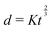Dynamic duos: the building blocks of dimensional mechanics
Marc A.
Fardin
 *ab,
Mathieu
Hautefeuille
*ab,
Mathieu
Hautefeuille
 c and
Vivek
Sharma
c and
Vivek
Sharma
 bd
bd
aCNRS, Institut Jacques Monod, Université de Paris, F-75013 Paris, France. E-mail: marc-antoine.fardin@ijm.fr
bThe Academy of Bradylogists, France
cInstitut de Biologie Paris Seine, UMR 7622, Sorbonne Université, 7 quai Saint Bernard, 75005 Paris, France
dDepartment of Chemical Engineering, University of Illinois at Chicago, Chicago, Illinois 60608, USA
First published on 17th June 2024
Abstract
Mechanics studies the relationships between space, time, and matter. These relationships can be expressed in terms of the dimensions of length 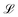 , time
, time 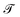 , and mass
, and mass 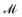 . Each dimension broadens the scope of mechanics. Historically, mechanics emerged from geometry, which considers quantities like lengths or areas, with dimensions of the form
. Each dimension broadens the scope of mechanics. Historically, mechanics emerged from geometry, which considers quantities like lengths or areas, with dimensions of the form  . With the Renaissance, quantities combining space and time were considered, like speed, acceleration and later diffusivity, all of the form
. With the Renaissance, quantities combining space and time were considered, like speed, acceleration and later diffusivity, all of the form 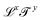 . Eventually, mechanics reached its full potential by including “mass-carrying” quantities such as mass, force, momentum, energy, action, power, viscosity, etc. These standard mechanical quantities have dimensions of the form
. Eventually, mechanics reached its full potential by including “mass-carrying” quantities such as mass, force, momentum, energy, action, power, viscosity, etc. These standard mechanical quantities have dimensions of the form 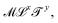 where x and y are integers. In this contribution, we show that, thanks to this dimensional structure, these mass-carrying quantities can be readily arranged into a table such that x and y increase along the row and column, respectively. Ratios of quantities in the same rows provide characteristic lengths, while those in the same columns yield characteristic times, encompassing a great variety of physical phenomena from atomic to astronomical scales. Most generally, we show that selecting duos of mechanical quantities that are neither on the same row nor column of the table yields dynamics, where one mechanical quantity is understood as impelling motion, while the other impedes it. The force and the mass are the prototypes of impelling and impeding factors, but many other duos are possible. We present examples from the physical and biological realms, including planetary motion, sedimentation, explosions, fluid flows, turbulence, diffusion, cell mechanics, capillary and gravity waves, and spreading, pinching, and coalescence of drops and bubbles. This review provides a novel synthesis revealing the power of scaling or dimensional analysis, to understand processes governed by the interplay of two mechanical quantities. This elementary decomposition of space, time and motion into pairs of mechanical factors is the foundation of “dimensional mechanics”, a method that this review wishes to promote and advance. Pairs are the fundamental building blocks, but they are only a starting point. Beyond this simple world of mechanical duos, we envision a richer universe that beckons with an interplay of three, four, or more quantities, yielding multiple characteristic lengths, times, and kinematics. This review is complemented by online video lectures, which initiate a discussion on the elaborate interplay of two or more mechanical quantities.
where x and y are integers. In this contribution, we show that, thanks to this dimensional structure, these mass-carrying quantities can be readily arranged into a table such that x and y increase along the row and column, respectively. Ratios of quantities in the same rows provide characteristic lengths, while those in the same columns yield characteristic times, encompassing a great variety of physical phenomena from atomic to astronomical scales. Most generally, we show that selecting duos of mechanical quantities that are neither on the same row nor column of the table yields dynamics, where one mechanical quantity is understood as impelling motion, while the other impedes it. The force and the mass are the prototypes of impelling and impeding factors, but many other duos are possible. We present examples from the physical and biological realms, including planetary motion, sedimentation, explosions, fluid flows, turbulence, diffusion, cell mechanics, capillary and gravity waves, and spreading, pinching, and coalescence of drops and bubbles. This review provides a novel synthesis revealing the power of scaling or dimensional analysis, to understand processes governed by the interplay of two mechanical quantities. This elementary decomposition of space, time and motion into pairs of mechanical factors is the foundation of “dimensional mechanics”, a method that this review wishes to promote and advance. Pairs are the fundamental building blocks, but they are only a starting point. Beyond this simple world of mechanical duos, we envision a richer universe that beckons with an interplay of three, four, or more quantities, yielding multiple characteristic lengths, times, and kinematics. This review is complemented by online video lectures, which initiate a discussion on the elaborate interplay of two or more mechanical quantities.
1 Introduction
Mechanics is the bedrock of physics and exerts influence across all sciences. Mechanics has had such a far reaching impact on our understanding of the natural world that it is hard to contain it under a single definition. In the 19th century, an effort to bring new disciplines like thermodynamics and electromagnetism into its fold prompted Fourier,1 Gauss and Weber,2 Maxwell and Kelvin,3,4 and their contemporaries to formulate one of the most commonly accepted definitions of mechanics.3,5 Mechanics deals with the relationships between space, time and matter, usually quantified by the dimensions of length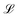 , time
, time 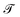 , and mass
, and mass 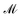 .
.
Mechanics in a general sense includes geometric quantities, with dimensions of the form 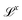 (lengths, areas, etc.). More broadly, mechanics also includes kinematic quantities, with dimensions
(lengths, areas, etc.). More broadly, mechanics also includes kinematic quantities, with dimensions 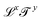 (speed, acceleration, diffusivity, etc.). These kinematic quantities describe motion, but without any reference to the “causes” of these motions. The quest for these causes led to the definition of mass, and all its derivatives: force, density, momentum, energy, action, power, stiffness, pressure, viscosity, etc. The bestiary of mechanics includes many creatures, all cast from the same mold. All these mechanical quantities have dimensions of the form
(speed, acceleration, diffusivity, etc.). These kinematic quantities describe motion, but without any reference to the “causes” of these motions. The quest for these causes led to the definition of mass, and all its derivatives: force, density, momentum, energy, action, power, stiffness, pressure, viscosity, etc. The bestiary of mechanics includes many creatures, all cast from the same mold. All these mechanical quantities have dimensions of the form 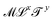 ; they are “mass-carrying quantities”. As we will see in this review, this shared structure allows a representation of the mechanical quantities in a plane with coordinates x and y, representing the exponents of the space and time dimensions. Moreover, since x and y are usually integers, the standard mechanical quantities can be arranged into a table, which can act as a great guide for researchers and teachers, and a perfect cheat sheet for students. We have spent the last three years toying with this enigmatic map of mechanical quantities. We constructed this table in order to provide a Rosetta stone to help translate knowledge across the boundaries of the diverse sub-fields of science. We invite readers to contribute to this table, and to suggest additions or modifications.
; they are “mass-carrying quantities”. As we will see in this review, this shared structure allows a representation of the mechanical quantities in a plane with coordinates x and y, representing the exponents of the space and time dimensions. Moreover, since x and y are usually integers, the standard mechanical quantities can be arranged into a table, which can act as a great guide for researchers and teachers, and a perfect cheat sheet for students. We have spent the last three years toying with this enigmatic map of mechanical quantities. We constructed this table in order to provide a Rosetta stone to help translate knowledge across the boundaries of the diverse sub-fields of science. We invite readers to contribute to this table, and to suggest additions or modifications.
Our investigations led us to reformulate the dimensional approach to mechanics, which we are sharing through a series of lectures on a https://www.youtube.com/@naturesnumbers channel that we created for this purpose (https://www.youtube.com/@naturesnumbers). These lectures explain in detail how to use this table to identify the “causes” of a wide range of motions, enabling the transformation of a kinematic description into a dynamical understanding. These videos serve as a supplementary material to this review, which focuses on the decomposition of geometric or kinematic quantities into ratios of mechanical quantities. From a dimensional perspective, this kind of decomposition is elementary, but it has far reaching consequences on the understanding of the relationship between mechanics and motion, and it provides a systematic way to approach the “causes” of motion.
The basis for this dimensional approach to mechanics dates back to Archimedes. To measure volume Ω, Archimedes proposed expressing it as a ratio, Ω = m/ρ, where m is the mass of the object and ρ is its density. This old formula seems so elementary today that we do not realize the great leap that it encompasses: a geometric quantity (the volume) is derived from a ratio of two mechanical quantities (the mass and the density). Dimensionally, the logic is flawless: 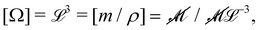 where the brackets return the dimensions of their content. The extra dimension of mass is a sort of “dummy” dimension, disappearing from the final result, a very useful intermediary in the computation.
where the brackets return the dimensions of their content. The extra dimension of mass is a sort of “dummy” dimension, disappearing from the final result, a very useful intermediary in the computation.
Almost two thousand years after Archimedes, Newton extended this logic even further. What Newton sought to compute was not a geometric quantity, but a kinematic one, acceleration a; however, he used the same principle. He expressed acceleration as the ratio between two mechanical quantities: a = F/m. Again, the dimensions match: 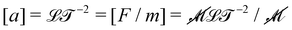 . The example is so classical that it may not seem too impressive today.
. The example is so classical that it may not seem too impressive today.
Fast forward almost three centuries to the 1940s and consider this other example, often found in textbooks on dimensional analysis.6 The Second World War is raging and the British physicist G. I. Taylor is trying to compute the dynamics of an explosion blast. Experiments suggest that the radius of explosion follows a ‘power law’ of time, d(t) ≃ Ktα. To understand the value of the kinematic prefactor K, Taylor uses the old trick again. In this context, Taylor identifies the energy E of the bomb and the density ρ of the ambient air as the relevant mechanical parameters. Then, the dimensions of the mechanical ratio provide an answer: 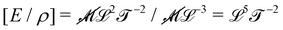 . Taylor concludes that
. Taylor concludes that 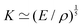 , that is
, that is 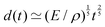 .7,8 Not so trivial anymore!
