A syndromic diagnostic assay on a macrochannel-to-digital microfluidic platform for automatic identification of multiple respiratory pathogens†
Cheng
Dong‡
a,
Fei
Li‡
 bc,
Yun
Sun
c,
Dongling
Long
d,
Chunzhao
Chen
e,
Mengyan
Li
bc,
Yun
Sun
c,
Dongling
Long
d,
Chunzhao
Chen
e,
Mengyan
Li
 f,
Tao
Wei
gh,
Rui P.
Martins
i,
Tianlan
Chen
*c and
Pui-In
Mak
*ij
f,
Tao
Wei
gh,
Rui P.
Martins
i,
Tianlan
Chen
*c and
Pui-In
Mak
*ij
aSchool of Intelligent Systems Science and Engineering/JNU-Industry School of Artificial Intelligence, Jinan University, Zhuhai 519000, China
bDepartment of Biomedical Engineering, Jinan University, Guangzhou, 510632, China
cDigifluidic Biotech Ltd., Zhuhai 519000, China. E-mail: oscar.chen@digifluidic.com
dZhuhai Center for Disease Control and Prevention, Zhuhai 519087, China
eAdvanced Interdisciplinary Institute of Environment and Ecology, Beijing Normal University, Zhu Hai 519087, China
fDepartment of Chemistry and Environmental Science, New Jersey Institute of Technology, Newark, 07102, USA
gDepartment of Bioengineering, College of Food Science, South China Agricultural University, Guangzhou, 510640, China
hPan Asia (Jiangmen) Institute of Biological Engineering and Health, Jiangmen, 529080, China
iState-Key Laboratory of Analog and Mixed-Signal VLSI, Institute of Microelectronics, University of Macau, Taipa, Macau SAR 999078, China. E-mail: pimak@um.edu.mo
jFaculty of Science and Technology, University of Macau, Taipa, Macau SAR 999078, China
First published on 9th November 2023
Abstract
The worldwide COVID-19 pandemic has changed people's lives and the diagnostic landscape. The nucleic acid amplification test (NAT) as the gold standard for SARS-CoV-2 detection has been applied in containing its transmission. However, there remains a lack of an affordable on-site detection system at resource-limited areas. In this study, a low cost “sample-in-answer-out” system incorporating nucleic acid extraction, purification, and amplification was developed on a single macrochannel-to-digital microfluidic chip. The macrochannel fluidic subsystem worked as a world-to-chip interface receiving 500–1000 μL raw samples, which then underwent bead-based extraction and purification processes before being delivered to DMF. Electrodes actuate an eluent dispensed to eight independent droplets for reverse transcription quantitative polymerase chain reaction (RT-qPCR). By reading with 4 florescence channels, the system can accommodate a maximum of 32 detection targets. To evaluate the proposed platform, a comprehensive assessment was conducted on the microfluidic chip as well as its functional components (i.e., extraction and amplification). The platform demonstrated a superior performance. In particular, using clinical specimens, the chip targeting SARS-CoV-2 and Flu A/B exhibited 100% agreement with off-chip diagnoses. Furthermore, the fabrication of chips is ready for scaled-up manufacturing and they are cost-effective for disposable use since they are assembled using a printed circuit board (PCB) and prefabricated blocks. Overall, the macrochannel-to-digital microfluidic platform coincides with the requirements of point-of-care testing (POCT) because of its advantages: low-cost, ease of use, comparable sensitivity and specificity, and availability for mass production.
Introduction
The nucleic acid amplification test (NAT) is a technology that detects a particular deoxyribonucleic acid (DNA) or ribonucleic acid (RNA) sequence. It thus could sensitively and specifically1 detect a few copies of a given nuclei acid with a small sample size. Various NAT technologies have long been used in many fields such as molecular research, genetic testing, forensics, agriculture and clinical medicine.2 Among them, reverse transcription-quantitative PCR (RT-qPCR) has been widely accepted as the gold standard in many clinical diagnostics, such as detecting various pathogens in the respiratory tract, intestinal tract, and reproductive systems.3,4 This is attributed to its well-recognized properties of quantification, sensitivity, and specificity.The worldwide pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has significantly changed the daily lives of billions and deeply impacted global development in economic, political, and social domains. As an infectious respiratory virus, its main clinical manifestations were reported similar to influenza A/B viruses and other common cold-induced bacteria/viruses, resulting in complicating the diagnosis of COVID-19 from symptoms.5 Unfortunately, SARS-CoV-2 is potentially pre- or oligosymptomatically transmitted6 and elicits relatively high community transmission (674 million cases) and mortality (6.8 million deaths).7 Rapid diagnostic assays, therefore, are of urgent need for containing the spread of COVID-19 and/or other infectious diseases. Previous research reported that NAT could timely and easily detect pathogens from specimens such as nasopharyngeal and oropharyngeal swabs upon symptom onset.8 However, several drawbacks still exist. It requires complex equipment in a centralized laboratory and highly skilled technicians to operate it. Meanwhile, this kind of offsite detection introduces many uncertainties regarding timeliness and safety. Thus, fully automatic equipment is imperative for decentralized use in source-limited regions.
