Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A graft-to strategy of poly(vinylphosphonates) on dopazide-coated gold nanoparticles using in situ catalyst activation†
Philipp Weingarten‡
a,
Sophie R. Thomas‡b,
Ana Luiza de Andrade Querino‡bc,
Kerstin Halamaa,
Moritz Kränzleina,
Angela Casini *b and
Bernhard Rieger
*b and
Bernhard Rieger *a
*a
aWACKER-Chair of Macromolecular Chemistry, Catalysis Research Center, School of Natural Sciences, Department of Chemistry, Technical University of Munich, Lichtenbergstraße 4, D-85748 Garching b. München, Germany. E-mail: rieger@tum.de
bChair of Medicinal and Bioinorganic Chemistry, Department of Chemistry, School of Natural Sciences, Technical University of Munich, Lichtenbergstraße 4, D-85748 Garching b. München, Germany. E-mail: angela.casini@tum.de
cDepartment of Chemistry, Universidade Federal de Minas Gerais, Belo Horizonte, MG 31270-901, Brazil
First published on 8th March 2024
Abstract
A modular synthetic pathway for poly(diethyl vinylphosphonates) grafting-to gold nanoparticles is presented. Utilising an azide-dopamine derivative as nanoparticle coating agent, alkyne–azide click conditions were used to covalently tether the polymer to gold nanoparticles leading to stable and well distributed colloids for different applications.
Polymers are materials that have constantly evolved for different applications from the beginning of their discovery until today. One of the most important classes of polymers are functional copolymers, which can be prepared through different methods and feature distinct functionalities, enabling their versatile use.1 Rare-earth metal-mediated group-transfer polymerisation (REM-GTP) of polar Michael monomers using yttrium-based catalysts has emerged as a valuable tool for synthesising functional polymers consisting of different microstructures.2 Thus, BCPs were carefully tuned to improve their physico-chemical properties, such as lower critical solution temperature (LCST), micelle size or pH responsive behaviour.3,4 Additionally, functional side-chains (allyl groups) and end-groups enhanced the scope of application for these polymers by facilitating the attachment of ionic moieties or biologically active molecules.5 Interestingly, non-toxic poly(diethyl vinylphosphonate) (PDEVP)-containing block copolymers (BCPs) hold promise as candidates for encapsulation and delivery of anti-cancer agents.4,6,7 Functional side-chains of such polymers could also enhance their drug delivery potential by creating crosslinked nanoparticles.6
To date, poly(vinyl phosphonate)-based polymers could be successfully applied for surface coatings in a few studies. For example, flame retardant properties were utilised as macroscopic polymer films on polycarbonates showing good performance due to their high thermal stability and morphological behaviour upon flame treatment.8 On a molecular scale, a grafting-from strategy including methyl methacrylate and DEVP was successfully developed to obtain polymer brushes on silicon surfaces with temperature and pH-responsive characteristics that might serve as proton conducting films or scaffolds for cell growth.9 Moreover, azide end-group functionalised PDEVP have been utilised to immobilise the polymer on multi-walled carbon nanotubes.10
To the best of our knowledge, no other poly(vinyl phosphonate)-based surface coating applications are known. However, coatings with functional poly(vinyl phosphonates) could be very relevant for different applications of nanomaterials. Thus, in search of anchoring groups for the modification of surfaces with functional PDEVP, dopamine appeared as a popular choice, specifically for grafting-to methods.11 The catechol moiety of polydopamine (PDA) offers strong binding on an array of different material surfaces, including superhydrophobic surfaces.12 For proof-of-concept experiments, gold nanoparticles (AuNPs) were selected as the surface material due to their advantageous properties, such as high surface area to volume ratio, established characterisation methods, and interesting electronic and physical properties.13 Moreover, examples of PDA-coated gold nanoparticles (PDA@AuNPs), synthesised by self-polymerisation of dopamine on the gold surfaces, are already reported to have well controlled Au core size and PDA coating layer thickness.14,15 Thus, PDA@AuNPs have been used in various applications, most commonly as therapeutic15,16 and sensing agents,17 with some reports of their use in catalysis18 and energy19 conversion.