.7,8 Not so trivial anymore!
From Archimedes to Newton and Taylor, the procedure remains the same. A geometric or more broadly kinematic quantity (without any mass dimension) can always be expressed as a ratio of mechanical quantities. The only thing that varies from one example to another is the pair of mechanical quantities that are involved in the decomposition. In any case, the dimension of mass comes to the rescue, providing a means to understand sizes, durations and motions of all sorts, from a ratio, or “balance”, or “struggle”, between “competing” mechanical factors.
Even if we restrict ourselves to the standard mechanical quantities in Table 1, there are hundreds of possible pairs, and quite a few with a rich history. The purpose of this review is to discuss a few of these pairs. Each pair tells a unique story, synthesizing different “physics”, and retracing the steps of those who sought to explore this mechanical landscape.
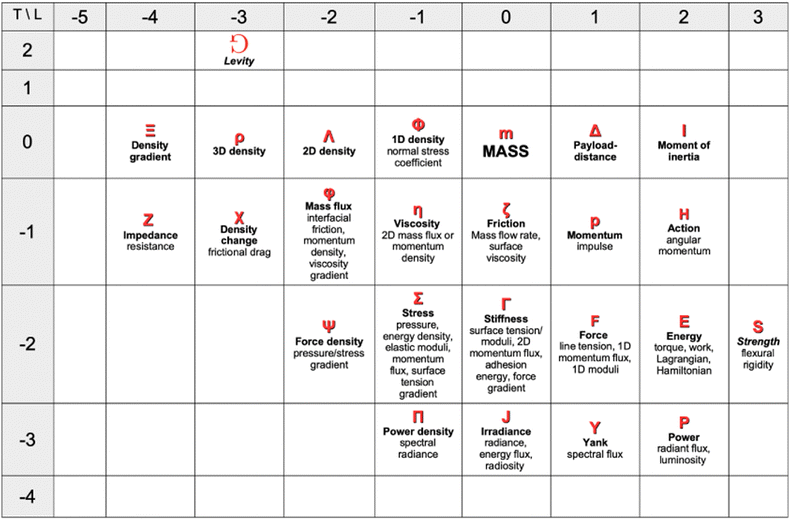
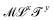 . Values of the exponents x and y refer to the columns and rows. A given quantity associated with a choice of couple (x, y) usually has several names. The symbol and the name in bold are the one we chose when generically referring to that quantity in this review. The dimensions of a mechanical quantity dictate its symbol. We systematically use the same symbols for all the mechanical quantities that share the same dimensions. For instance, we do not use “P” for pressure, “G” for elastic modulus and “σ” for shear stress, we just use Σ for all quantities with dimensions
. Values of the exponents x and y refer to the columns and rows. A given quantity associated with a choice of couple (x, y) usually has several names. The symbol and the name in bold are the one we chose when generically referring to that quantity in this review. The dimensions of a mechanical quantity dictate its symbol. We systematically use the same symbols for all the mechanical quantities that share the same dimensions. For instance, we do not use “P” for pressure, “G” for elastic modulus and “σ” for shear stress, we just use Σ for all quantities with dimensions 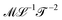 . We have found useful to define the levity and strength, which are respectively discussed in Sections 4.4.2 and 3.1.7. Please feel free to complete this table in any way you see fit
. We have found useful to define the levity and strength, which are respectively discussed in Sections 4.4.2 and 3.1.7. Please feel free to complete this table in any way you see fit
Pairs of mechanical quantities are the building blocks of the relationship between mechanics and kinematics, but they are only a starting point. If motion can be understood from the interplay of mechanical quantities, what can we expect from the interaction of three, four or even more quantities? We asked ourselves these very questions three years ago and we have been working on answering them ever since; our https://www.youtube.com/@naturesnumbers documents this journey. In this review, we solely focus on the interplay of pairs of mechanical quantities, but we will return later with more on the impact of additional players.
To illustrate the scope of a dimensional analysis of mechanics we will use examples from a wide spectrum of fields. This diversity constrains us to limit our citations to a few papers, which can be used as gates toward larger bodies of literature. Our background in fluid dynamics, soft matter, and biophysics, has biased us toward references from these fields. For instance, we are indebted to several reviews and textbooks on spreading, pinching and coalescence, including works by Dussan,9 de Gennes,10 Leger and Joanny,11 Middleman,12 Oron et al.,13 McKinley,14 Starov et al.,15 Kalliadasis and Thiele,16 Craster and Matar,17 Bonn et al.,18 Popescu et al.,19 de Gennes et al.,20 Snoeijer and Andreotti,21 Lu et al.,22 Bico et al.,23 Andreotti and Snoeijer,24 Lohse et al.,25 and Lohse and Zhang.26 However, we have tried as much as possible to diversify our references to include a literature more familiar to biologists and engineers. In particular, for explosions we relied on works by Bethe et al.,27 Glasstone et al.,28 Sedov,29 Krehl,30 Westine et al.,31 Kinney and Graham32 and Sachdev.33 For biological systems, we relied on works by Thompson,34 Mitchison and Cramer,35 Alt,36 Sheetz,37 Roberts et al.,38 Lecuit and Lenne,39 Le Clainche and Carlier,40 Pollard and Cooper,41 Phillips et al.,42 Marchetti et al.43 and Schwarz and Safran.44 We have also benefited from seminal texts on dimensional analysis, including those by Fourier,1 Maxwell,3 Buckingham,45 Rayleigh,46 Bridgman,47 Barenblatt,6,48 and Santiago.49
In this review, terms first appearing between ‘single quotes’ are technical terms from the literature. A search for this term on the Web will generally yield its definition. Terms appearing in italics are those we first define here, or which substantially deviate from traditional usage. Terms appearing between “double quotes” are actual quotes, or colloquialisms. The sign ‘≡’ symbolizes a definition, where the left-hand side is a shorthand notation for the right-hand side. The sign ‘≃’ means that the two sides of the equation are expected to be of the same ‘order of magnitude’ (other authors may use ∝ or ∼). The sign ‘∼’ will be used to state an incomplete scaling relation, as in d ∼ tα, where “incomplete” means that the left and right-hand sides do not have the same dimensions. The sign ‘=’ refers to a standard equality, which is presumably exact.
Links to the https://www.youtube.com/@naturesnumbers are given at the beginning of each associated section.
2 The mechanical quantities
https://youtu.be/XDQ21ngtpkI?si=a1PeLW6ICFV8bP5iMechanics includes geometry, kinematics and everything beyond, if it can be expressed with the inclusion of the dimension of mass. So the most generous definition of the term “mechanical quantity” could encompass any quantity with dimensions of the form 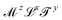 , where x, y and z could a priori be real numbers. However, this is not how we will use this term in this review. We will refer to quantities with dimensions of the form
, where x, y and z could a priori be real numbers. However, this is not how we will use this term in this review. We will refer to quantities with dimensions of the form 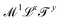 as mechanical quantities. We will use the term kinematic quantities to describe quantities with dimensions of the form
as mechanical quantities. We will use the term kinematic quantities to describe quantities with dimensions of the form 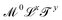 , with x ≠ 0 and y ≠ 0. And we will use the adjectives geometric/spatial and chronometric/temporal to describe
, with x ≠ 0 and y ≠ 0. And we will use the adjectives geometric/spatial and chronometric/temporal to describe 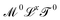 and
and 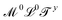 (x ≠ 0 and y ≠ 0), respectively. What about quantities like
(x ≠ 0 and y ≠ 0), respectively. What about quantities like 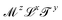 (z ≠ 0, z ≠ 1)? We will disregard them due to the fact that they can be reduced to mechanical quantities by factorization:
(z ≠ 0, z ≠ 1)? We will disregard them due to the fact that they can be reduced to mechanical quantities by factorization: 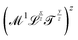 .
.
Examples of geometric quantities include not only the well-known length 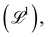 area
area 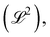 and volume
and volume 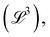 but also more technical quantities like the ‘wavenumber’
but also more technical quantities like the ‘wavenumber’ 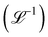 . Chronometric quantities include the duration or period
. Chronometric quantities include the duration or period 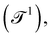 or the frequency
or the frequency 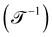 . The three most well-known examples of kinematic quantities are speed or velocity
. The three most well-known examples of kinematic quantities are speed or velocity 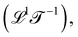 acceleration
acceleration 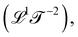 and diffusivity
and diffusivity 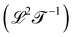 . Progressive time derivatives of the position beyond acceleration lead to the so-called ‘jerk’, ‘snap’, ‘crackle’ and ‘pop’, but these colorful terms are seldom used. More broadly, as we will see later, there are many more possible kinematic quantities, although they are less known and rarely have names.
. Progressive time derivatives of the position beyond acceleration lead to the so-called ‘jerk’, ‘snap’, ‘crackle’ and ‘pop’, but these colorful terms are seldom used. More broadly, as we will see later, there are many more possible kinematic quantities, although they are less known and rarely have names.
Now, what about mechanical quantities 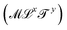 ? In a colloquial sense, mechanical quantities are the “forces” that in turn push or pull, drive or resist, impel or impair, all the processes at play behind space, time and their combination: motion. We have seen a few examples of these mechanical quantities in the introduction: mass (x = 0, y = 0), density (x = −3, y = 0), force (x = 1, y = −2), and energy (x = 2, y = −2). Each quantity is specified by its coordinates (x, y), so mechanical quantities can be represented as points on a plane. As we said, a priori, the coordinates x and y could take any value, but the small integers are of notable importance. Indeed the well-known geometric, kinematic and mechanical quantities have integer exponents. Investigating the reasons for this preference for integers is a fascinating task, but it goes beyond the scope of this review. In this review, we will only take note of this fact, and we will use it to our advantage. Because if the coordinates are small integers, we can represent the standard mechanical quantities they correspond to in a table. Thus, standard mechanical quantities refer to mechanical quantities where the exponents x and y are small integers, but since these are the only mechanical quantities we will be dealing with here, we will drop the adjective “standard”.
