M. Du, Red raspberries suppress NLRP3 inflammasome and attenuate metabolic abnormalities in diet-induced obese mice, J. Nutr. Biochem., 2018, 53, 96–103 CrossRef CAS PubMed.
- R. Fan, M. You, A. M. Toney, J. Kim, D. Giraud, Y. Xian, F. Ye, L. Gu, A. E. Ramer-Tait and S. Chung, Red raspberry polyphenols attenuate high-fat diet-driven activation of NLRP3 inflammasome and its paracrine suppression of adipogenesis via histone modifications, Mol. Nutr. Food Res., 2020, 64(15), e1900995 CrossRef PubMed.
- I. I. López-Tenorio, A. Domínguez-López, Á. Miliar-García, G. N. Escalona-Cardoso, S. A. Real-Sandoval, A. Gómez-Alcalá and M. E. Jaramillo-Flores, Modulation of the mRNA of the Nlrp3 inflammasome by morin and PUFAs in an obesity model induced by a high-fat diet, Food Res. Int., 2020, 137, 109706 CrossRef PubMed.
- L. Rodrigues, C. M. Donado-Pestana, M. H. C. Moura, R. Rossi, E. Silva, É. V. M. Pessoa and M. I. Genovese, Phenolic compounds from jaboticaba (Plinia jaboticaba (Vell.) Berg) ameliorate intestinal inflammation and associated endotoxemia in obesity, Food Res. Int., 2021, 141, 110139 CrossRef CAS PubMed.
- D. Liu, X. Zeng, X. Li, J. L. MehtaL and X. Wang, Role of NLRP3 inflammasome in the pathogenesis of cardiovascular diseases, Basic Res. Cardiol., 2018, 113, 5 CrossRef PubMed.
- D. Lv, X. Cheng, L. Tang and M. Jiang, The cardioprotective effect of total flavonoids on myocardial ischemia/reperfusion in rats, Biomed. Pharmacother., 2017, 88, 277–284 CrossRef CAS PubMed.
- I. Roth, R. Casas, M. Ribó-Coll, M. Doménech, R. M. Lamuela-Raventós and R. Estruch, Acute consumption of Andalusian aged wine and gin decreases the expression of genes related to atherosclerosis in men with high cardiovascular risk: Randomized intervention trial, Clin. Nutr., 2019, 38(4), 1599–1606 CrossRef CAS PubMed.
- H. Feng, S. Q. Mou, W. J. Li, N. Zhang, Z. Y. Zhou, W. Ding, Z. Y. Bian and H. H. Lia, Resveratrol Inhibits Ischemia-Induced Myocardial Senescence Signals and NLRP3 Inflammasome Activation, Oxid. Med. Cell. Longevity, 2020, 2020, 2647807 Search PubMed.
- Y. Li, R. Wang, L. Xue, Y. Yang and F. Zhi, Astilbin protects against cerebral ischaemia/reperfusion injury by inhibiting cellular apoptosis and ROS-NLRP3 inflammasome axis activation, Int. Immunopharmacol., 2020, 84, 106571 CrossRef CAS PubMed.
- Z. Sun, W. Lu, N. Lin, H. Lin, J. Zhang, T. Ni, L. Meng, C. Zhang and H. Guo, Dihydromyricetin alleviates doxorubicin-induced cardiotoxicity by inhibiting NLRP3 inflammasome through activation of SIRT1, Biochem. Pharmacol., 2020, 175, 113888 CrossRef CAS PubMed.
- L. J. Xu, R. C. Chen, X. Y. Ma, Y. Zhu, G. B. Sun and X. B. Sun, Scutellarin protects against myocardial ischemia-reperfusion injury by suppressing NLRP3 inflammasome activation, Phytomedicine, 2020, 68, 153169 CrossRef CAS PubMed.
- B. Yang, Y. Sun, C. Lv, W. Zhang and Y. Chen, Procyanidins exhibits neuroprotective activities against cerebral ischemia reperfusion injury by inhibiting TLR4-NLRP3 inflammasome signal pathway, Psychopharmacology, 2020, 237(11), 3283–3293 CrossRef CAS PubMed.