Therefore, we report here the synthesis of alkyne-terminated poly(vinyl phosphonates) via in situ catalyst activation and a facile method to graft these polymers to the surface of plasmonic dopazide-stabilised AuNPs via click chemistry in aqueous conditions. Taking inspiration from our previous work,20 in situ σ-bond activation of [Cp2Y(CH2TMS)(THF)] (Y, TMS = trimethylsilyl, THF = tetrahydrofuran) using an alkyne-containing sym-collidine derivative was used to achieve alkyne-terminated polymers (Scheme 1 and S1†). The synthetic route was extended to different types of Michael monomers (DEVP, 2-vinyl pyridine (2VP), iso-propenyl oxazoline (iPOx), and a BCP consisting of DEVP and diallyl vinylphosphonate (DAlVP)) (Scheme S2†). Successful catalyst activation, end-group functionalisation, and polymerisation was demonstrated by electrospray ionisation mass spectrometry (ESI-MS), size exclusion chromatography (SEC), and 1H and 1H DOSY NMR spectroscopy on the purified polymers (Fig. S1–S16 and Table S1†). Apart from temperature responsive PiPOx and hydrophobic and pH responsive P2VP, PDEVP offers hydrophilicity, temperature responsiveness and a high degree of biocompatibility,21 and therefore was selected for anchoring to AuNPs. To tether the functional PDEVP to the dopamine coated AuNPs, a dopamine derivative was selected,22 and its amino group replaced with an azido moiety via a diazo-transfer,23 providing a robust functional group for popular click reactions (Scheme 1, see ESI for details†).
To demonstrate the presence of the viable alkyne as an end-group, a qualitative study was performed utilising ruthenium-catalysed azide–alkyne click (RuAAC) conditions to react the alkyne with the fluorescent compound 3-azido-7-hydroxycoumarin (see ESI for details, Fig. S17†). Photoluminescence studies of the obtained polymer showed fluorescence at the expected wavelength (Fig. S18†). These reaction conditions were the basis for the decoration of dopazide-coated AuNPs, which is the first step to creating hybrid materials consisting of PDEVP and AuNPs.
Before the click reaction can be performed, the dopazide ligand must first be coated onto the AuNP surface via a ligand exchange reaction with citrate stabilised AuNPs (citrate@AuNPs) to achieve dopazide@AuNPs (Scheme 1).24 The water-soluble ruby-red dopazide@AuNPs were then purified by centrifugation to remove the excess of free dopazide and citrate. The final step to conjugate the alkyne terminated polymer to the dopazide@AuNPs was performed via a click reaction with the Cp*RuCl(cod) catalyst using similar conditions as described above (Scheme 1, see ESI for details†).
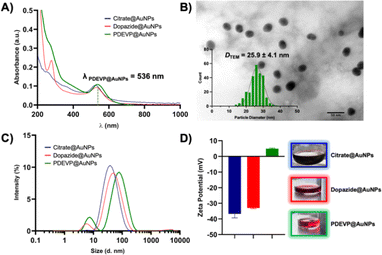
The characterisation of the PDEVP@AuNPs was then attained via different methods and directly compared to the precursor AuNPs (dopazide@AuNPs and citrate@AuNPs), as summarised in Table 1. UV-visible absorption spectroscopy was first used to evaluate the presence of AuNPs due to the typical surface plasmon resonance (SPR) band at 520–530 nm (Fig. 1A), indicative of spherical NPs with an average size of ca. 20 nm.25 The PDEVP@AuNPs feature a SPR band around 536 nm, comparable to the band of the dopazide@AuNPs and citrate@AuNPs (Fig. 1A). Furthermore, a broad peak around 270 nm appears that can be attributed to the presence of the polymer; slightly blue-shifted compared to the intense π–π* intraligand transition of the polymer alone (ca. 290 nm) and of the dopazide@AuNPs (ca. 280 nm) (Fig. S19†).