? In a colloquial sense, mechanical quantities are the “forces” that in turn push or pull, drive or resist, impel or impair, all the processes at play behind space, time and their combination: motion. We have seen a few examples of these mechanical quantities in the introduction: mass (x = 0, y = 0), density (x = −3, y = 0), force (x = 1, y = −2), and energy (x = 2, y = −2). Each quantity is specified by its coordinates (x, y), so mechanical quantities can be represented as points on a plane. As we said, a priori, the coordinates x and y could take any value, but the small integers are of notable importance. Indeed the well-known geometric, kinematic and mechanical quantities have integer exponents. Investigating the reasons for this preference for integers is a fascinating task, but it goes beyond the scope of this review. In this review, we will only take note of this fact, and we will use it to our advantage. Because if the coordinates are small integers, we can represent the standard mechanical quantities they correspond to in a table. Thus, standard mechanical quantities refer to mechanical quantities where the exponents x and y are small integers, but since these are the only mechanical quantities we will be dealing with here, we will drop the adjective “standard”.
In Table 1, we have tabulated the mechanical quantities we could find in the literature, highlighting the fact that they may bear different names depending on the context. Surprisingly, such a table does not seem to have been drawn before, although, as we will see, it provides a great way to understand the mechanical underpinning of space-time. The table is organized around mass (x = y = 0), with columns determined by the exponent x, and rows by the exponent y. You can think of each mechanical quantity as being located in the dimensional space with two coordinates (x, y). We will use the symbol Q(x,y) to designate the mechanical quantity with dimensions 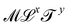 , and the symbol K(x, y) to designate the kinematic quantity with dimensions
, and the symbol K(x, y) to designate the kinematic quantity with dimensions 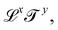 or simply Q and K when the exponents are implicit.
or simply Q and K when the exponents are implicit.
Table 1 is a map of the explorations of mechanics in the past centuries, but this mechanical universe is still mostly uncharted territory. The quantities on this table were discovered step by step. Just a few centuries ago, the table would have been mostly empty. Beyond mass, density, force and momentum, contemporaries of Newton had very little to work with. Newton himself formalized the concept of ‘viscosity’, while his rival Hooke was quantifying the concept of ‘stiffness’. It is the painstaking recording of natural phenomena that progressively enlarged the mechanical cartography. And this exploration is still ongoing. There are blank spots to fill. We took the liberty of naming two quantities we felt deserved their place, but for which we could not find names in the literature: levity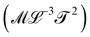 and strength
and strength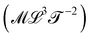 . A famous example of levity is the inverse of the gravitational constant G. Almost equally famous examples of strength are ℏc, and kce2, where ℏ, kc, c and e are respectively the Planck and Coulomb constants, the speed of light, and the elementary charge. These two expressions respectively represent the “strength” of the nuclear and electromagnetic interactions. We will return to these important examples later in the review.
. A famous example of levity is the inverse of the gravitational constant G. Almost equally famous examples of strength are ℏc, and kce2, where ℏ, kc, c and e are respectively the Planck and Coulomb constants, the speed of light, and the elementary charge. These two expressions respectively represent the “strength” of the nuclear and electromagnetic interactions. We will return to these important examples later in the review.
Each mechanical quantity can a priori be independent from the others. However, as we shall see, mechanical quantities are revealed through their interactions with one another. The most elementary form of such interaction is between pairs of mechanical quantities. Ratios of different mechanical quantities can produce space, time and motion.
Ratios of quantities in the same row produce purely spatial results:
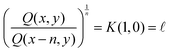 | (1) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) will generally be used to refer to any kind of length, size or distance, when this length is constant. We will rather use the symbol d when referring to a variable length. When multiple lengths are present, we may occasionally use alternate symbols for lengths, like h for height, or r for radius.
will generally be used to refer to any kind of length, size or distance, when this length is constant. We will rather use the symbol d when referring to a variable length. When multiple lengths are present, we may occasionally use alternate symbols for lengths, like h for height, or r for radius.
Ratios of quantities in the same column produce purely temporal results:
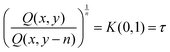 | (2) |
Ratios of quantities on a diagonal of slope −1 produce speeds:
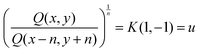 | (3) |
Ratios of quantities on a diagonal of slope −2 produce acceleration:
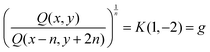 | (4) |
These ratios giving rise to length, time, speed, and acceleration are the most well-known, but we shall see that others are of interest. Note also that the relationship between two mechanical quantities is sometimes encoded in the very names of these quantities. In particular, the quantity Q(x − 2, y − 1) can be thought of as the flux of Q(x, y). For instance, stress can be thought of as a flux of momentum. The quantity Q(x − 3, y) can be thought of as the density of the quantity Q(x, y). For instance, stress can be thought of as a density of energy. However, except in a few instances where traditions obliged us (as with the mass flux, or the force–density), we tried to use names that did not explicitly refer to a parent quantity. All quantities on the table are related to another, but every quantity exists in its own right.
As illustrated by the different names of the mechanical quantities shown in Table 1, the overall spatial dimension of a particular context can lead to confusing usage of the same words. For instance, in a 2D setting, one may call the stiffness Q(0, −2) an elasticity (this is very common in the study of cells and tissues43,44). Our choice here will be to use the 3D naming conventions written in bold in Table 1. Note that the existence of many names for a quantity with the same dimensions is correlated to the existence of many units as well. For instance, Table 1 will make it quite obvious that stiffness is sometimes expressed in N m−1, where a Newton is a unit of force, or in J m−2, where a Joule is a unit of energy. Although less conventional, it could very well be given in poise m s−1, where a poise is a unit of viscosity. We have found that the table of mechanical quantities can make it easier to juggle with all these overlapping names and units.
3 The mechanics of space or time
Before addressing the relationship between mechanics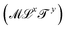 and kinematics
and kinematics 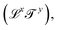 that is between mass-carrying quantities and motion, we should first discuss how space or time can be separately understood mechanically. Any constant length or duration can be decomposed into a pair of mechanical factors. At the very least, these factors are interpreted as providing a way to compute the values of lengths or durations, but they can also be regarded as the “origin”, or “reason”, or “cause” behind these lengths or durations.
that is between mass-carrying quantities and motion, we should first discuss how space or time can be separately understood mechanically. Any constant length or duration can be decomposed into a pair of mechanical factors. At the very least, these factors are interpreted as providing a way to compute the values of lengths or durations, but they can also be regarded as the “origin”, or “reason”, or “cause” behind these lengths or durations.
3.1 Simple lengths
https://youtu.be/QiZ--8tkMdo?si=2IERNoMuT8Jq1A6wAs mentioned in the introduction, Archimedes showed us the way when it comes to relating geometry and mechanics, when he expressed volume as the ratio of mass and density Ω = m/ρ. The story has been told a thousand times; it is the original “Eureka!” moment. https://math.nyu.edu/~crorres/Archimedes/Crown/bilancetta.html shows the importance it had on the mechanical Renaissance.50
If the mass and density of an object are known, then its volume is known. For a simple volume, like that of a cube, we can simply compute the volume from the knowledge of the length of the side ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , as Ω =
, as Ω = ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 3. Conversely, for a given volume, we can always compute the length of the side of a cube with the same volume, as
3. Conversely, for a given volume, we can always compute the length of the side of a cube with the same volume, as 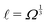 , which represents a sort of “average size” of the object. Using the notations introduced in the previous section (eqn (1)), we can write:
, which represents a sort of “average size” of the object. Using the notations introduced in the previous section (eqn (1)), we can write:
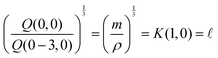 | (5) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) to denote any length, when no confusion is possible. Once we shall start dealing with multiple such lengths simultaneously, we will introduce more specific notations. In particular, the length built from the quantities Q1 and Q2 shall be called
to denote any length, when no confusion is possible. Once we shall start dealing with multiple such lengths simultaneously, we will introduce more specific notations. In particular, the length built from the quantities Q1 and Q2 shall be called ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) Q1Q2. So in the example from Archimedes, the average size is
Q1Q2. So in the example from Archimedes, the average size is ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) mρ, and the volume is
mρ, and the volume is ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) mρ3. Note that the order of the indices does not matter, so
mρ3. Note that the order of the indices does not matter, so ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) mρ =
mρ = ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ρm. We will come back to this important point in Section 4.4.
ρm. We will come back to this important point in Section 4.4.
Objects can have all sorts of shapes, with different heights, widths and lengths. When a measurement of the “size” of this object is performed, this measure may not exactly coincide with the size 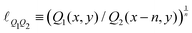 . For instance, if the object is spherical, its volume will be Ω = (4π/3)r3, where r is the radius, so
. For instance, if the object is spherical, its volume will be Ω = (4π/3)r3, where r is the radius, so 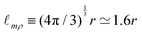 . If we call radius r the “size”, then this size is only approximately given by the ratio of mass and density,
. If we call radius r the “size”, then this size is only approximately given by the ratio of mass and density, 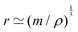 . Because tracking the fine effects of shape is often challenging, and because we do not seek precision but generality, we will often rely on this approximate equality sign ‘≃’.
. Because tracking the fine effects of shape is often challenging, and because we do not seek precision but generality, we will often rely on this approximate equality sign ‘≃’.