Microfluidics is an eloquent technology for picoliter- to microliter-sized droplet manipulation. It has been applied in diverse decentralized NAT diagnoses which have been partially commercialized such as GeneXpert® from Cepheid, FilmArray® from BioFire Diagnosis, Cobas Liat® from Roche, etc. They are all fully automatic and portable point-of-care test (POCT) devices with self-contained, single use, biosafe, microfluidic cartridges for NATs. Digital microfluidics (DMF) based on electrowetting-on-dielectric (EWOD) provides an alternative for POCT devices and cartridges. This technology has been developed for over 40 years and is widely employed for multiple purposes.9,10 Many researchers have developed their unique DMF devices for specific applications or cutting edge research goals, such as various omics sample preparation,11–13 online monitoring of chemicals,14 antimicrobial susceptibility test,15 and even pollutant monitoring.16 The DMF chip discretely actuates droplets on an array of electrodes without external pumps or valves or complicated geometries, and it can be simply fabricated by photolithography.17 DMF, thus, is readily available for commercial use such as NAT, liberating tedious operations in the laboratory.18 However, indeed, the development and commercialization of DMF are sluggish, which have been hindered by the gap between research and market.18 Several companies (e.g., Nanolytics, NuGEN, Advanced Liquid Logic) are dedicated to DMF commercialization mostly in the area of biomedical utilization. Successful products proved that DMF is a feasible tool for various biological applications; nevertheless, the failures of pioneering products such as NeoPrep Library Prep System by Illumina (famous for its sequencing equipment) remind us of the challenges of engineering and manufacturing.19 Thus, many efforts were made to increase the reliability through the design of the circuitry,20 optimization of reagents,21,22 and other approaches.23–25
However, the feasibility of DMF technology in the field of qPCR nucleic acid detection remains to be verified due to challenges such as sample acceptance, seamless connection between extraction and amplification, and fluorescence detection. In the meantime, the scarcity of research into DMF commercialization also provided an impetus to propose this study, particularly with regard to the design of DMF chips. Herein, an all-in-one NAT system was presented, named Virus Hunter 2.0 (VH 2.0). This study aimed to provide insights into the diagnostic applications of DMF from its design and commercialization. Compared with the previous version VH 1.0, the following improvements were made to realize a “sample-in-to-result-out” fully automatic process: (1) VH 2.0 integrated automatic nucleic acid extraction process; (2) all reagents are stored in the chip through diverse approaches depending on their properties and forms; (3) the macrochannel-to-digital microfluidics is adopted. It served as a world-to-chip interface which receives hundreds of microliters of samples and exports a limited volume of liquid for DMF manipulation; specially designed electrodes for droplets overcome the height difference from macrochannel to digital microfluidics; (4) the thin-film heater on the chip is similar to a previous version because it provides an accurate temperature control and miniaturized device. In this version we improve the performance of the temperature control by changing the resistor's location and the geometry of the copper foil. Moreover, as a commercial product, the performance and reliability of the system have been strictly evaluated before mass production, including the performances of discrete modules, integrated chips, and clinical specimens and a wide variety of aspects. We also compared VH 2.0 with other POCT platforms targeting respiratory pathogens based on microfluidics. In summary, this study improved the automation and integration levels of a DMF-based nucleic acid detection platform, which brings the DMF technology significantly closer to commercialization.
Experimental
Reagents and materials
Pseudo-viruses of SARS-CoV-2 and influenza A/B with a concentration of 108 copies per mL were purchased from Sangon Biotech (Shanghai, China). Upon receipt, they were immediately aliquoted into 1.5 mL micro-centrifuge tubes and stored at −80 °C for future use. Primers and probes were designed to target the M1 gene from influenza A, the hemagglutinin gene from influenza B, and nucleocapsid protein and ORF1 genes from SARS-CoV-2 according to previous research26 and synthesized by Sangon Biotech (Shanghai, China). Sequences are indicated in Table S1 in the ESI.† Magnetic beads and commercial pathogen nucleic acid extraction kits were purchased from EasyDiagnosis Co., Ltd. (Wuhan, China). All the other materials were purchased from Sangon Biotech (Shanghai, China) if not specifically indicated.VH 2.0 system
Validation of the VH 2.0 biochip performance
| Efficiency = 10−1/slope − 1. |
Results and discussion
Temperature control
A uniplanar heater together with an NTC thermistor achieved rapid and accurate on-chip heating and sensing. By optimizing the resistor's pattern on the PCB and implementing PID control (Fig. 2c), it is possible to generate a homogenous temperature within the paralleled qPCR reaction spots at a relatively short response reaching an asymptotic state (90% of the set temperature). That was about 13 s drop from 95 °C to 60 °C and took about 4 s raising up to 95 °C (Fig. 2a). At the asymptotic state, fine adjustments were made to approach the set temperature and then retained at the setpoint. Temperatures fluctuated with <±1.27 °C (2.11% of the set temperature) at the conditioning period (the first few seconds upon set temperature) and <±0.5 °C (0.49% of the set temperature) at the stable period (Fig. 2a and b). As heat was lost at the edge of the heating region, some self-compensations were needed to counterbalance the side effects.30 A serpentine resistor was patterned in an hourglass-shaped geometry (Fig. 2d), while copper foil was configured for a uniform thermal distribution. Traditional thermal loading units were usually built in the device with metal blocks. However, the performance was associated with thermal conductivity, heat capacity, and thermal coupling to the environment of the substrate. The on-chip thin-film heater (heating) and NTC thermistor (sensing) embedded in the PCB were closer to the heating area (i.e., qPCR chamber), making it a short transient regime and quick response by closed-loop control. Therefore, the thermal program was strictly followed for the set temperature with minor discrepancies (Fig. 2a). Despite the heating module being moved onto the chip, the production cost (<2 USD per PCB substrate) is acceptable for disposable use due to the highly developed PCB manufacture. On the other hand, the utilization of on-chip thermal unit miniaturized outline dimensions of the devices, which is beneficial for POCT. Frankly, apart from the contact-based Joule heating, some oncoming technologies are promising and attract interest from many communities, such as surface acoustic waves,31 microwave32 and lasers.33 However, their drawbacks in terms of fabrication, integration, cost, and accuracy of the position and temperature control, etc. are evident34 so far.Droplet manipulation on macrochannel-to-digital microfluidic platform
In this macrochannel-to-digital platform, 500 μL raw sample underwent nucleic acid extraction through an RNA extraction module; pure RNA was transferred into 60 μL elution buffer at the elution room where the interface of the macrochannel and digital microfluidics is. At the end of the RNA extraction process and the start of the dispensing process, there is a critical interface of the macro-dielectric layer and channel structure, enabling an effective bonding of two patterns of microfluidics. The elution room was entirely situated on the dielectric layer at a height of 1.2 mm, while the height of the paralleled PCB and ITO glass was 0.6 mm. Hence the height difference trapped the elution buffer in this well by surface tension, as shown in Fig. 3a. To actuate the large droplet escaping from the interface, an electrode spanned across the elution room and oil-filled chamber was patterned. Plugging high voltage to adjacent electrodes stepwise could force the eluent to enter into the digital microfluidic controlled region, as shown in Fig. 3b and ESI† video. After the extraction process, the eluent was then actuated to an array of L-shaped electrodes for reaction droplet generation, which is approximately 5.55 μL of each droplet. Their volumes have been determined and discussed in the ESI† (Methods, Results and discussion, and Fig. S3).The system exhibited eminent stability over a hundred tests of the on-chip “sample-to-answer” workflow. Differing from previous digital-to-microchannel infrastructures, the macrochannel was exploited because it is well-suited for bead-based RNA extraction at a volume of hundreds of microliters. Previous studies combined DMF with microchannel networks by multi-layer design35 or side-on pattern36 for handling droplets. However, microchannels could not accommodate large volumes of samples. Meanwhile, the sophisticated structure may introduce more uncertainties which impede their commercialization. In contrast, magnetic beads were reported to be compatible with DMF by (1) immobilizing beads and refreshing droplets37 or (2) extracting and fusing beads with sequential droplets.38 Herein, a device was developed with the latter mechanism. The released RNA from viruses was adhered to magnetic beads and dragged to sequential cavities for different purposes of treatments, eventually accessing the elution room.