| Sample | DTEM [nm] | Dh [nm] | ζ-Potential [mV] |
|---|---|---|---|
| Citrate@AuNPs | 25.8 ± 7.1 | 32 ± 0.9 | −38.2 ± 2.5 |
| Dopazide@AuNPs | 27.8 ± 7.5 | 48 ± 0.5 | −31.5 ± 1.0 |
| PDEVP@AuNPs | 25.9 ± 4.1 | 78.8 ± 1.5 | +4.9 ± 0.4 |
| Test (i) (polymer w/o alkyne) | — | 122.9 ± 9.8 | +3.1 ± 0.3 |
| Test (ii) (no cat.) | — | 70.0 ± 2.0 | −6.7 ± 0.4 |
The average particle size and size distribution of the PDEVP@AuNPs was next evaluated by transmission electron microscopy (TEM) (Table 1). A representative TEM image is reported in Fig. 1B, showing the well-distributed monodisperse quasi-spherical PDEVP@AuNPs with an average diameter (DTEM) of 25.9 ± 4.1 nm, which is comparable to the precursor AuNPs (DTEM dopazide@AuNPs = 27.8 ± 7.5 nm and DTEM citrate@AuNPs = 25.8 ± 7.1 nm, Fig. S20, S21,† and Table 1) and in line with literature values.26 Additionally, the TEM images showed no apparent aggregation of the AuNPs, highlighting their stability throughout the reaction steps. Furthermore, dynamic light scattering (DLS) can be used to measure both the core NP size and the size of the surface coating of the AuNPs in solution. Via this method, analysis of an aqueous solution of the PDEVP@AuNPs revealed an average hydrodynamic diameter (Dh) of 78.8 ± 1.5 (Fig. 1C and Table 1), suggesting a polymer shell-AuNP structure. This is also apparent, albeit to a lesser extent, in the dopazide@AuNPs and citrate@AuNPs with smaller Dh values of 48 ± 0.5 and 32 ± 0.9 nm, respectively (Fig. 1C and Table 1), due to the shorter linker and stabiliser sizes. The presence of the polymer clicked to the dopazide@AuNPs was further confirmed by measuring the surface ζ-potential of the different NP solutions (Fig. 1D and Table 1). An aqueous PDEVP@AuNP solution showed a small ζ-potential of +4.9 ± 0.4 mV, as expected from a neutral polymer shell. This is at variance to the precursor AuNPs featuring negative ζ-potentials (Fig. 1D and Table 1), as also seen for similar systems in the literature.27
Fourier transform infrared (FTIR) spectra showed the loss of the azide stretch at ca. 2000 cm−1 from the dopazide ligand after the click reaction to form the PDEVP@AuNPs (Fig. S22†). Furthermore, to estimate the degree of ligand functionalisation on the PDEVP@AuNPs, thermogravimetric analysis (TGA) was performed. From the TGA-profile (Fig. S23†), one broad event is observed starting at ca. 100 °C until 472 °C, with a total weight loss of approximately 30 wt%, likely to be attributed to the loss of the organic bound ligands.
To rule out the possibility that the polymer was merely adsorbed to the NP surface, a control experiment was performed using the same reaction conditions as the title reaction, but featuring a structurally similar polymer without the alkyne moiety (test (i)).20 Upon reaction completion, a colour change of the solution was observed, from red to pale pink, and the solution became slightly turbid, revealing the presence of suspended particles. The results of the characterisation are summarised in Table 1 (test i). The UV-vis absorption spectrum of test (i) appeared comparable to that of the PDEVP@AuNPs with an SPR band at 536 nm and a broad peak in the UV region at ca. 260–270 nm (Fig. S24†); the latter being attributable to polymer coating the NP surfaces. The presence of suspended/aggregated NPs was further evidenced by the DLS measurement, with test (i) displaying a Dh of 122.9 ± 9.8 nm, significantly larger than the PDEVP@AuNPs (Table 1 and Fig. S25A†). The ζ-potential was slightly positive, similar to what was observed for the PDEVP@AuNPs (Fig. S25B†). Furthermore, the IR spectrum showed a significant concentration of the dopazide moiety in the supernatant (Fig. S26†), in line with the observed colour change and turbidity of the solution upon polymer addition.
A second control reaction was performed using the dopazide@AuNPs and the polymer with an alkyne moiety, in absence of the catalyst (test ii). Interestingly, even without the catalyst, the click reaction can occur, albeit to a lower extent.28 Nevertheless, the incomplete click reaction can be observed in the UV-vis spectrum in Fig. S27,† featuring two absorption bands for test ii at 230 and 280 nm, likely corresponding to a combination of the PDEVP polymer alone, dopazide@AuNPs and PDEVP@AuNPs. The DLS and ζ-potential results (Table 1) further support this hypothesis (Fig. S28A and B,† respectively). Additionally, the FTIR spectrum of test ii still displays the azide peak at ca. 2000 cm−1, as expected due to the reaction only occurring partially (Fig. S29†). Overall, these results point towards the successful engrafting of the polymer on the AuNP surfaces via click chemistry to provide a well-defined nanostructure.