In all generality, lengths can be built by combining an arbitrary number of quantities (geometric, kinematic, mechanical and even beyond) such that the overall dimension is a length. However, in this review, we will focus on cases where the decomposition only involves two mechanical quantities. We will refer to these lengths as simple lengths, and as we will see they have been useful in a very wide range of situations. Even if we restrict ourselves to the standard mechanical quantities tabulated in Table 1, there are over sixty pairs that can produce lengths. We will only discuss a few, enough to illustrate the generality of this mechanical approach to space:
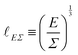
| (6) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΣ applies particularly in the context of explosions. In the introduction, we mentioned the scaling derived by Taylor for the dynamics of the radius of an explosion,
EΣ applies particularly in the context of explosions. In the introduction, we mentioned the scaling derived by Taylor for the dynamics of the radius of an explosion, 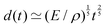 . We will return to this scaling later in the review; it concerns a type of motion connected to a ratio of energy and density. Evidently, this motion cannot continue indefinitely and eventually a ‘final blast radius’ is reached.28,31,32,51–54 This radius gives the extent of the zone where most damages occur. For the nuclear test studied by Taylor (Trinity), the energy is that of the bomb, around E ≃ 1014 J, and the stress is the bulk modulus of the air, which is not far from the atmospheric pressure Σ ≃ 105 Pa. Overall, this gives:
. We will return to this scaling later in the review; it concerns a type of motion connected to a ratio of energy and density. Evidently, this motion cannot continue indefinitely and eventually a ‘final blast radius’ is reached.28,31,32,51–54 This radius gives the extent of the zone where most damages occur. For the nuclear test studied by Taylor (Trinity), the energy is that of the bomb, around E ≃ 1014 J, and the stress is the bulk modulus of the air, which is not far from the atmospheric pressure Σ ≃ 105 Pa. Overall, this gives: 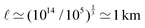 . As a comparison, bombs of 1 Mt, 10 Mt and 100 Mt of TNT (where 1 ton of TNT is equal to 4.184 gigajoules) correspond to blasts radii of 3 km, 8 km, and 16 km, respectively.28 The length
. As a comparison, bombs of 1 Mt, 10 Mt and 100 Mt of TNT (where 1 ton of TNT is equal to 4.184 gigajoules) correspond to blasts radii of 3 km, 8 km, and 16 km, respectively.28 The length ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΣ also applies to blast cavities of underground explosions, where Σ is the elastic modulus of the ground materials.55
EΣ also applies to blast cavities of underground explosions, where Σ is the elastic modulus of the ground materials.55
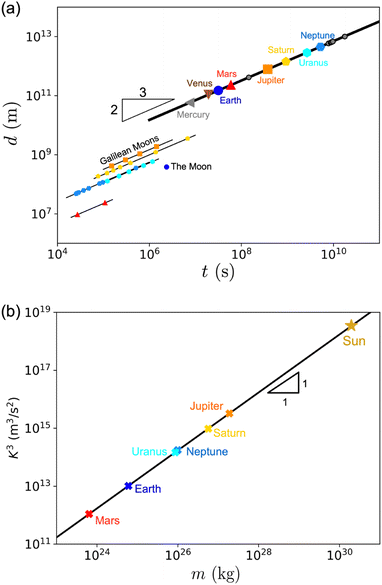
As illustrated in Fig. 1a, the same formula can be applied all the way up to supernovae explosions,56,57 which release energies on the order of 1044 J, and can extend their blast to a distance of at least 1019 m (i.e., over 300 parsecs). This gives a pressure of the interstellar medium of E/![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 3 ≃ 1044/1019×3 ≃ 10−13 Pa, which is the right order of magnitude.57 In regions of interstellar space with even smaller pressures, the supernova remnants can extend even further.
3 ≃ 1044/1019×3 ≃ 10−13 Pa, which is the right order of magnitude.57 In regions of interstellar space with even smaller pressures, the supernova remnants can extend even further.
 | ||
Fig. 1 Examples of simple lengths. (a) The Crab nebula, a supernova remnant, which will grow for millennia up to a size 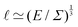 (here (here ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 1017 m). Image credit: NASA (public domain). (b) Earth, Titan and the Moon, three astronomical bodies with sizes of the form ≃ 1017 m). Image credit: NASA (public domain). (b) Earth, Titan and the Moon, three astronomical bodies with sizes of the form ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ Σ/Ψ (here ≃ Σ/Ψ (here ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 6371, 2575, 1737 km). Image credit: NASA (public domain). (c) Manilius crater on the Moon, an example of length given by ≃ 6371, 2575, 1737 km). Image credit: NASA (public domain). (c) Manilius crater on the Moon, an example of length given by 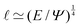 (here (here ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 19 km). Image credit: NASA (public domain). (d) Illustration of the capillary length on a pendant drop before its fall (public domain), with size ≃ 19 km). Image credit: NASA (public domain). (d) Illustration of the capillary length on a pendant drop before its fall (public domain), with size 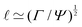 (here (here ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 3 mm). (e) An elastic substrate is deformed by capillarity at the contact line, over a distance ≃ 3 mm). (e) An elastic substrate is deformed by capillarity at the contact line, over a distance ![[small script l]](https://www.rsc.org/images/entities/i_char_e146.gif) ≃Γ/Σ (here ≃Γ/Σ (here ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 10 μm).23 (f) A thin film of oil on water produces an interference pattern testifying of its small thickness expressed as ≃ 10 μm).23 (f) A thin film of oil on water produces an interference pattern testifying of its small thickness expressed as 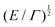 (public domain). (g) Beyond the optical resolution, the diffraction pattern of an atomic crystal obtained by electron crystallography reveals the typical size of the atoms (public domain). This size if of the form (public domain). (g) Beyond the optical resolution, the diffraction pattern of an atomic crystal obtained by electron crystallography reveals the typical size of the atoms (public domain). This size if of the form ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ S/E or ≃ S/E or ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ H/p (here ≃ H/p (here ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 0.1 nm). ≃ 0.1 nm). | ||
The length ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΣ can also be used in situations far from explosions. For instance, in microscopic physics influenced by thermal effects, E can be the thermal energy E = kBΘ, where kB is the Boltzmann constant and Θ is the temperature. This Boltzmann constant conveniently enables the translation of a temperature into an energy, incorporating thermodynamics into the realm of mechanics. In this context, the equation
EΣ can also be used in situations far from explosions. For instance, in microscopic physics influenced by thermal effects, E can be the thermal energy E = kBΘ, where kB is the Boltzmann constant and Θ is the temperature. This Boltzmann constant conveniently enables the translation of a temperature into an energy, incorporating thermodynamics into the realm of mechanics. In this context, the equation 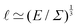 is better known as Σ
is better known as Σ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 3 ≃ kBΘ, which is called the ‘ideal gas law’, and is usually written as ‘PV = nkBT’, where V/n ≃
3 ≃ kBΘ, which is called the ‘ideal gas law’, and is usually written as ‘PV = nkBT’, where V/n ≃ ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 3 is the average volume associated with each microscopic constituent, n is the number of these constituents, and P, V and T are the symbols usually used for pressure, volume and temperature, respectively (note that we shall not use these notations in this review). This denomination is a bit misleading since this formula is not restricted to ideal gases but can be useful for connecting (thermal) energy, pressure, and microscales, for a wide variety of materials. For instance, in 1905, Einstein's theory of Brownian motion was developed with the postulate that the osmotic pressure due to a dilute suspension of particles was analogous to that of molecular solutes.58a In some cases, the stress Σ can then be interpreted as an elastic modulus. For instance, in polymer physics,
3 is the average volume associated with each microscopic constituent, n is the number of these constituents, and P, V and T are the symbols usually used for pressure, volume and temperature, respectively (note that we shall not use these notations in this review). This denomination is a bit misleading since this formula is not restricted to ideal gases but can be useful for connecting (thermal) energy, pressure, and microscales, for a wide variety of materials. For instance, in 1905, Einstein's theory of Brownian motion was developed with the postulate that the osmotic pressure due to a dilute suspension of particles was analogous to that of molecular solutes.58a In some cases, the stress Σ can then be interpreted as an elastic modulus. For instance, in polymer physics, ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΣ gives the typical ‘blob size’. Assuming room temperature (E ≃ 4 × 10−21 J) and a modulus around Σ ≃ 100 Pa, which is typical for soft gels, then
EΣ gives the typical ‘blob size’. Assuming room temperature (E ≃ 4 × 10−21 J) and a modulus around Σ ≃ 100 Pa, which is typical for soft gels, then 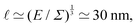 a scale characteristic of the biological frontier of physics.59 Clearly, the same formula can underpin very different interpretations. Similar formulas have also been used to explain the size of cells, where the thermal energy is multiplied with the effective number of proteins in the cell.60–62
a scale characteristic of the biological frontier of physics.59 Clearly, the same formula can underpin very different interpretations. Similar formulas have also been used to explain the size of cells, where the thermal energy is multiplied with the effective number of proteins in the cell.60–62
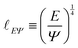
| (7) |
The length ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΨ can also be used to describe the average ‘height of a Brownian particle’ in sedimentation or centrifugation. There, E is the thermal energy and the force–density is Ψ ≃ ρg, with g = g0 for sedimentation and g = rω2 for centrifugation (where r is the distance from the axis of rotation at rate ω).69 For instance, at room temperature E ≃ 4 × 10−21 J, and if ρ ≃ 103 kg m−3, then
EΨ can also be used to describe the average ‘height of a Brownian particle’ in sedimentation or centrifugation. There, E is the thermal energy and the force–density is Ψ ≃ ρg, with g = g0 for sedimentation and g = rω2 for centrifugation (where r is the distance from the axis of rotation at rate ω).69 For instance, at room temperature E ≃ 4 × 10−21 J, and if ρ ≃ 103 kg m−3, then 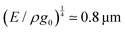 . Particles below this size are ‘Brownian’, and they remain suspended or dispersed, with a number distribution at any height set by the sedimentation–diffusion equilibrium.70
. Particles below this size are ‘Brownian’, and they remain suspended or dispersed, with a number distribution at any height set by the sedimentation–diffusion equilibrium.70
Note that for objects embedded in a fluid, the force–density will generally be built from ‘buoyancy’, i.e., from the difference in density with the surrounding medium Ψ ≃ |ρf − ρ|g. In particular, in the case of Brownian particles, the density difference between dispersed particles and the outside medium determines the length scale that can be identified as the upper limit to the size of Brownian particles. The argument explains why metal nanoparticles are Brownian only below 100 nm, whereas polymer microbeads can be over a micron. Centrifugation can provide a much larger value of effective g, and therefore leads to sedimentation and separation.69
An example of a characteristic size of the form ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΨ for which the force–density is not the weight density occurs for drop impact. When the viscosity of the drop is negligible compared to its inertia and surface-tension, some studies have found that the maximum drop radius after impact is given by
EΨ for which the force–density is not the weight density occurs for drop impact. When the viscosity of the drop is negligible compared to its inertia and surface-tension, some studies have found that the maximum drop radius after impact is given by 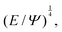 where E ≃ ρr3u2 is the kinetic energy of the impacting drop of radius r, speed u and density ρ. The force–density Ψ ≃ Γ/r2 originates from capillarity.71 We will see in Section 3.1.6 that other studies suggest a different scaling.
where E ≃ ρr3u2 is the kinetic energy of the impacting drop of radius r, speed u and density ρ. The force–density Ψ ≃ Γ/r2 originates from capillarity.71 We will see in Section 3.1.6 that other studies suggest a different scaling.