- J. Zhao, Z. Wang, Z. Yuan, S. Lv and Q. Su, Baicalin ameliorates atherosclerosis by inhibiting NLRP3 inflammasome in apolipoprotein E-deficient mice, Diabetes Vasc. Dis. Res., 2020, 17(6), 1479164120977441 CAS.
- H. Guo, L. Zhu, P. Tang, D. Chen, Y. Li, J. Li and C. Bao, Carthamin yellow improves cerebral ischemia-reperfusion injury by attenuating inflammation and ferroptosis in rats, Int. J. Mol. Med., 2021, 47(4), 52 CrossRef CAS PubMed.
- Y. W. Hsiao, Y. N. Tsai, Y. T. Huang, S. H. Liu, Y. J. Lin, L. W. Lo, Y. F. Hu, F. P. Chung, S. F. Lin, S. L. Chang, S. Higa and S. A. Chen, Rhodiola crenulata reduces ventricular arrhythmia through mitigating the activation of IL-17 and inhibiting the MAPK signaling pathway, Cardiovasc. Drugs Ther., 2021, 35(5), 889–900 CrossRef CAS PubMed.
- J. Jiang, X. Gu, H. Wang and S. Ding, Resveratrol improves cardiac function and left ventricular fibrosis after myocardial infarction in rats by inhibiting NLRP3 inflammasome activity and the TGF-β1/SMAD2 signaling pathway, PeerJ, 2021, 9, e11501 CrossRef PubMed.
- J. Luo, J. Chen, C. Yang, J. Tan, J. Zhao, N. Jiang and Y. Zhao, 6-Gingerol protects against cerebral ischemia/reperfusion injury by inhibiting NLRP3 inflammasome and apoptosis via TRPV1/FAF1 complex dissociation-mediated autophagy, Int. Immunopharmacol., 2021, 100, 108146 CrossRef CAS PubMed.
- D. C. Ma, N. N. Zhang, Y. N. Zhang and H. S. Chen, Salvianolic Acids for Injection alleviates cerebral ischemia/reperfusion injury by switching M1/M2 phenotypes and inhibiting NLRP3 inflammasome/pyroptosis axis in microglia in vivo and in vitro, J. Ethnopharmacol., 2021, 270, 113776 CrossRef CAS PubMed.
- Y. Xu, X. Gao, L. Wang, M. Yang and R. Xie, Bakuchiol ameliorates cerebral ischemia-reperfusion injury by modulating NLRP3 inflammasome and Nrf2 signaling, Respir. Physiol. Neurobiol., 2021, 292, 103707 CrossRef CAS PubMed.
- W. X. Zheng, W. Q. He, Q. R. Zhang, J. X. Jia, S. Zhao, F. J. Wu and X. L. Cao, Baicalin Inhibits NLRP3 inflammasome activity via the AMPK signaling pathway to alleviate cerebral ischemia-reperfusion injury, Inflammation, 2021, 44(5), 2091–2105 CrossRef CAS PubMed.
- R. Huang, Z. Zhao, X. Jiang, W. Li, L. Zhang, B. Wang and H. Tie, Liposomal chrysin attenuates hepatic ischaemia-reperfusion injury: possible mechanism via inhibiting NLRP3 inflammasome, J. Pharm. Pharmacol., 2022, 74(2), 216–226 CrossRef PubMed.
- X. Wei, Y. Wu, H. Pan, Q. Zhang, K. He, G. Xia, H. Xia, S. Lin and H. C. Shang, Proteomics revealed that mitochondrial function contributed to the protective effect of Herba Siegesbeckiae against cardiac ischemia/reperfusion injury, Front. Cardiovasc. Med., 2022, 9, 895797 CrossRef CAS PubMed.
- Y. Zhang and G. RuXian, Didymin, a natural flavonoid, relieves the progression of myocardial infarction via inhibiting the NLR family pyrin domain containing 3 inflammasome, Pharm. Biol., 2022, 60(1), 2319–2327 CrossRef CAS PubMed.
- H. M. Lee, J. J. Kim, H. J. Kim, M. Shong, B. J. Ku and E. K. Jo, Upregulated NLRP3 inflammasome activation in patients with type 2 diabetes, Diabetes, 2013, 62(1), 194–204 CrossRef CAS PubMed.