In summary, we report here a straightforward protocol for grafting alkyne terminated polyesters to the surface of plasmonic dopazide-stabilised AuNPs via click chemistry in aqueous conditions in situ. Comprehensive characterisation of the PDEVP@AuNPs and its precursors was performed by different methods to confirm the successful click reaction, demonstrating the formation of single polymeric specie, as well as the stability of the NPs during the multiple steps. Combining the tuneability of the BCPs' properties with the herein reported alkyne at the polymer's end-group means that precise modification under mild conditions is possible. Thus, the accessibility to orthogonal functionalisation of surfaces, proteins, and biologically relevant peptides or drugs with BCPs is enhanced. More specifically, PDEVP@AuNPs are interesting for different applications, such as the obtainment of nanosensors via the ordered deposition of nanoparticles on nanofibers,29 or other highly curved surfaces, as well as for applications in continuous flow catalysis or lithography on silicon substrates.30 Further, the possibility to stably coat surfaces with BCPs is interesting to confer different properties to the material depending on the type of monomers and their functionalisation. From the perspective of the AuNPs for biomedical applications, such as drug delivery, imaging or for the photothermal therapy of tumours,31 colloids featuring a dense brush polymer surface can display high stability and tuneable pharmacokinetic properties. In general, polymeric ligands improve the long-term stability of AuNPs and increase hydrophilicity of the surface. In addition, AuNPs size can be adjusted with polymer coating. Finally, BCPs could represent a new class of antifouling coatings to render NPs more biocompatible.32 All these features can be rationally implemented in AuNPs exploiting BCPs' highly versatile combinatorial properties, to achieve finely engineered nanomaterials.
Conflicts of interest
There are no conflict of interest to declare.Acknowledgements
Authors acknowledge support from the TUM Innovation Network “Artificial Intelligence Powered Multifunctional Material Design” (ARTEMIS). S. R. T. acknowledges the TUM Global Postdoc Fellowship Scheme for funding. A. L. A. Q. acknowledges the funding from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES, Finance Code 001). M. K. is grateful for the PhD scholarship from the Studienstiftung des deutschen Volkes. Dr Carsten Peters (TUM) is kindly acknowledged for his assistance recording TEM data.References
- (a) F. H. Schacher, P. A. Rupar and I. Manners, Angew. Chem., Int. Ed., 2012, 51, 7898–7921 CrossRef CAS PubMed; (b) G. Gaucher, M.-H. Dufresne, V. P. Sant, N. Kang, D. Maysinger and J.-C. Leroux, J. Controlled Release, 2005, 109, 169–188 CrossRef CAS PubMed; (c) H.-C. Kim, S.-M. Park and W. D. Hinsberg, Chem. Rev., 2010, 110, 146–177 CrossRef CAS PubMed; (d) B. S. Soller, S. Salzinger and B. Rieger, Chem. Rev., 2016, 116, 1993–2022 CrossRef CAS PubMed.
- (a) F. Adams, P. T. Altenbuchner, P. D. L. Werz and B. Rieger, RSC Adv., 2016, 6, 78750–78754 RSC; (b) N. Zhang, S. Salzinger and B. Rieger, Macromolecules, 2012, 45, 9751–9758 CrossRef CAS; (c) M. Kränzlein, T. M. Pehl, F. Adams and B. Rieger, Macromolecules, 2021, 54, 10860–10869 CrossRef.
- (a) A. Saurwein, A. Schaffer, C. Wieser and B. Rieger, RSC Adv., 2021, 11, 1586–1594 RSC; (b) A. Schaffer, M. Kränzlein and B. Rieger, Macromolecules, 2020, 53, 4345–4354 CrossRef CAS.
- P. T. Altenbuchner, P. D. L. Werz, P. Schöppner, F. Adams, A. Kronast, C. Schwarzenböck, A. Pöthig, C. Jandl, M. Haslbeck and B. Rieger, Chem.–Eur. J., 2016, 22, 14576–14584 CrossRef CAS PubMed.