On-chip RNA extraction
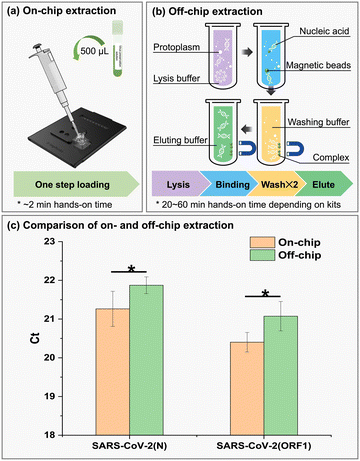
The extracted RNA from 107 SARS-CoV-2 pseudoviruses through on- and off-chip methods was simultaneously loaded into the qPCR instrument. The cycle thresholds by on-chip extraction were 21.26 ± 0.45 for the N gene and 20.40 ± 0.25 for ORF1 detection, while they are 21.87 ± 0.22 and 21.08 ± 0.38, respectively, extracted by an operator using a commercialized kit (Fig. 4). It has approximately 0.5 difference between N and ORF1 gene detection, though from the same concentration of SARS-CoV-2. This may result from the different amplification efficiency of the two primers and the template sequences. By statistical analysis, on-chip assay could reach the thresholds with less recycles compared to off-chip assays, suggesting that the chip-based extraction performed better than off-chip extraction. Due to the small volume used in the chip, the magnetic beads may adequately come in contact with washing and elution buffers. Consequently, chip extracted RNA concentration and/or purity may be slightly higher than that by hand.On-chip multiplex amplification
For the on-chip amplification module, all pathogenic genes were positively detected at the lowest detected concentration (i.e., 15 copies per test), demonstrating a detection limit likely lower than 15 copies per test. Interestingly, on the fluorescence channel of HEX registration, the ORF1 gene was detected at the lowest cycle threshold among the four channels, which indicates the highest sensitivity of HEX on the device. The off-chip examination exhibited the highest sensitivity on the channel of ROX registration, suggesting that Flu A was the most sensitively detected pathogen in off-chip qPCR devices. Compared with the lower volume (∼2 μL) used in the VH 1.0 system, the volume of ∼5.55 μL per droplet was believed to increase the detection sensitivity. It was reported that LODs of VH 1.0 were close to 100 copies per test with either LAMP or multiplex qPCR.27,39 Assuming the RNA concentration in the eluent is 2.7 copies per μL (i.e., 15 copies per test), it has 4.5‰ possibility of taking an empty droplet in 2 μL according to Poisson distribution, which is four orders of magnitude compared with that of the 5.55 μL droplet. Ji et al. developed a microfluidic disc-direct RT-qPCR (dirRT-qPCR) assay for rapid multiplex detection of SARS-CoV-2 and influenza A and B viral infection in pharyngeal swab samples. It could detect 20 copies per test of all three pathogens in as little as 2 μL of samples,40 which are comparable with VH 2.0. However, it should be noted that these LODs were given by actual measurement results without statistical evaluation (e.g., Probit regression). Limited to the sample size (<20 reactions), LOD was also farfetched in this section and for off-chip complete workflow (in the next section); thus, they were not presented.Meanwhile, the low inter-assay and intra-assay variations (<5.00%, data not shown) demonstrated the homogeneity and reproducibility of the amplification module, which may be attributed to the uniformity of the droplet volume and the subtle thermal actuation unit. In addition, both on- and off-chip amplification have a high correlation coefficient (R2 >0.99, Table 1). The high linearity of amplification is prerequisite for quantitation and high sensitivity of integrated chips. Meanwhile, the linearity (i.e., R2) of on-chip amplification performed comparably with that of off-chip amplification indicated by two-tailed Student t-test coefficient, p = 0.13 > 0.05. Without manual intervention, the stochastic error induced by users' operation was minimized. The volumes of the on-chip droplets were measured based on the documented method41 (detailed in the ESI†). The digital microfluidics exhibited inherent superiority in precisely manipulating tiny droplets (i.e., droplet splitting and moving), conferring the stable performance of the RT-qPCR.