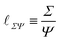
| (8) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ Σ/ρg0. The simplest examples consider that the stress is the isotropic pressure. Such a formula is particularly useful in the context of the formation of astronomical objects like planets or stars.72 In these cases, the size of a ball of matter is understood as a compromise between the compression by gravity and the resistance of an internal pressure. The size is then that of the planet or star. In this context, eqn (8) is sometimes referred to as ‘the hydrostatic equilibrium’ and written as Σ ≃
≃ Σ/ρg0. The simplest examples consider that the stress is the isotropic pressure. Such a formula is particularly useful in the context of the formation of astronomical objects like planets or stars.72 In these cases, the size of a ball of matter is understood as a compromise between the compression by gravity and the resistance of an internal pressure. The size is then that of the planet or star. In this context, eqn (8) is sometimes referred to as ‘the hydrostatic equilibrium’ and written as Σ ≃ ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) Ψ, where the internal pressure equilibrates the weight per unit volume,73 as in the context of barometers where pressure was first defined.74 As an example, in the case of Earth, ρg0 ≃ 5 × 104 N m−3, and
Ψ, where the internal pressure equilibrates the weight per unit volume,73 as in the context of barometers where pressure was first defined.74 As an example, in the case of Earth, ρg0 ≃ 5 × 104 N m−3, and ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 6 × 106 m, giving Σ ≃ 1011 Pa. Such pressure typically corresponds to the elastic modulus of metallic or amorphous solids constituting the Earth (≃100 GPa). Through this scaling, the various sizes of astronomical objects shown in Fig. 1b are directly related to their densities and elastic moduli.72 For more information, we refer the reader to a pedagogical presentation on scaling approaches to the size of stars and planets, which some of us have recently published.75
≃ 6 × 106 m, giving Σ ≃ 1011 Pa. Such pressure typically corresponds to the elastic modulus of metallic or amorphous solids constituting the Earth (≃100 GPa). Through this scaling, the various sizes of astronomical objects shown in Fig. 1b are directly related to their densities and elastic moduli.72 For more information, we refer the reader to a pedagogical presentation on scaling approaches to the size of stars and planets, which some of us have recently published.75
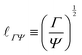
| (9) |
When a drop hangs from a leaf as shown in Fig. 1d, it may grow in size only up to the capillary length, after which it will fall. Generally, gravitation flattens drops of size larger than ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ΓΨ, forming puddles, while capillarity keeps smaller drops spherical. The capillary length also influences the shape of a meniscus near an immersed or floating object.20 Understanding the interplay between gravity and capillarity can actually be used to determine surface tension, using pendant drop analysis or using capillary rise.20
ΓΨ, forming puddles, while capillarity keeps smaller drops spherical. The capillary length also influences the shape of a meniscus near an immersed or floating object.20 Understanding the interplay between gravity and capillarity can actually be used to determine surface tension, using pendant drop analysis or using capillary rise.20
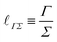
| (10) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ΓΣ may be called the ‘elasto-adhesive’ length.76
ΓΣ may be called the ‘elasto-adhesive’ length.76
When Γ comes from surface-tension, the length ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ΓΣ is often called the ‘elasto-capillary length’.23 This length is relevant for the spreading of drops on soft substrates, associating surface energy and elasticity.24 The elasticity can be that of the spreading object or of its environment. For instance, as illustrated in Fig. 1e,
ΓΣ is often called the ‘elasto-capillary length’.23 This length is relevant for the spreading of drops on soft substrates, associating surface energy and elasticity.24 The elasticity can be that of the spreading object or of its environment. For instance, as illustrated in Fig. 1e, ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ΓΣ applies to the height of the wetting ridge near the solid–liquid–air triple line,77 where the surface tension Γ acts perpendicularly to a substrate of elastic modulus Σ. Experimentally, a drop of glycerol (Γ ≃ 63 mN m−1) on a soft silicone gel (Σ ≃ 2.4 kPa) produces a ridge of about 12 μm.78 Other orders of magnitude can be obtained, for instance, a length of 30 nm was found for tricresyl phosphate (Γ ≃ 28.5 mN m−1) on a silicone elastomer (Σ ≃ 0.6 MPa).79
ΓΣ applies to the height of the wetting ridge near the solid–liquid–air triple line,77 where the surface tension Γ acts perpendicularly to a substrate of elastic modulus Σ. Experimentally, a drop of glycerol (Γ ≃ 63 mN m−1) on a soft silicone gel (Σ ≃ 2.4 kPa) produces a ridge of about 12 μm.78 Other orders of magnitude can be obtained, for instance, a length of 30 nm was found for tricresyl phosphate (Γ ≃ 28.5 mN m−1) on a silicone elastomer (Σ ≃ 0.6 MPa).79
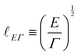
| (11) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 3, where
3, where ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) is the film thickness. Disjoining pressure and the Hamaker constant play an important role in the climbing and spreading of thin films,11,19,83 and in setting the nano-topography of foam films.84
is the film thickness. Disjoining pressure and the Hamaker constant play an important role in the climbing and spreading of thin films,11,19,83 and in setting the nano-topography of foam films.84
Note that the linear stability analysis of both freestanding and supported ultra-thin films results in a prediction of a spinodal-like instability into thick-thin regions, with a typical size ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ΓΨ given in eqn (9), where the force–density is defined as the gradient of disjoining pressure, i.e. Ψ ≃ E/
ΓΨ given in eqn (9), where the force–density is defined as the gradient of disjoining pressure, i.e. Ψ ≃ E/![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΓ4. This length scale has been observed experimentally in spinodal dewetting and spinodal stratification.16,85
EΓ4. This length scale has been observed experimentally in spinodal dewetting and spinodal stratification.16,85
As mentioned in Section 3.1.2, in the context of drop impact, when the viscosity of the drop is negligible, some studies have found that the maximum drop radius after impact is a simple length of the form ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΨ, where E is the kinetic energy of the drop. In contrast, other studies suggest that the maximum radius may be of the form given by
EΨ, where E is the kinetic energy of the drop. In contrast, other studies suggest that the maximum radius may be of the form given by ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) EΓ.86–88
EΓ.86–88
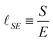
| (12) |
The length ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) SE can then be used to express the size of an atom, using S0 and the Hartree energy E = meu2, where me is the mass of the electron and where u = α0c is the semi-classical speed of the electron (α0 ≃ 1/137 is the ‘fine structure constant’ we shall discuss later, c is the speed of light). Under these assumptions eqn (12) gives
SE can then be used to express the size of an atom, using S0 and the Hartree energy E = meu2, where me is the mass of the electron and where u = α0c is the semi-classical speed of the electron (α0 ≃ 1/137 is the ‘fine structure constant’ we shall discuss later, c is the speed of light). Under these assumptions eqn (12) gives ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) = e2/(4πε0meu2), which is the first historical expression of the ‘Bohr radius’91 (an alternate way of writing this radius will be given in the next sub-section).
= e2/(4πε0meu2), which is the first historical expression of the ‘Bohr radius’91 (an alternate way of writing this radius will be given in the next sub-section).