- (a) C. Schwarzenböck, A. Schaffer, P. Pahl, P. J. Nelson, R. Huss and B. Rieger, Polym. Chem., 2018, 9, 284–290 RSC; (b) K. Halama, A. Schaffer and B. Rieger, RSC Adv., 2021, 11, 38555–38564 RSC; (c) J. Kim, H. Y. Jung and M. J. Park, Macromolecules, 2020, 53, 746–763 CrossRef CAS.
- C. Schwarzenböck, P. J. Nelson, R. Huss and B. Rieger, Nanoscale, 2018, 10, 16062–16068 RSC.
- (a) F. Adams, P. Pahl and B. Rieger, Chem.–Eur. J., 2018, 24, 509–518 CrossRef CAS PubMed; (b) P. T. Altenbuchner, P. D. L. Werz, P. Schöppner, F. Adams, A. Kronast, C. Schwarzenböck, A. Pöthig, C. Jandl, M. Haslbeck and B. Rieger, Chem.–Eur. J., 2016, 22, 14576–14584 CrossRef CAS PubMed.
- D. Lanzinger, S. Salzinger, B. S. Soller and B. Rieger, Ind. Eng. Chem. Res., 2015, 54, 1703–1712 CrossRef CAS.
- N. Zhang, S. Salzinger, F. Deubel, R. Jordan and B. Rieger, J. Am. Chem. Soc., 2012, 134, 7333–7336 CrossRef CAS PubMed.
- M. Kränzlein, T. M. Pehl, K. Halama, P. F. Großmann, T. Kratky, A. M. Mühlbach and B. Rieger, Macromol. Mater. Eng., 2023, 308, DOI:10.1002/mame.202200635.
- (a) A. B. Asha, Y. Chen and R. Narain, Chem. Soc. Rev., 2021, 50, 11668–11683 RSC; (b) T. M. Lutz, C. Kimna, A. Casini and O. Lieleg, Mater. Today Bio, 2022, 13, 100203 CrossRef CAS PubMed.
- (a) F. Wu, J. Li, K. Zhang, Z. He, P. Yang, D. Zou and N. Huang, ACS Appl. Mater. Interfaces, 2016, 8, 109–121 CrossRef CAS PubMed; (b) Y. Liu, K. Ai and L. Lu, Chem. Rev., 2014, 114, 5057–5115 CrossRef CAS PubMed; (c) S. M. Kang, N. S. Hwang, J. Yeom, S. Y. Park, P. B. Messersmith, I. S. Choi, R. Langer, D. G. Anderson and H. Lee, Adv. Funct. Mater., 2012, 22, 2949–2955 CrossRef CAS PubMed.
- S. J. Amina and B. Guo, Int. J. Nanomed., 2020, 15, 9823–9857 CrossRef CAS.
- X. Liu, J. Cao, H. Li, J. Li, Q. Jin, K. Ren and J. Ji, ACS Nano, 2013, 7, 9384–9395 CrossRef CAS.
- N. N. Heris, L. Baghani, F. Khonsari, R. Varshochian, R. Dinarvand and F. Atyabi, J. Drug Delivery Sci. Technol., 2023, 87, 104869 CrossRef CAS.
- L. N. Nguyen, N. Kaushik, P. Lamichhane, S. Mumtaz, R. Paneru, P. Bhartiya, J. S. Kwon, Y. K. Mishra, L. Q. Nguyen, N. K. Kaushik and E. H. Choi, Green Chem., 2020, 22, 6588–6599 RSC.
- (a) S. Xu, G. Zhang, B. Fang, Q. Xiong, H. Duan and W. Lai, ACS Appl. Mater. Interfaces, 2019, 11, 31283–31290 CrossRef CAS PubMed; (b) G. Xu, J. Hou, Y. Zhao, J. Bao, M. Yang, H. Fa, Y. Yang, L. Li, D. Huo and C. Hou, Sens. Actuators, B, 2019, 287, 428–436 CrossRef CAS; (c) H.-C. Lee, C.-Y. Wang and C.-H. Lin, Sens. Actuators, B, 2014, 191, 204–210 CrossRef CAS; (d) J. Zhang, L. Mou and X. Jiang, Anal. Chem., 2018, 90, 11423–11430 CrossRef CAS PubMed.