| Target | Standard curve | R 2 | Mean Ct of 15 copies/T | CV of 15 copies/T (%) | Detection rate |
|---|---|---|---|---|---|
| a It could be 0.999 if the curve does not include the lowest concentration. The negative controls (sample without template) did not acquire Ct values. | |||||
| On-chip | |||||
| Flu A | y = −3.00x + 39.44 | 0.971a | 35.60 | 0.37 | 16/16 |
| Flu B | y = −2.91x + 36.61 | 0.994 | 33.16 | 2.41 | 16/16 |
| SARS-CoV-2(N) | y = −3.52x + 41.25 | 0.991 | 36.94 | 3.54 | 16/16 |
| SARS-CoV-2(ORF1) | y = −3.18x + 32.57 | 0.996 | 28.85 | 0.25 | 16/16 |
| Off-chip | |||||
| Flu A | y = −4.09x + 32.28 | 0.990 | 27.68 | 4.54 | 20/20 |
| Flu B | y = −3.99x + 35.28 | 0.990 | 30.76 | 4.33 | 20/20 |
| SARS-CoV-2(N) | y = −4.17x + 39.15 | 0.994 | 34.25 | 3.72 | 19/20 |
| SARS-CoV-2(ORF1) | y = −4.11x + 39.46 | 0.993 | 34.56 | 1.79 | 19/20 |
Performance of VH 2.0 integrated platform
To evaluate the performance of integrated chips, series concentrations of pseudoviruses were tested and the limit of detection with 95% detection possibility (LOD95) was estimated by Probit regression (derived from >40 reactions). Same as the amplification module of the chip, the integrated chip also exhibited a high linearity (Table 2), illustrating the robustness of the extraction module as well as the whole chip. Both on-chip and off-chip detection could be linearly fitted with a coefficient of determination R2 higher than 0.99. The HEX registered the highest sensitivity among four fluorescence channels; therefore, the LOD95 of SARS-CoV-2 (ORF1) could achieve 302 copies per mL. Other pathogens can be stably detected as low as approximately 1000 copies per mL. The numbered test counts were all positively detected by an off-chip qPCR instrument, resulting in the non-deducible LOD95. However, they were most likely lower than the minimum detected concentrations (i.e., 1000 copies per mL for Flu B/ORF1/N and 100 copies per mL for Flu A). The LOD for integrated chip assay was conspicuously lower than that of amplification parts. Such a discrepancy was caused by integration of the extraction process, which inevitably led to the loss of nucleoid acid. Moreover, the impurities from samples or extraction reagents may also inhibit the amplification.42 The qPCR efficiencies ranged from 100% to 110% on-chip, while the off-chip assays varied from 94% to 97%. Fluorescence was not detected from any negative samples (derived from preservation solution) and samples spiked with other pathogens that were not the target of detection, declaring the high specificity of integrated chips.| Target | Standard curve | Linear range (copies per mL) | R 2 | Efficiency (%) | LOD95 (copies per mL) |
|---|---|---|---|---|---|
| NA: not available.a The coefficient of determination R2 lower than 0.99 was marked. | |||||
| On-chip | |||||
| Flu A | y = −3.10x + 43.54 | 102–106 | 0.996 | 110 | 1027.19 |
| Flu B | y = −3.20x + 43.90 | 102–106 | 0.995 | 106 | 1258.11 |
| SARS-CoV-2(N) | y = −3.31x + 42.56 | 102–106 | 0.989a | 100 | 943.91 |
| SARS-CoV-2(ORF1) | y = −3.24x + 40.01 | 102–107 | 0.991 | 104 | 302.22 |
| Off-chip | |||||
| Flu A | y = −3.48x + 46.16 | 103–106 | 1.000 | 94 | NA |
| Flu B | y = −3.47x + 49.24 | 102–106 | 0.996 | 94 | NA |
| SARS-CoV-2(N) | y = −3.42x + 44.22 | 103–107 | 0.993 | 96 | NA |
| SARS-CoV-2(ORF1) | y = −3.40x + 45.23 | 102–107 | 0.994 | 97 | NA |
PCR efficiency is the fraction of target molecules that are copied in one cycle, which is an important indicator of the quantitative process. Results showed that the values were all within the widely accepted range (i.e., 90–110%) that is recommended for qPCR reactions.43 Typically, efficiency is significantly impacted by the instruments, replicates, and concentrations used for standard curve generation.44 A total of 72 reactions in 9 independent chips for at least 5 concentrations were performed in this research, guaranteeing the reliability of the evaluation. Thus, in our experiment, the differences between on- and off-chip tests may be largely contributed by different instruments.
Performance in clinical application
Eleven SARS-CoV-2 and Flu A positive clinical specimens and four negative specimens from healthy people were tested. Results show a 100% coincidence between on- and off-chip assays for these two pathogen detection. Considering the satisfactory performances of the chip in detecting pseudoviruses, the Ct values measured by on- and off-chip methods presented a strong positive correlation as expected (Fig. 5 and Table S3†), especially for Flu A and ORF1 gene detection. The Pearson's coefficients were both higher than 0.9 with p values lower than 0.01. The N gene detection was a little off but the p value was low as well (0.0002), indicating the positive correlation between on- and off-assays is convincing. Numbered clinical specimens ineludibly introduced bias that may not cover all the possibilities, for instance, the weakly positive samples may be challenging for detection. The lowest pathogen load among tested samples appeared in No. 11 Flu A specimen (Table S4†), which was reported as 36.07 by VH 2.0. Also, the Ct value of No. 10 Flu A specimen was 35.11 which is close to the LOD95 (the corresponding Ct is 34.20). These two clinical samples were probabilistic detected by chip (7/8). Thanks to the eight parallel reaction models on chip, the possibilities of misdiagnosis were reduced. For SARS-CoV-2, the detection of two specific genes can double verify the accuracy of the results. Thus, the chip leads to a robust and convincing result in clinical applications.The stability of VH 2.0 biochips
Concentrations near the LOD95 were selected for stability assay. VH 2.0 biochips (with all reagents prestored) exhibited relatively stable Ct values at all registered fluorescence channels (Fig. 5), which were 34.09 ± 0.26 for Flu A, 34.03 ± 0.24 for Flu B, 32.64 ± 0.20 for SARS-CoV-2(N), and 33.12 ± 0.19 for SARS-CoV-2(ORF1). It demonstrated that the detection of Flu A/B and SARS-CoV-2 (ORF1/N) was reproducible and the chips were stable within the three-month monitoring duration. Although a slight fluctuation of Ct values was observed during the 3 months, the differences were statistically insignificant according to Student's t-test. All p-values within the cohort were greater than 0.05 (Table S5†). Also, the intra-assay CVs were in the range from 1.72% to 4.70% and inter-assay CVs were around 0.7%. Both were consistently acceptable (less than 5%) under various conditions.The stability assay is an essential step for scaling up the products. Ideally, the long-term stability test should be conducted on the day of product deterioration, demonstrating availability within the shelf-life. However, due to time constraints, only three months were monitored after the date of manufacture. Indeed, the stability of the chip includes the stability of all the reagents, filled oil, encapsulation, lifetime of the coating and electronics on the chips. To guarantee the superior performance of the products, a stability test report for each component was acquired from suppliers based on their qualified assays. On the other hand, we made many attempts to improve the stability during the fabrication, such as the reagent dehydration technology, brand of oil, sealing technology, etc. The DMF chips can be stably used under practical conditions for more than 1 year according to the first-generation product.