- C. Wang, Y. Pan, Q. Y. Zhang, F. M. Wang and L. D. Kong, Quercetin and allopurinol ameliorate kidney injury in STZ-treated rats with regulation of renal NLRP3 inflammasome activation and lipid accumulation, PLoS One, 2012, 7(6), e38285 CrossRef CAS PubMed.
- Q. Y. Zhang, Y. Pan, R. Wang, L. L. Kang, Q. C. Xue, X. N. Wang and L. D. Kong, Quercetin inhibits AMPK/TXNIP activation and reduces inflammatory lesions to improve insulin signaling defect in the hypothalamus of high fructose-fed rats, J. Nutr. Biochem., 2014, 25(4), 420–428 CrossRef CAS PubMed.
- X. An, Y. Zhang, Y. Cao, J. Chen, H. Qin and L. Yang, Punicalagin Protects Diabetic Nephropathy by Inhibiting Pyroptosis Based on TXNIP/NLRP3 Pathway, Nutrients, 2020, 12(5), 1516 CrossRef CAS PubMed.
- N. Lalitha, B. Sadashivaiah, T. R. Ramaprasad and S. A. Sing, Anti-hyperglycemic activity of myricetin, through inhibition of DPP-4 and enhanced GLP-1 levels, is attenuated by co-ingestion with lectin-rich protei, PLoS One, 2020, 15(4), e0231543 CrossRef CAS PubMed.
- Q. Ma, Q. Yang, J. Chen, C. Yu, L. Zhang, W. Zhou and M. Chen, Salvianolic Acid A Ameliorates Early-Stage Atherosclerosis Development by Inhibiting NLRP3 Inflammasome Activation in Zucker Diabetic Fatty Rats, Molecules, 2020, 25(5), 1089 CrossRef CAS PubMed.
- G. R. Chai, S. Liu, H. W. Yang and X. L. Chen, Quercetin protects against diabetic retinopathy in rats by inducing heme oxygenase-1 expression, Neural Regener. Res., 2021, 16(7), 1344–1350 CrossRef CAS PubMed.
- J. Yao, Y. Li, Y. Jin, Y. Chen, L. Tian and W. He, Synergistic cardioptotection by tilianin and syringin in diabetic cardiomyopathy involves interaction of TLR4/NF-κB/NLRP3 and PGC1a/SIRT3 pathways, Int. Immunopharmacol., 2021, 96, 107728 CrossRef CAS PubMed.
- C. Zhang, X. Li, X. Hu, Q. Xu, Y. Zhang, H. Liu, Y. Diao, X. Zhang, L. Li, J. Yu, H. Yin and J. Peng, Epigallocatechin-3-gallate prevents inflammation and diabetes -Induced glucose tolerance through inhibition of NLRP3 inflammasome activation, Int. Immunopharmacol., 2021, 93, 107412 CrossRef CAS PubMed.
- T. Zheng, Q. Wang, F. Bian, Y. Zhao, W. Ma, Y. Zhang, W. Lu, P. Lei, L. Zhang, X. Hao and L. Chen, Salidroside alleviates diabetic neuropathic pain through regulation of the AMPK-NLRP3 inflammasome axis, Toxicol. Appl. Pharmacol., 2021, 416, 115468 CrossRef CAS PubMed.
- Y. Liu, R. Sun, X. Lin, L. Wu, H. Chen, S. Shen, Y. Li, Y. Wei and G. Deng, Procyanidins and its metabolites by gut microbiome improves insulin resistance in gestational diabetes mellitus mice model via regulating NF-κB and NLRP3 inflammasome pathway, Biomed. Pharmacother., 2022, 151, 113078 CrossRef CAS PubMed.
- R. Yang, J. Chen, Q. Jia, X. Yang and S. Mehmood, Epigallocatechin-3-gallate ameliorates renal endoplasmic reticulum stress-mediated inflammation in type 2 diabetic rats, Exp. Biol. Med., 2022, 247(16), 1410–1419 CrossRef CAS PubMed.
- Z. T. Zhang, W. J. He, S. M. Deng, S. H. Xu, X. Zeng, Z. M. Qian, Z. Q. Chen, S. M. Wang and D. Tang, Trilobatin alleviates non-alcoholic fatty liver disease in high-fat diet plus streptozotocin-induced diabetic mice by suppressing NLRP3 inflammasome activation, Eur. J. Pharmacol., 2022, 933, 175291 CrossRef CAS PubMed.