In plasma and electrolytes, the strength S0 also appears in the definition of the Bjerrum and Debye lengths.82 The ‘Bjerrum length’ follows eqn (12) with S = S0/εr, which takes into account the dimensionless relative dielectric constant εr, and E = kBΘ. This length provides the scale at which the electrostatic and thermal effects exert comparable influence. It arises in the context of electrolytes and colloidal dispersions.82,92 For water at room temperature, εr ≃ 80 and the Bjerrum length is around 0.7 nm. The different assumptions leading from S/E to the Bohr or Bjerrum lengths are summarized here:
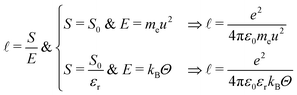 | (13) |
In the context of plasmas and electrolytes, the ‘Debye length’ also involves a strength S0. The Debye length characterizes the screening of electrostatic interactions between two charges in the presence of other charges. In colloidal dispersions, the Debye length depends on ion concentration, making it a property of the solution, unlike the Bjerrum length which depends on the solvent and its dielectric constant.92 In this context one can define an effective stiffness as a density of strength, Γ = S0/r3, which is then combined with the thermal energy using eqn (11). Here, the distance r is the mean distance between electrons and 1/r3 is the electron number density, so Γ can be understood as a charge density expressed in units of mass, length and time. The Debye length can vary widely, from the atomic scale in the solar core to thousands of kilometers in the intergalactic medium. In electrolyte media, encountered in soft matter and within cells, the strength is built from the number density of ions, whereas in semiconductors, the number density of dopants makes the relevant contribution.93,94
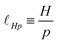
| (14) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ S0/E. The kinetic energy can be written in terms of the mass me of the electron and its speed u = α0c, so
≃ S0/E. The kinetic energy can be written in terms of the mass me of the electron and its speed u = α0c, so ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ S0/(meuα0c) ≃ S0/(pα0c), where p = meu is the momentum of the electron. Historically, the dimensionless ‘fine structure constant’ α0 was understood precisely in this fashion, as the ratio between the speed of the electron and the speed of light.95 However, α0 quickly showed up in other situations, particularly in the comparison between the electromagnetic strength S0 and the nuclear strength ℏc, since α0 = S0/ℏc. Using this formula we can rewrite the Bohr radius as
≃ S0/(meuα0c) ≃ S0/(pα0c), where p = meu is the momentum of the electron. Historically, the dimensionless ‘fine structure constant’ α0 was understood precisely in this fashion, as the ratio between the speed of the electron and the speed of light.95 However, α0 quickly showed up in other situations, particularly in the comparison between the electromagnetic strength S0 and the nuclear strength ℏc, since α0 = S0/ℏc. Using this formula we can rewrite the Bohr radius as ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ ℏ/p. Expressed in this way the Bohr radius is understood as the ‘de Broglie wavelength of the electron’. Generally, when H is the quantum of action ℏ, eqn (14) encompasses one of the central concept of quantum mechanics, the relationship between waves and particles.96
≃ ℏ/p. Expressed in this way the Bohr radius is understood as the ‘de Broglie wavelength of the electron’. Generally, when H is the quantum of action ℏ, eqn (14) encompasses one of the central concept of quantum mechanics, the relationship between waves and particles.96
We have seen that the Bohr radius can be expressed by two different pairs of mechanical quantities, S and E or H and p. The existence of multiple mechanical decompositions is not at all unique to this case. Any length can always be decomposed into a ratio of two mechanical quantities, but this decomposition is not unique, and this plurality encourages a diversity of mechanical models. The pair chosen in a particular situation depends on the broader context in which the length is found, and on historical circumstances. A more complete investigation of this “plurality” would require more than two mechanical quantities and is therefore out of the scope of this review. We will say a few more words about this in the conclusion.
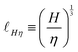
| (15) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 1 Å. So if we multiply the viscosity with the typical volume of a water molecule we get an action: H ≃ η
≃ 1 Å. So if we multiply the viscosity with the typical volume of a water molecule we get an action: H ≃ η![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 3 ≃ 10−33 J s. This value is not far from the quantum of action, ℏ ≃ 10−34 J s, so the viscosity of water almost corresponds to one quantum of action per molecule. Although this link between viscosity and action is here only sketched by rough orders of magnitude, it can be formalized more rigorously, as exemplified in a recent paper by Trachenko and Brazhkin.97
3 ≃ 10−33 J s. This value is not far from the quantum of action, ℏ ≃ 10−34 J s, so the viscosity of water almost corresponds to one quantum of action per molecule. Although this link between viscosity and action is here only sketched by rough orders of magnitude, it can be formalized more rigorously, as exemplified in a recent paper by Trachenko and Brazhkin.97
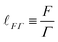
| (16) |
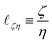
| (17) |
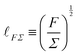
| (18) |
In eqn (16), Γ can be interpreted as the stiffness of a material behaving as a spring, then eqn (16) is just Hooke's law, F = Γ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) FΓ, where F and
FΓ, where F and ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) FΓ are usually understood as variables. In the context of spreading drops or cells, this ratio can represent a balance between a driving force F and surface tension or stiffness Γ. For cell spreading, the length
FΓ are usually understood as variables. In the context of spreading drops or cells, this ratio can represent a balance between a driving force F and surface tension or stiffness Γ. For cell spreading, the length ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) FΓ can be used to characterize the portion of the cell behind the edge, which is rich in a very dynamic polymer called ‘actin’.35,38,41 The polymerization of actin can be associated with a ‘protrusion force’ F, which is balanced by a surface energy Γ, with contributions form the plasma membrane, the cell stiffness, and the adhesion with the substrate.98,99
FΓ can be used to characterize the portion of the cell behind the edge, which is rich in a very dynamic polymer called ‘actin’.35,38,41 The polymerization of actin can be associated with a ‘protrusion force’ F, which is balanced by a surface energy Γ, with contributions form the plasma membrane, the cell stiffness, and the adhesion with the substrate.98,99
In eqn (17), the length is ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ζη ≡ ζ/η, which is a ratio between ‘friction’ or ‘mobility’ and viscosity. This equation is more often seen in the form ζ = η
ζη ≡ ζ/η, which is a ratio between ‘friction’ or ‘mobility’ and viscosity. This equation is more often seen in the form ζ = η![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ζη, in the context of Stokes drag,100,101 where it gives the effective friction ζ on an object of size
ζη, in the context of Stokes drag,100,101 where it gives the effective friction ζ on an object of size ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ζη moving slowly in a fluid of viscosity η. Indeed, at high viscosity and low speed, the frictional force is proportional to speed u, and given by F ≃ ζu ≃ η
ζη moving slowly in a fluid of viscosity η. Indeed, at high viscosity and low speed, the frictional force is proportional to speed u, and given by F ≃ ζu ≃ η![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ζηu. This connection is the basis for Brownian motion in the Stokes Einstein relation,101 and thus lies at the heart of colloidal physics and chemistry.102
ζηu. This connection is the basis for Brownian motion in the Stokes Einstein relation,101 and thus lies at the heart of colloidal physics and chemistry.102
In eqn (18), length is defined as the ratio between a force and a stress. This formula is more often seen as Σ ≃ F/![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 2, which defines stress from the force F over the area
2, which defines stress from the force F over the area ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 2. From this perspective, the stress is typically understood as intensive, whereas the force is extensive but normalized by the area. When F and Σ are independent constants,
2. From this perspective, the stress is typically understood as intensive, whereas the force is extensive but normalized by the area. When F and Σ are independent constants, ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) FΣ is a simple length in its own right. This is, for instance, the case in the physics of nematic and polar materials, which includes a large class of living systems.43,44 In this context,
FΣ is a simple length in its own right. This is, for instance, the case in the physics of nematic and polar materials, which includes a large class of living systems.43,44 In this context, ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) FΣ is sometimes called the ‘nematic length’, where F is understood as the ‘Frank constant’, which represents 1D elasticity associated with differences in alignment, and where Σ represents the energy per unit volume associated with the alignment of the nematic components.103 This length scale determines the typical extent of orientational boundary layers.43 Another important simple length in the study of active matter is the crossover from ‘wet’ to ‘dry’ active particles, which can be written as
FΣ is sometimes called the ‘nematic length’, where F is understood as the ‘Frank constant’, which represents 1D elasticity associated with differences in alignment, and where Σ represents the energy per unit volume associated with the alignment of the nematic components.103 This length scale determines the typical extent of orientational boundary layers.43 Another important simple length in the study of active matter is the crossover from ‘wet’ to ‘dry’ active particles, which can be written as ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ηχ, where η is the viscosity of the embedding fluid, and χ is understood as a so-called ‘frictional drag’.43
ηχ, where η is the viscosity of the embedding fluid, and χ is understood as a so-called ‘frictional drag’.43
3.2 Simple times
https://youtu.be/KNeNx9mi2ao?si=CSSXh1kCe7_DcNKvWe have seen that ratios of mechanical quantities can produce length scales that show up in a wide variety of situations. In these examples, a length emerges from a sort of “balance” between conflicting “forces”, where the term “force” is used quite generously to encompass any mechanical quantity  . Similarly, pairs of mechanical quantities can be used to understand time, durations and periods, leading to what we can call simple times. We will use the symbol τ when no ambiguity is possible, and τQ1Q2 when specificity is required. Over thirty such simple times can be derived from the standard quantities of Table 1. We list here the ones we shall discuss in this section:
. Similarly, pairs of mechanical quantities can be used to understand time, durations and periods, leading to what we can call simple times. We will use the symbol τ when no ambiguity is possible, and τQ1Q2 when specificity is required. Over thirty such simple times can be derived from the standard quantities of Table 1. We list here the ones we shall discuss in this section:
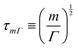
| (19) |
In the context of the dynamics of droplets, the mass is usually given by m ≃ ρr3, where ρ is the density of the fluid and r is the radius of the droplet. In this context one speaks of the ‘Rayleigh time’,104 which applies to the oscillation frequency of drops, as well as to the contact time of rebounding drops.105 Despite very different rebound profiles depending on the impact speed, the contact time remains the same and is set by τmΓ. The timescale also appears in capillarity-driven flows of ‘inviscid fluids’ (i.e., negligible viscosity).12,14,104,106,107
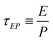
| (20) |
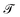 . Common units of energy, like kilowatt-hour, reflect this proximity, with 1 kW h = 3.6 × 106 J s−1 s, so simply 3.6 × 106 J. For a given energy E, the time scale in eqn (20) gives the expected time range when the energy consumption rate is the power P. This time scale can be used for a wide variety of purposes, to estimate how long you can drive on a full tank, as well as the life expectancy of the Sun.
. Common units of energy, like kilowatt-hour, reflect this proximity, with 1 kW h = 3.6 × 106 J s−1 s, so simply 3.6 × 106 J. For a given energy E, the time scale in eqn (20) gives the expected time range when the energy consumption rate is the power P. This time scale can be used for a wide variety of purposes, to estimate how long you can drive on a full tank, as well as the life expectancy of the Sun.
A typical small car will have something like 70 horsepower, so P ≃ 53 × 103 W. The energy comes from the fuel. Assuming a gas tank of 35 liters of standard fuel, with 8.9 kW h liter−1, yields E ≃ 109 J per gas tank. Then, τEP ≃ 5 hours. This is roughly how long this car can drive without refueling.