Comparison with existing platforms
The pandemic caused by SARS-CoV-2 has not only upended the daily lives of billions but also changed the diagnostic landscape. NATs have partially replaced conventional culture and antigen detection assays because of their capability of fast and accurate detection. A number of commercialized NAT-based platforms have been developed for identifying respiratory pathogens. Here, the VH 2.0 system was comprehensively compared with 9 POCT platforms, 16 panels including 9 US FDA-cleared or EUA respiratory panels, 3 previous versions of our product, 2 representative respiratory panels, and 2 panels developed by research groups (Table 3).| Manufacturer | Platform (device) | Panel (chip or cartridge) | Methodology | Targets | Turnaround time (min) | Throughput | NA extraction integrated | NA FDA authorized | Sensitivity (%) | Specificity (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| RV: respiratory viruses; RP: respiratory pathogens; FDA: Food and Drug Administration; EUA: emergency use authorization.a Need to pipet 200 μL of the sample to the sample delivery device and vortex for 10 s. | |||||||||||
| Digifluidic Biotech | VH 2.0 | RP panel v3 | DMF + multiplex RT-qPCR | 3 (max. 32) | 120 | 1–10 | Yes | No | 100 | 100.0 | This research |
| VH 1.0 | RP panel v1 | 4 | 90 | No | 94.0 | 100.0 | 52 | ||||
| RP panel v2 | 11 | 93.3 | 100.0 | 39 | |||||||
| Foodborne pathogens | DMF + LAMP | 4 | 30 | — | — | 27 | |||||
| Cepheid | GeneXpert | CoV-2 plus | MF + multiplex RT-qPCR | 1 | 20–30 | 1–80 | Yes | EUA | 95.2 | 95.0 | 53 |
| CoV-2/Flu/RSV plus | 3 | 45 | Yes | 100.0 | 98.3 | ||||||
| BioFire Diagnostics | Torch | RP panel | Nest-PCR-endpoint melt curve analysis | 21 | 60 | 1–12 | Yes | Yes | 84.5–97.9 | 96.1–100 | 54 |
| RP 2.1 plus panel | 23 | 45 | 97.1 | 99.3 | 55 | ||||||
| SPOTFIRE | R panel | 15 | 15 | 1–4 | 96.3–100 | 90.6–100 | 56 | ||||
| GenMark Dx | ePlex | RP panel | DMF + voltammetry | 20 | 90 | 3–24 | Noa | Yes | 95.4 | 99.7 | 54 and 57 |
| RP panel 2 | 21 | Yes | EUA | 91.4–98.3 | 99.2–99.7 | 57 and 58 | |||||
| Luminex Molecular Diagnostics | MAGPIX | xTAG RVP v1 | Fluorescence-labeled bead array + cytometry | 20 | 240 | 1–96 | No | Yes | 87.2–91.2 | 99.7–100 | 54 and 59 |
| xTAG RVP fast v2 | 19 | 78.8–92 | 97–99.6 | 54 and 60 | |||||||
| Baicare Biotechnology | Onestart | RP panel | DMF + monoplex RT-qPCR | 21 | 90 | 1 | Yes | No | — | 100.0 | 61 |
| Kansas State University | — | SARS-CoV-2 | DMF + multiplex RT-qPCR | 1 | 17/42 | 1 | No | — | — | — | 62 |
| Xiamen University | RCD | RP panel | PRA + Cas12a | 2 | 30 | 1 | No | — | 100 | 100 | 63 |
The chosen platforms were reported to be able to detect 1–23 respiratory pathogens in a single detection. Multiplex RT-qPCR as one of the most developed technologies has been widely used for routine diagnostic applications. However, limited by the number of fluorophores (typically 4 or 5 channels), only several targets can be detected per reaction. Thus, tagging oligonucleotide cleavage and extension (TOCE), dual priming oligonucleotide (DPO), and multiplex ligation-dependent probe amplification (MLPA) were developed to overcome such limitations by specially designed primers and probes. These technologies have been implemented in the products of Seegene such as SeeplexII RV15 and AnyplexII RV 16.45 However, the sophisticated primer and probe need experienced users and results have to be analysed by bundled software. Notably, microfluidic technology combined with RT-qPCR is an alternative methodology to increase the detection targets. In detail, DMF technology is propitious to generate many droplets. Combined with multiplex qPCR, the detection targets can be multiplied. As listed in Table 3, our DMF systems, Onestart from Baicare and a platform from Kansas State University adopted DMF plus multiplex qPCR as a basic principle. Apart from these, DMF is also compatible with other nucleic acid probing technologies, such as eSensor™46,47 from GenMark and recombinase polymerase amplification (RPA) coupled with DETECTR48 adopted by the RCD platform. Among them, eSensor™ relies on competitive DNA hybridization and electrochemical detection. It has been successfully commercialized, and its Respiratory Pathogen Panel 2 was authorized for emergency use by the US Food & Drug Administration (FDA) in 2020.49 However, both methods have their pros and cons. Due to electrochemistry utilizing molecular hybridization without amplification, it is capable of miniaturizing the device, reducing the risk of aerosol contamination, and enhancing the specificity; nevertheless, the most severe limitation is the detection limit, which is far below that of fluorometric assays.50 On the other hand, isothermal amplification such as real-time loop-mediated isothermal amplification (LAMP) and RPA dramatically shorten the reaction time while simplifying the structure of the heating module, whereas its sensitivity and specificity cannot be compared with qPCR51 and it is challenging for either primer design or absolute quantification. In addition to DMF, channel-based microfluidics (CMF) has been employed by GeneXpert from Cepheid and FilmArray from BioFire Diagnostics. CMF requires extra pumps or valves to control fluid, leading to a higher cost for each diagnosis compared with DMF.