- Y. Chen, Y. Zheng, R. Chen, J. Shen, S. Zhang, Y. Gu, J. Shi and G. Meng, Dihydromyricetin Attenuates Diabetic Cardiomyopathy by Inhibiting Oxidative Stress, Inflammation and Necroptosis via Sirtuin 3 Activation, Antioxidants, 2023, 12(1), 200 CrossRef CAS PubMed.
- Y. Chen, Y. Zheng, R. Chen, J. Shen, S. Zhang, Y. Gu, J. Shi and G. Meng, Dihydromyricetin Attenuates Diabetic Cardiomyopathy by Inhibiting Oxidative Stress, Inflammation and Necroptosis via Sirtuin 3 Activation, Antioxidants, 2023, 12(1), 200 CrossRef CAS PubMed.
- B. Das, C. Sarkar, V. S. Rawat, D. Kalita, S. Deka and A. Agnihotri, Promise of the NLRP3 inflammasome inhibitors in in vivo disease models, Molecules, 2021, 26(16), 4996 CrossRef CAS PubMed.
- A. Wree, M. D. McGeough, C. A. Peña, M. Schlattjan, H. Li, M. E. Inzaugarat, K. Messer, A. Canbay, H. M. Hoffman and A. E. Feldstein, NLRP3 inflammasome activation is required for fibrosis development in NAFLD, J. Mol. Med., 2014, 92, 1069–1082 CrossRef CAS PubMed.
- S. Gaul, A. Leszczynska, F. Alegre, B. Kaufmann, C. D. Johnson, L. A. Adams, A. Wree, G. Damm, D. Seehofer, C. J. Calvente, D. Povero, T. Kisseleva, A. Eguchi, M. D. McGeough, H. M. Hoffman, P. Pelegrin, U. Laufs and A. E. Feldstein, Hepatocyte pyroptosis and release of inflammasome particles induce stellate cell activation and liver fibrosis, J. Hepatol., 2021, 74(1), 156–167 CrossRef CAS PubMed.
- C. Ding, Y. Zhao, X. Shi, N. Zhang, G. Zu, Z. Li, J. Zhou, D. Gao, L. Lv, X. Tian and J. Yao, New insights into salvianolic acid A action: Regulation of the TXNIP/NLRP3 and TXNIP/ChREBP pathways ameliorates HFD-induced NAFLD in rats, Sci. Rep., 2016, 6, 28734 CrossRef CAS PubMed.
- S. L. Glisan, C. Ryan, A. P. Neilson and J. D. Lambert, Cranberry extract attenuates hepatic inflammation in high-fat-fed obese mice, J. Nutr. Biochem., 2016, 37, 60–66 CrossRef CAS PubMed.
- D. Porras, E. Nistal, S. Martínez-Flórez, S. Pisonero-Vaquero, J. L. Olcoz, R. Jover, J. González-Gallego, M. V. García-Mediavilla and S. Sánchez-Campos, Protective effect of quercetin on high-fat diet-induced non-alcoholic fatty liver disease in mice is mediated by modulating intestinal microbiota imbalance and related gut-liver axis activation, Free Radical Biol. Med., 2017, 102, 188–202 CrossRef CAS PubMed.
- H. M. Song, X. Li, Y. Y. Liu, W. P. Lu, Z. H. Cui, L. Zhou, D. Yao and H. M. Zhang, Carnosic acid protects mice from high-fat diet-induced NAFLD by regulating MARCKS, Int. J. Mol. Med., 2018, 42(1), 193–207 CAS.
- T. Zheng, X. Yang, W. Li, Q. Wang, L. Chen, D. Wu, F. Bian, S. Xing and S. Jin, Salidroside attenuates high-fat diet-induced nonalcoholic fatty liver disease via AMPK-dependent TXNIP/NLRP3 Pathway, Oxid. Med. Cell. Longevity, 2018, 2018, 8597897 Search PubMed.
- X. Yang, Y. Deng, Y. Tu, D. Feng and W. Liao, Nobiletin mitigates NAFLD via lipophagy and inflammation, Food Funct., 2022, 13(19), 10186–10199 RSC.