The principle behind the formula in eqn (20) remains the same for all kinds of fuel and all kinds of systems consuming this fuel. In particular, this formula can also be used to obtain an estimate of the lifetime of a star like the Sun. In this case, the power is well estimated by the solar luminosity, and P ≃ 3.8 × 1026 W.73 If the fuel of the Sun were standard gasoline as in the car, then the lifetime of the Sun would only be around 3000 years, according to eqn (20). This is obviously not the case.
So what is the fuel of the Sun? The quest to answer this question spanned from the mid 19th to the mid 20th century and involved some of the greatest minds of that time. The story has been told beautifully in a paper by Shaviv.108 An important step in the quest was to consider the energy to be due to the self-gravitation of the Sun, so E ≃ Gm2/![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , where m and
, where m and ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) are respectively the mass and size of the Sun. In this scenario, the power of the Sun, that is its luminosity is due to the gravitational potential energy. This time scale is sometimes called the ‘thermal timescale’, or the ‘Kelvin–Helmholtz timescale’, to honor Kelvin and Helmholtz contributions to this field of research. However, as noted by Shaviv,108 August Ritter was the first to derive this formula. This timescale plays an important role in astronomy, in particular to set the timescale of the collapse of protostars; however, it fails to estimate the age of the Sun and similar stars. Indeed, using
are respectively the mass and size of the Sun. In this scenario, the power of the Sun, that is its luminosity is due to the gravitational potential energy. This time scale is sometimes called the ‘thermal timescale’, or the ‘Kelvin–Helmholtz timescale’, to honor Kelvin and Helmholtz contributions to this field of research. However, as noted by Shaviv,108 August Ritter was the first to derive this formula. This timescale plays an important role in astronomy, in particular to set the timescale of the collapse of protostars; however, it fails to estimate the age of the Sun and similar stars. Indeed, using ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 7 × 108 m and m ≃ 2 × 1030 kg, we get τEP ≃ 30 million years. The inadequacy of this figure with the geological records led to intense debate, and the controversy was only resolved at the beginning of the 20th century, when it was realized that the fuel of the Sun is nuclear.108 By considering the conversion of hydrogen into helium, it was estimated that the energy of the Sun is around E ≃ 6 × 10−4mc2, giving τEP ≃ 10 billion years, which is the currently accepted order of magnitude, and is sometimes called the ‘nuclear time scale’.73
≃ 7 × 108 m and m ≃ 2 × 1030 kg, we get τEP ≃ 30 million years. The inadequacy of this figure with the geological records led to intense debate, and the controversy was only resolved at the beginning of the 20th century, when it was realized that the fuel of the Sun is nuclear.108 By considering the conversion of hydrogen into helium, it was estimated that the energy of the Sun is around E ≃ 6 × 10−4mc2, giving τEP ≃ 10 billion years, which is the currently accepted order of magnitude, and is sometimes called the ‘nuclear time scale’.73
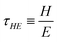
| (21) |
In the special case where the energy is the thermal energy (E = kBΘ), the time τ = ℏ/kBΘ is called the ‘Debye time’.109 The inverse of this time scale provides an important thermal cut-off in the propagation of waves in crystal lattices. At room temperature, the Debye time is around twenty femtoseconds. Note that the term ‘Debye time’ can also be used in a slightly different way, as the time for charge relaxation, for diffusion across the Debye length.110 The two formulas could be reconciled by using the relationship between viscosity and action, as given in eqn (15).
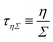
| (22) |
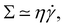 where
where ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) is the deformation rate. The time scale is then
is the deformation rate. The time scale is then ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) −1. In general, the deformation rate is not a constant. However, in complex fluids there are often remarkable values of
−1. In general, the deformation rate is not a constant. However, in complex fluids there are often remarkable values of ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) . For instance, many materials display rather elastic properties on short time scales, and are viscous on longer time scales.111 These materials are usually called ‘visco-elastic’, and the threshold between short and long time scales is the ‘relaxation time’ τ. In ‘Maxwell's model’, which is the simplest model of visco-elastic fluid, the elasticity of the material Σ, the viscosity, and the relaxation time are connected by the equation τ ≃ η/Σ.111,112 The greater the viscosity, the longer the time, and the greater the elasticity, the shorter the time. The relaxation time scale of visco-elastic fluids can range from milliseconds to decades.111,112 At any rate, in simple visco-elastic fluids the time τ is a constant of the material and it can be used to understand the transition between different flow regimes.14,111,113
. For instance, many materials display rather elastic properties on short time scales, and are viscous on longer time scales.111 These materials are usually called ‘visco-elastic’, and the threshold between short and long time scales is the ‘relaxation time’ τ. In ‘Maxwell's model’, which is the simplest model of visco-elastic fluid, the elasticity of the material Σ, the viscosity, and the relaxation time are connected by the equation τ ≃ η/Σ.111,112 The greater the viscosity, the longer the time, and the greater the elasticity, the shorter the time. The relaxation time scale of visco-elastic fluids can range from milliseconds to decades.111,112 At any rate, in simple visco-elastic fluids the time τ is a constant of the material and it can be used to understand the transition between different flow regimes.14,111,113
In more complex visco-elastic fluids beyond Maxwell's model, there can be more than one relaxation time.111,114 Polymer solutions typically exhibit a spectrum of relaxation times. In addition, some materials may behave as Maxwell fluids under small deformations, but display flow-induced changes in their structure at higher deformations. For instance, worm-like micelle111 solutions have a viscosity η1 at low deformation rates, and above a threshold ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) 1 a different flow-induced “phase” of viscosity η2 is generated and coexists at constant stress Σp with the original one until a second threshold
1 a different flow-induced “phase” of viscosity η2 is generated and coexists at constant stress Σp with the original one until a second threshold ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) 2. Above this threshold the whole material has viscosity η2. This phenomenon is usually called ‘shear-banding’.115 Both
2. Above this threshold the whole material has viscosity η2. This phenomenon is usually called ‘shear-banding’.115 Both ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) 1−1 ≃ η1/Σp and
1−1 ≃ η1/Σp and ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) 2−1 ≃ η2/Σp are time scales of the form η/Σ. More broadly, remarkable values of viscosity or stress can occur in a large class of ‘complex’ or ‘non-Newtonian’ fluids, including shear-thinning, shear-thickening and yield-stress fluids. In turn, these quantities provide multiple ways to construct time scales of the form given in eqn (22).111
2−1 ≃ η2/Σp are time scales of the form η/Σ. More broadly, remarkable values of viscosity or stress can occur in a large class of ‘complex’ or ‘non-Newtonian’ fluids, including shear-thinning, shear-thickening and yield-stress fluids. In turn, these quantities provide multiple ways to construct time scales of the form given in eqn (22).111
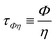
| (23) |
![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) , where
, where ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) ≃ u/
≃ u/![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) is a velocity gradient over the distance
is a velocity gradient over the distance ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) . In contrast, normal stresses arise from inertia and are usually of the form Σn ≃ ρu2 ≃ ρ
. In contrast, normal stresses arise from inertia and are usually of the form Σn ≃ ρu2 ≃ ρ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 2
2![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) 2. This stress is sometimes called the ‘dynamic pressure’, or the ‘Ram pressure’ in astrophysics.116 For Newtonian or non-Newtonian fluids, normal stresses can be expressed as Σn ≃ Φ
2. This stress is sometimes called the ‘dynamic pressure’, or the ‘Ram pressure’ in astrophysics.116 For Newtonian or non-Newtonian fluids, normal stresses can be expressed as Σn ≃ Φ![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) 2, such that
2, such that 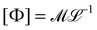 , the same dimensions as a 1D mass-density. In the Newtonian case, Φ ≃ ρ
, the same dimensions as a 1D mass-density. In the Newtonian case, Φ ≃ ρ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 2, but in non-Newtonian fluids, including magnetic fluids relevant to astrophysics, the normal stress coefficient Φ can be completely independent of ρ and inertia in general.117 Whereas the positive value of Φ for Newtonian fluids tend to generate centrifugal forces pushing a rotating fluid outward, for non-Newtonian fluids, Φ can be negative and push the material inward in the so-called ‘rod-climbing’ or ‘Weissenberg effect’.111,112 This is but one among many examples of non-Newtonian normal stress effects.
2, but in non-Newtonian fluids, including magnetic fluids relevant to astrophysics, the normal stress coefficient Φ can be completely independent of ρ and inertia in general.117 Whereas the positive value of Φ for Newtonian fluids tend to generate centrifugal forces pushing a rotating fluid outward, for non-Newtonian fluids, Φ can be negative and push the material inward in the so-called ‘rod-climbing’ or ‘Weissenberg effect’.111,112 This is but one among many examples of non-Newtonian normal stress effects.
For non-Newtonian fluids, the normal stress coefficient Φ is a material property as important as viscosity, and disconnected from the density. It is an independent mechanical quantity, from which a time scale τΦη can be constructed, as in eqn (23). In simple visco-elastic models like Maxwell's model, this time scale is identical to the relaxation time τηΣ. Indeed, in Maxwell's model, one has Φ ≃ ητηΣ.111 This identity is not true in general. Currently, the differences between the two non-Newtonian time scales are most often investigated in the context of flows with an extensional component, where the dual effects of normal stresses and relaxation time are factored into the differences between shear and extensional rheology.14,118
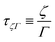
| (24) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , where Σ is the ‘traction stress’ exerted by the cells on the substrate, and the width
, where Σ is the ‘traction stress’ exerted by the cells on the substrate, and the width ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) near the edge is given by the nematic length discussed with eqn (18).
near the edge is given by the nematic length discussed with eqn (18).