The VH 2.0 instrument mounted 4 fluorescence channels and the tailor-made chip has 8 reaction rooms, resulting in the theoretical detection capacity reaching 32 targets. Further, the self-developed lysis buffer was designed for both viruses and bacteria, allowing the detection target to be expanded to infectious bacteria, such as S. pneumoniae, H. influenzae, S. aureus, S. enterica, and S. flexneri, which could be lysed by the buffer. Globally, evidence shows the flexibility and feasibility of DMF for detecting multiple pathogens.
The single run time of RP v3 of Digifluidic Biotech is approximately 2 h, which is relatively longer than that of other panels. It results from two reasons: (1) the prestored reagents are incompatible with fast PCR which mainly depends on the efficiency of enzymes; (2) lower power was selected for stable thermal control and matching with present PCR reagents. Unremitting efforts are made to shorten the turnaround time such as the development of fast/extreme qPCR, corresponding reagents, and improvement of the thermal module. To meet the requirements of point-of-care testing, VH 2.0 was designed as an independent cartridge integrating a nucleic acid extraction process on the chip as GeneXpert, FilmArray, and Onestart do. All the reagents required by extraction and reaction have been prestored in the corresponding cavities, thereby no extra pipetting is needed. Compared to VH 1.0, such update greatly shortened the hands-on time and simplified the diagnosis, lifting the need for experienced operators. Although batchwise testing systems (e.g., BD Viper™ XTR/LT system, Cobas® 4800/6800/8800 system, etc.) automatically possess high throughput per analysis station, these systems are costly and have large footprints which constrain scale-up testing and deployment in low-resource settings for mass screening. Coincidently, portable all-in-one POCT devices such as VH 2.0 complement batchwise systems.
Conclusions
This study developed an all-in-one diagnostic platform based on a macrochannel-to-digital microfluidic platform for automatic identification of multiple viruses. The special designs for on-chip thermal control and hybrid microfluidics enable stability and ease of fabrication. PCB and prefabricated blocks were employed for chip construction, which can reduce the cost and rejection rate during mass production. It is therefore beneficial to product commercialization. This research involved three respiratory pathogens (i.e., SARS-CoV-2, Flu A, and Flu B) for a proof-of-concept study. Through comprehensive evaluations, this platform exhibited a stable performance for its extraction, amplification modules, and sample-to-answer process. Notably, its sensitivity, specificity, detection limit, and amplification efficiency are comparable to that of the off-chip RT-qPCR process. The platform could easily extend to a maximum of 32 targets by simply changing the primers and probes. Furthermore, the platform dramatically reduces the hands-on time and the professional requirements of operators and equipment compared with traditional PCR. Overall, this research made certain academic and application merits for DMF in the field of diagnosis.Author contributions
Conceptualization and methodology: Dr. Cheng Dong, Dr. Tianlan Chen, Dr. Fei Li. Investigation, formal analysis, and writing – original draft: Yun Sun, Dr. Fei Li. Visualization: Dr. Fei Li, Dr. Chunzhao Chen, Dr. Cheng Dong. Funding acquisition, project administration, and resources: Dr. Tianlan Chen, Dr. Cheng Dong, Dr. Rui P. Martins, Dr. Pui-In Mak, Dongling Long. All authors were involved in writing – review & editing.Conflicts of interest
There are no conflicts to declare.Acknowledgements
We acknowledge the support from the Zhuhai Industry-University-Research Cooperation Project Grant (ZH22017002200012PWC), the Guangdong Basic and Applied Basic Research Foundation Grant (2021A1515110623), the Special Project of Commercialization of Scientific and Research Findings of Hong Kong and Macao in Guangdong Grant (2022A0505030022), and the Social Development Field Science and Technology Plan Project of Zhuhai (ZH22036201210189PWC).Notes and references
- R. Wölfel, V. M. Corman, W. Guggemos, M. Seilmaier, S. Zange, M. A. Müller, D. Niemeyer, T. C. Jones, P. Vollmar and C. Rothe, Nature, 2020, 581, 465–469 CrossRef PubMed.
- Z. Ballard and A. Ozcan, Nat. Biomed. Eng., 2018, 2, 629–630 CrossRef PubMed.
- K. J. Cortez, E. Roilides, F. Quiroz-Telles, J. Meletiadis, C. Antachopoulos, T. Knudsen, W. Buchanan, J. Milanovich, D. A. Sutton, A. Fothergill, M. G. Rinaldi, Y. R. Shea, T. Zaoutis, S. Kottilil and T. J. Walsh, Clin. Microbiol. Rev., 2008, 21, 157–197 CrossRef CAS.
- C. A. Muzny, R. J. Blackburn, R. J. Sinsky, E. L. Austin and J. R. Schwebke, Clin. Infect. Dis., 2014, 59, 834–841 CrossRef.
- S. Zayet, N. d. J. Kadiane-Oussou, Q. Lepiller, H. Zahra, P.-Y. Royer, L. Toko, V. Gendrin and T. Klopfenstein, Microbes Infect., 2020, 22, 481–488 CrossRef CAS PubMed.
- C. Rothe, M. Schunk, P. Sothmann, G. Bretzel, G. Froeschl, C. Wallrauch, T. Zimmer, V. Thiel, C. Janke and W. Guggemos, N. Engl. J. Med., 2020, 382, 970–971 CrossRef PubMed.