- (a) S. Mei, Z. Kochovski, R. Roa, S. Gu, X. Xu, H. Yu, J. Dzubiella, M. Ballauff and Y. Lu, Nano-Micro Lett., 2019, 11, 83 CrossRef CAS PubMed; (b) B.-Z. Yang, Z.-Y. Su and A. F.-J. Jou, ACS Appl. Bio Mater., 2021, 4, 2821–2828 CrossRef CAS PubMed.
- M. Xu, L. Yan, Y. Zhu, Y. Li, X. Song and L. Yin, J. Mater. Sci.: Mater. Electron., 2020, 31, 6698–6705 CrossRef CAS.
- B. S. Soller, S. Salzinger, C. Jandl, A. Pöthig and B. Rieger, Organometallics, 2015, 34, 2703–2706 CrossRef CAS.
- C. Schwarzenböck, S. I. Vagin, W. R. Heinz, P. J. Nelson and B. Rieger, Macromol. Rapid Commun., 2018, 39, e1800259 CrossRef PubMed.
- R. Mrówczyński, R. Markiewicz and J. Liebscher, Polym. Int., 2016, 65, 1288–1299 CrossRef.
- S. Pagoti, D. Dutta and J. Dash, Adv. Synth. Catal., 2013, 355, 3532–3538 CrossRef CAS.
- (a) R. P. Briñas, M. Maetani and J. J. Barchi, J. Colloid Interface Sci., 2013, 392, 415–421 CrossRef PubMed; (b) J.-G. Wang, X. Hua, M. Li and Y.-T. Long, ACS Appl. Mater. Interfaces, 2017, 9, 3016–3023 CrossRef CAS PubMed.
- S. Link and M. A. El-Sayed, J. Phys. Chem. B, 1999, 103, 4212–4217 CrossRef CAS.
- K. H. S. Sy, L. W. C. Ho, W. C. Y. Lau, H. Ko and C. H. J. Choi, Langmuir, 2018, 34, 14033–14045 CrossRef CAS PubMed.
- (a) C. Li, Z. Liu and P. Yao, RSC Adv., 2016, 6, 33083–33091 RSC; (b) C. K. K. Choi, J. Li, K. Wei, Y. J. Xu, L. W. C. Ho, M. Zhu, K. K. W. To, C. H. J. Choi and L. Bian, J. Am. Chem. Soc., 2015, 137, 7337–7346 CrossRef CAS PubMed.
- I. L. Librando, A. G. Mahmoud, S. A. C. Carabineiro, M. F. C. Da Guedes Silva, F. J. Maldonado-Hódar, C. F. G. C. Geraldes and A. J. L. Pombeiro, Catalysts, 2022, 12, 45 CrossRef CAS.
- (a) H. Zhu, J.-F. Masson and C. G. Bazuin, ACS Appl. Nano Mater., 2020, 3, 516–529 CrossRef CAS; (b) H. Zhu, F. Lussier, C. Ducrot, M.-J. Bourque, J. P. Spatz, W. Cui, L. Yu, W. Peng, L.-É. Trudeau, C. G. Bazuin and J.-F. Masson, ACS Appl. Mater. Interfaces, 2019, 11, 4373–4384 CrossRef CAS PubMed.
- (a) E. Han, M. Leolukman, M. Kim and P. Gopalan, ACS Nano, 2010, 4, 6527–6534 CrossRef CAS PubMed; (b) J. Zhou, C. Zhang and Y. Wang, Polym. Chem., 2019, 10, 1642–1649 RSC.
- (a) O. S. Muddineti, B. Ghosh and S. Biswas, Int. J. Pharm., 2015, 484, 252–267 CrossRef CAS PubMed; (b) G. Ajnai, A. Chiu, T. Kan, C.-C. Cheng, T.-H. Tsai and J. Chang, J. Exp. Clin. Med., 2014, 6, 172–178 CrossRef CAS.
- P. Bevilacqua, S. Nuzzo, E. Torino, G. Condorelli, M. Salvatore and A. M. Grimaldi, Nanomaterials, 2021, 11, 780 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Experimental section, Fig. S1–S29. See DOI: https://doi.org/10.1039/d4ra01116c |
| ‡ These authors contributed equally to this manuscript. |
| This journal is © The Royal Society of Chemistry 2024 |