- X. Zhu, X. Lin, P. Zhang, Y. Liu, W. Ling and H. Guo, Upregulated NLRP3 inflammasome activation is attenuated by anthocyanins in patients with nonalcoholic fatty liver disease: A case-control and an intervention study, Clin. Res. Hepatol. Gastroenterol., 2022, 46(4), 101843 CrossRef CAS PubMed.
- Y. Zhou, S. Wang, T. Wan, Y. Huang, N. Pang, X. Jiang, Y. Gu, Z. Zhang, J. Luo and L. Yang, Cyanidin-3-O-β-glucoside inactivates NLRP3 inflammasome and alleviates alcoholic steatohepatitis via SirT1/NF-κB signaling pathway, Free Radical Biol. Med., 2020, 160, 334–414 CrossRef CAS PubMed.
- B.-C. Zhang, W.-M. Li, R. Guo and Y. W. Xu, Salidroside decreases atherosclerotic plaque formation in low-density lipoprotein receptor-deficient mice, J. Evidence-Based Complementary Altern. Med., 2012, 2012, 907508 Search PubMed.
- T. Wang, H. Xu, R. Dong, S. Wu, Y. Guo and D. Wang, Effectiveness of targeting the NLRP3 inflammasome by using natural polyphenols: A systematic review of implications on health effects, Food Res. Int., 2023, 112567 CrossRef CAS PubMed.
- S. Reagan-Shaw, M. Nihal and N. Ahmad, Dose translation from animal to human studies revisited, FASEB J., 2008, 22(3), 659–661 CrossRef CAS PubMed.
- J. Pérez-Jiménez, L. Fezeu, M. Touvier, N. Arnault, C. Manach, S. Hercberg, P. Galan and A. Scalbert, Dietary intake of 337 polyphenols in French adults, Am. J. Clin. Nutr., 2010, 93(6), 1220–1228 CrossRef PubMed.
- N. P. Bondonno, F. Dalgaard, C. Kyro, K. Murray, C. P. Bondonno, J. R. Lewis, K. D. Croft, G. Gislason, A. Scalbert, A. Cassidy, A. Tjonneland, K. Overvad and J. M. Hodgson, Flavonoid intake is associated with lower mortality in the Danish Diet Cancer and Health Cohort, Nat. Commun., 2019, 10, 3651 CrossRef PubMed.
- V. Neveu, J. Perez-Jiménez, F. Vos, V. Crespy, L. du Chaffaut, L. Mennen, C. Knox, R. Eisner, J. Cruz, D. Wishart and A. Scalbert, Phenol-Explorer: an online comprehensive database on polyphenol contents in foods, Database, 2010, 2010, bap024 CrossRef CAS PubMed.
- F. Martín-Sánchez, C. Diamond, M. Zeitler, A. I. Gómez, A. Baroja-Mazo, J. Bagnall, D. Spiller, M. White, M. J. D. Daniels, A. Mortellaro, M. Peñalver, P. Paszek, J. P. Steringer, W. Nickel, D. Brough and P. Pelegrín, Inflammasome-dependent IL-1β release depends upon membrane permeabilization, Cell Death Differ., 2016, 23(7), 1219–1231 CrossRef PubMed.
- A. Cortés-Martín, M. V. Selma, F. A. Tomás-Barberán, A. González-Sarrías and J. C. Espín, Where to look into the puzzle of polyphenols and health? The postbiotics and gut microbiota associated with human metabotypes, Mol. Nutr. Food Res., 2020, 64(9), e1900952 CrossRef PubMed.
- R. Giacco, G. Costabile, G. Fatati, L. Frittitta, M. Maiorino, G. Marelli, M. Parillo, D. Pistis, C. Tubili, C. Vetrani and M. Vitale, Effects of polyphenols on cardio-metabolic risk factors and risk of type 2 diabetes. A joint position statement of the Diabetes and Nutrition Study Group of the Italian Society of Diabetology (SID), the Italian Association of Dietetics and Clinical Nutrition (ADI) and the Italian Association of Medical Diabetologists (AMD), Nutr., Metab. Cardiovasc. Dis., 2020, 30(3), 355–367 CrossRef CAS PubMed.
- L. Condezo-Hoyos, C. Gazi and J. Pérez-Jiménez, Design of polyphenol-rich diets in clinical trials: a systematic review, Food Res Int., 2021, 149, 110655 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2023 |