- Center for Systems Science and Engineering, COVID-19 dashboard, Center for Systems Science and Engineering at Johns Hopkins University, 2023, https://coronavirus.jhu.edu Search PubMed.
- J. Zhao, Q. Yuan, H. Wang, W. Liu, X. Liao, Y. Su, X. Wang, J. Yuan, T. Li and J. Li, Clin. Infect. Dis., 2020, 71, 2027–2034 CrossRef CAS PubMed.
- K. Choi, A. H. Ng, R. Fobel and A. R. Wheeler, Annu. Rev. Anal. Chem., 2012, 5, 413–440 CrossRef CAS.
- M. Washizu, IEEE Trans. Ind. Appl., 1998, 34, 732–737 CrossRef CAS.
- Q. Zhang, X. Xu, L. Lin, J. Yang, X. Na, X. Chen, L. Wu, J. Song and C. Yang, Lab Chip, 2022, 22, 1971–1979 RSC.
- X. Xu, L. Lin, J. Yang, W. Qian, R. Su, X. Guo, L. Cai, Z. Zhao, J. Song and C. Yang, Nano Today, 2022, 46, 101596 CrossRef CAS.
- J. Lamanna, E. Y. Scott, H. S. Edwards, M. D. Chamberlain, M. D. M. Dryden, J. Peng, B. Mair, A. Lee, C. Chan, A. A. Sklavounos, A. Heffernan, F. Abbas, C. Lam, M. E. Olson, J. Moffat and A. R. Wheeler, Nat. Commun., 2020, 11, 5632 CrossRef CAS PubMed.
- A. Das, C. Weise, M. Polack, R. D. Urban, B. Krafft, S. Hasan, H. Westphal, R. Warias, S. Schmidt, T. Gulder and D. Belder, J. Am. Chem. Soc., 2022, 144, 10353–10360 CrossRef CAS PubMed.
- W. Qiu and S. Nagl, ACS Sens., 2021, 6, 1147–1156 CrossRef CAS.
- S. Han, Q. Zhang, X. Zhang, X. Liu, L. Lu, J. Wei, Y. Li, Y. Wang and G. Zheng, Biosens. Bioelectron., 2019, 143, 111597 CrossRef CAS PubMed.
- S. Battat, D. A. Weitz and G. M. Whitesides, Lab Chip, 2022, 22, 530–536 RSC.
- R. Shen, S. Yi, P. Wang, P.-I. Mak, R. P. Martins and Y. Jia, TrAC, Trends Anal. Chem., 2022, 116826 Search PubMed.
- J. Li and C.-J. C. Kim, Lab Chip, 2020, 20, 1705–1712 RSC.
- Y. Xing, Y. Liu, R. Chen, Y. Li, C. Zhang, Y. Jiang, Y. Lu, B. Lin, P. Chen, R. Tian, X. Liu and X. Cheng, Lab Chip, 2021, 21, 1886–1896 RSC.
- R. Shen, Y. Jia, P.-I. Mak and R. P. Martins, Lab Chip, 2020, 20, 1928–1938 RSC.
- J. Li, N. S. Ha, T. L. Liu, R. M. van Dam and C.-J. ‘Cj’ Kim, Nature, 2019, 572, 507–510 CrossRef CAS.
- T.-W. Huang, T.-Y. Ho and K. Chakrabarty, Reliability-oriented broadcast electrode-addressing for pin-constrained digital microfluidic biochips, in 2011 IEEE/ACM International Conference on Computer-Aided Design (ICCAD), IEEE, 2011, pp. 448–455 Search PubMed.
- Z. Li, T. A. Dinh, T.-Y. Ho and K. Chakrabarty, Reliability-driven pipelined scan-like testing of digital microfluidic biochips, in 2014 IEEE 23rd Asian Test Symposium, IEEE, 2014, pp. 57–62 Search PubMed.
- S.-J. Wang, K. S.-M. Li and T.-Y. Ho, Design-for-Reliability and Probability-Based Fault Tolerance for Paper-Based Digital Microfluidic Biochips with Multiple Faults, in 2022 27th Asia and South Pacific Design Automation Conference (ASP-DAC), IEEE, 2022, pp. 62–67 Search PubMed.
- Y. Zai, C. Min, Z. Wang, Y. Ding, H. Zhao, E. Su and N. He, Lab Chip, 2022, 22, 3436–3452 RSC.
- M. Xie, T. Chen, X. Xin, Z. Cai, C. Dong and B. Lei, Food Control, 2022, 136, 108824 CrossRef CAS.
- V. Luu-The, N. Paquet, E. Calvo and J. Cumps, BioTechniques, 2005, 38, 287–293 CrossRef CAS.
- J. Pierson-Perry, J. Vaks, A. Durham, C. Fischer, C. Gutenbrunner, D. Hillyard, M. Kondratovich, P. Ladwig and R. Middleberg, Evaluation of Detection Capability for Clinical Laboratory Measurement Procedures, Clinical and Laboratory Standards Institute, Wayne, PA, USA, 2nd edn, 2012 Search PubMed.
- T.-M. Hsieh, C.-H. Luo, F.-C. Huang, J.-H. Wang, L.-J. Chien and G.-B. Lee, Sens. Actuators, B, 2008, 130, 848–856 CrossRef CAS.
- R. J. Shilton, V. Mattoli, M. Travagliati, M. Agostini, A. Desii, F. Beltram and M. Cecchini, Adv. Funct. Mater., 2015, 25, 5895–5901 CrossRef CAS.
- T. Markovic, S. Liu, I. Ocket and B. K. J. C. Nauwelaers, Int. J. Microw. Wirel. Technol., 2017, 9, 1591–1596 CrossRef.
- H. Kim, S. Vishniakou and G. W. Faris, Lab Chip, 2009, 9, 1230–1235 RSC.
- V. Miralles, A. Huerre, F. Malloggi and M.-C. Jullien, Diagnostics, 2013, 3, 33–67 CrossRef PubMed.
- M. Abdelgawad, M. W. L. Watson and A. R. Wheeler, Lab Chip, 2009, 9, 1046–1051 RSC.
- G. Sathyanarayanan, M. Haapala, C. Dixon, A. R. Wheeler and T. M. Sikanen, Adv. Mater. Technol., 2020, 5, 2000451 CrossRef CAS.
- Y. Fouillet, D. Jary, C. Chabrol, P. Claustre and C. Peponnet, Microfluid. Nanofluid., 2008, 4, 159–165 CrossRef.
- M. Shikida, K. Takayanagi, K. Inouchi, H. Honda and K. Sato, Sens. Actuators, B, 2006, 113, 563–569 CrossRef CAS.
- H. Huang, K. Huang, Y. Sun, D. Luo, M. Wang, T. Chen, M. Li, J. Duan, L. Huang and C. Dong, Micromachines, 2022, 13, 1650 CrossRef PubMed.
- M. Ji, Y. Xia, J. F.-C. Loo, L. Li, H.-P. Ho, J. He and D. Gu, RSC Adv., 2020, 10, 34088–34098 RSC.
- M. J. Kiani, A. Dehghan, M. Saadatbakhsh, S. J. Asl, N. M. Nouri and E. Pishbin, Lab Chip, 2023, 23(4), 748–760 RSC.
- I. G. Wilson, Appl. Environ. Microbiol., 1997, 63, 3741–3751 CrossRef CAS.
- J. Ruijter, C. Ramakers, W. Hoogaars, Y. Karlen, O. Bakker, M. Van den Hoff and A. Moorman, Nucleic Acids Res., 2009, 37, e45 CrossRef CAS.
- D. Svec, A. Tichopad, V. Novosadova, M. W. Pfaffl and M. Kubista, Biomol. Detect. Quantif., 2015, 3, 9–16 CrossRef CAS PubMed.
- C. H. Cho, B. Chulten, C. K. Lee, M. H. Nam, S. Y. Yoon, C. S. Lim, Y. Cho and Y. K. Kim, J. Clin. Virol., 2013, 57, 338–342 CrossRef CAS PubMed.
- S. D. Vernon, D. H. Farkas, E. R. Unger, V. Chan, D. L. Miller, Y.-P. Chen, G. F. Blackburn and W. C. Reeves, BMC Infect. Dis., 2003, 3, 12 CrossRef.
- J. E. Schmitz and Y.-W. Tang, Future Microbiol., 2018, 13, 1697–1708 CrossRef CAS PubMed.
- J. S. Chen, E. Ma, L. B. Harrington, M. Da Costa, X. Tian, J. M. Palefsky and J. A. Doudna, Science, 2018, 360, 436–439 CrossRef CAS.
- ePlex Respiratory Pathogen Panel 2 - Letter of Authorization, US Food & Drug Administration, 2020, https://www.fda.gov/media/142902/download#:~:text=This%20test%<?pdb_no 20is?>20is<?pdb END?>%20only%20authorized,and%20Cosmet Search PubMed.
- M. Lazerges and F. Bedioui, Anal. Bioanal. Chem., 2013, 405, 3705–3714 CrossRef CAS PubMed.
- J. Song, C. Liu, M. G. Mauk, S. C. Rankin, J. B. Lok, R. M. Greenberg and H. H. Bau, Clin. Chem., 2017, 63, 714–722 CrossRef CAS PubMed.
- W. Li, X. Jiang, H. Wang, M. Wang, A. Zhou, D. Luo, T. Chen and C. Dong, J. Trop. Med., 2022, 22, 1487–1492 Search PubMed.
- L. D. Noble, L. E. Scott, R. Munir, M. Du Plessis, K. Steegen, L. Hans, P. Marokane, P. Da Silva and W. S. Stevens, Diagnostics, 2022, 13, 34 CrossRef PubMed.
- E. B. Popowitch, S. S. O'Neill and M. B. Miller, J. Clin. Microbiol., 2013, 51, 1528–1533 CrossRef PubMed.
- bioMérieux, BIOFIRE® Respiratory 2.1 plus Panel, https://www.biomerieux-diagnostics.com/biofirer-respiratory-21-plus-panel Search PubMed.
- US FDA, 510(k) Premarket Notification of BIOFIRE SPOTFIRE Respiratory (R) Panel, 2023, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K213954 Search PubMed.
- J. Jarrett, K. Uhteg, M. S. Forman, A. Hanlon, C. Vargas, K. C. Carroll, A. Valsamakis and H. H. Mostafa, J. Clin. Virol., 2021, 135, 104737 CrossRef CAS.
- W. Zhen, E. Smith, R. Manji, D. Schron and G. J. Berry, J. Clin. Microbiol., 2020, 58, e00783-20 CrossRef.
- H.-K. Kim, S.-H. Oh, K. A. Yun, H. Sung and M.-N. Kim, J. Clin. Microbiol., 2013, 51, 1137–1141 CrossRef CAS PubMed.
- Luminex Corporation, xTAG® Respiratory Viral Panel (RVP) FAST v2, 2012, https://www.ld.ru/w/multiplex/RVP-FAST-v2-Brochure.pdf Search PubMed.
- E. Huang, Y. Wang, N. Yang, B. Shu, G. Zhang and D. Liu, Anal. Bioanal. Chem., 2021, 413, 1787–1798 CrossRef CAS PubMed.
- K.-L. Ho, H.-Y. Liao, H. M. Liu, Y.-W. Lu, P.-K. Yeh, J. Y. Chang and S.-K. Fan, Micromachines, 2022, 13, 196 CrossRef.
- Z. Sun, K.-F. Lin, Z.-H. Zhao, Y. Wang, X.-X. Hong, J.-G. Guo, Q.-Y. Ruan, L.-Y. Lu, X. Li, R. Zhang, C.-Y. Yang and B.-A. Li, Sci. China: Chem., 2022, 65, 630–640 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d3lc00728f |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2024 |