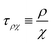
| (25) |
 or
or  in 2D). If the mass is constant, then τρχ ≡ ρ/χ ≃ n/ṅ, where ṅ ≃ ∂n/∂t. Thus, the time scale reflects the rate of change of the number of objects. For instance, in tissues of cells the time scale τρχ is associated with the characteristic time separating two cell divisions. This time scale is relevant for the spreading of tissues,122 as well as for some organisms like ants.123
in 2D). If the mass is constant, then τρχ ≡ ρ/χ ≃ n/ṅ, where ṅ ≃ ∂n/∂t. Thus, the time scale reflects the rate of change of the number of objects. For instance, in tissues of cells the time scale τρχ is associated with the characteristic time separating two cell divisions. This time scale is relevant for the spreading of tissues,122 as well as for some organisms like ants.123
4 The mechanics of motion
In the previous section, we investigated the mechanics of space or time, taken separately. Here, we will see how mechanical quantities can be used to rationalize motion, so we will address the connection between mechanics and kinematics, that is between mass-carrying quantities and space-time.We will present a few instructive examples of dynamic scalings from the literature, in particular those illustrated in Fig. 2.
 | ||
Fig. 2 Examples of dynamics associated with scalings of the form d ∼ tα, from the pinching of viscous liquid threads, to atomic explosions and the motions of living cells. (a) Side view of the pinching dynamics of a viscous glycerol filament,124 governed by d ≃ (Γ/η)t (eqn (31)), where the time t is the duration before pinch-off, so in this example the “actual time” runs from right to left. The length d is the radius of the filament, and Γ and η are the surface tension and viscosity of the fluid, respectively. (b) Side view of the first atomic explosion (Trinity test, 1945), discussed in the introduction.7 The dynamics of the blast radius d follow 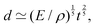 where E is the yield of the explosion and ρ is the density of the surrounding air. (c) Top view of the spreading of a silicon oil droplet that is being spin coated.125 The dynamics of the contact radius d follows where E is the yield of the explosion and ρ is the density of the surrounding air. (c) Top view of the spreading of a silicon oil droplet that is being spin coated.125 The dynamics of the contact radius d follows 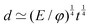 (eqn (38)), where E is the centrifugal energy and φ ≃ η/ (eqn (38)), where E is the centrifugal energy and φ ≃ η/![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) is an interfacial friction derived from the viscosity and size of the drop. (d) Top view of the spreading of a cell onto a rigid substrate covered with an extracellular matrix.99 The dynamics of the contact radius d follows is an interfacial friction derived from the viscosity and size of the drop. (d) Top view of the spreading of a cell onto a rigid substrate covered with an extracellular matrix.99 The dynamics of the contact radius d follows 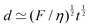 (eqn (51)), where F is the ‘protrusion force’ and η is the viscosity of the cell. (e) Top view of the spreading of an aggregate of cells onto a rigid substrate covered by extracellular matrix.126 The dynamics of the contact radius d follows (eqn (51)), where F is the ‘protrusion force’ and η is the viscosity of the cell. (e) Top view of the spreading of an aggregate of cells onto a rigid substrate covered by extracellular matrix.126 The dynamics of the contact radius d follows 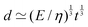 (eqn (39)), where E ≃ Γ (eqn (39)), where E ≃ Γ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 2 is the adhesion energy, and η is the viscosity of the aggregate. 2 is the adhesion energy, and η is the viscosity of the aggregate. | ||
4.1 General formula
In the previous section we only considered pairs of mechanical quantities on the same lines or on the same columns of Table 1. Pairs on the same line yield simple lengths, and pairs on the same column yield simple times. We now consider any arbitrary pair of mechanical quantities, Q1(x1, y1) and Q2(x2, y2). In this general case, the dimensions of the mechanical ratio combine space and time, and the dimension of mass naturally disappears: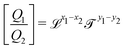 | (26) |
4.2 Simple speeds
https://youtu.be/VvMKwZSNEX4?si=l6aLuzYAhzjqjxfvSimilarly, just as pairs of mechanical quantities can combine to give simple lengths or times, they can also produce speeds. Indeed, for pairs of mechanical quantities satisfying x1 − x2 = y2 − y1, eqn (26) implies
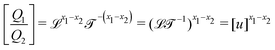 | (27) |
 | (28) |
These speeds give us a preview of the relationship between mechanics and motion, in the special case where this motion is ‘uniform’, i.e., at constant speed.
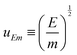
| (29) |
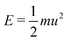 . In this context, the speed u is much smaller than the speed of light. Usually, this formula is used to compute the energy from a known mass m and speed u. However, the formula can be rearranged to express the speed from the mass and energy, as in eqn (29). The mass can be that of a projectile, like a canon ball, or a bullet, and the energy is that delivered by the gun.
. In this context, the speed u is much smaller than the speed of light. Usually, this formula is used to compute the energy from a known mass m and speed u. However, the formula can be rearranged to express the speed from the mass and energy, as in eqn (29). The mass can be that of a projectile, like a canon ball, or a bullet, and the energy is that delivered by the gun.
The speed uEm can be used to rationalize the speed of all sorts of projectiles, including bullets racing in a straight line, as well as debris flying in all directions, as in the case of explosions–small, large, or even astronomical. This kind of speed can, for instance, be used to describe the early stage of supernova explosions. For some types of supernovae (type Ia), both the mass and energy are known with some confidence. The mass of the ‘progenitor’, i.e., the exploding star, is around the same as that of our Sun, so m ≃ 2 × 1030 kg, and the energy is around E ≃ 2 × 1044 J. In this context, the early speed of the leading edge of the supernova remnant can be estimated from eqn (29), reaching a daunting 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 km per second!
000 km per second!
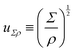
| (30) |
The sound speed uΣρ can also appear in other fields, for instance, in astrophysics, and magnetohydrodynamics, where it is sometimes called the ‘Alfvén speed’, when the stress is built from a magnetic field strength B as Σ = B2/μ0, where μ0 is the permeability of the vacuum.127 Note that just like the Boltzmann constant was used to translate a temperature into an energy ([kB] = [E]/[Θ]), and the permittivity ε0 was used to translate charges into a strength ([ε0] = [e]2/[S]), here the permeability is used to translate a magnetic field into a stress ([μ0] = [B]2/[Σ]). These translation constants allow one to remain within the 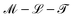 system.
system.
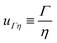
| (31) |
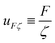
| (32) |
In eqn (32), the force F needs not be the weight. For instance, the standard acceleration of gravity can be superseded by a centrifugal acceleration, g ≃ rω2, which can be orders of magnitude larger than the standard g0. Then eqn (32) can be written as u ≃ mrω2/ζ, a formula very useful in biology, chemistry, and physics, to separate objects based on their different sedimentation speeds. The equation can be rearranged as u/g ≃ m/ζ. On the left, the sedimentation speed is divided by the effective acceleration, and is sometimes called the sedimentation coefficient, measured in Svedberg, after Theodor Svedberg, the Swedish chemist who got a Nobel prize for his study of colloids and proteins and the development of the ultracentrifuge.69,132,133 By definition, one Svedberg is equal to 10−13 s, and indeed speed divided by acceleration yields time. The right hand side of the equation reveals that this kinematic ratio of speed and acceleration can also be understood mechanically as a ratio of mass and friction. So the sedimentation coefficient is a simple time, τζm, built from the mass and friction, both belonging to a pair on the same column of Table 1, which we can add to the list of simple times we started in the previous section.
Note that the most general formulation of this simple speed does not require the force to be connected to any mass. For sedimentation and centrifugation the force is known, but in recent years, this simple formula has also been used the other way around, to estimate the magnitude of an unknown driving force F from a known friction, as in the case of motile cells or organisms. For instance, consider a swimming bacterium; between turning points, the bacterium moves at an approximately constant speed, u ≃ 30 μm s−1. From eqn (32), we can obtain an estimate of the driving force F from the speed u, if we also know friction ζ. In viscous fluids, as we saw in eqn (17), the friction can be related to the size ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) of the moving object and to the viscosity of the fluid, as ζ ≃
of the moving object and to the viscosity of the fluid, as ζ ≃ ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) η. The driving force can then be expressed as F ≃ η
η. The driving force can then be expressed as F ≃ η![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) u. For a swimming E. coli with u ≃ 30 μm s−1 and
u. For a swimming E. coli with u ≃ 30 μm s−1 and ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ 2 μm, the surrounding medium is around 10 times more viscous than water, so η ≃ 10−2 Pa s. Overall, F ≃ 10−12 N, i.e., one piconewton, which is indeed the correct order of magnitude, although the numerical prefactors we ignored can increase this force to a few tens of piconewtons.43,44
≃ 2 μm, the surrounding medium is around 10 times more viscous than water, so η ≃ 10−2 Pa s. Overall, F ≃ 10−12 N, i.e., one piconewton, which is indeed the correct order of magnitude, although the numerical prefactors we ignored can increase this force to a few tens of piconewtons.43,44
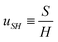
| (33) |
Note that the speed of light itself can be expressed using eqn (33). Since c = S0/ℏα0, we can define an action H = hα0, or a strength S = S0/α0, which would provide slightly different ways to think about the speed of light.
4.3 Non-uniform motion
https://youtu.be/jv6IKH7aO5s?si=55dEy2nCWsS_51GTFrom Antiquity to the Middle Ages, motion was practically synonymous with uniform motion, where distances grow linearly with time, as d = ut. The great leap made by Galileo, Kepler and Newton was in no small part driven by their departure from this narrow focus on motion at constant speed. Generations of thinkers had been fascinated by speed, with dimensions 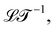 but the Renaissance shifted the attention toward acceleration,
but the Renaissance shifted the attention toward acceleration, 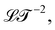 in particular with the study of free-fall, where the fallen distance grows quadratically, as
in particular with the study of free-fall, where the fallen distance grows quadratically, as 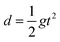 . Unfortunately, this revolutionary takeover turned into a new dogma, and for centuries acceleration became the imposed kinematic metric of motion. It was only toward the end of the 19th century that the existence of other types of motion resurfaced with the study of diffusion.134 For diffusive processes, distances grow proportionally to the square root of time,
. Unfortunately, this revolutionary takeover turned into a new dogma, and for centuries acceleration became the imposed kinematic metric of motion. It was only toward the end of the 19th century that the existence of other types of motion resurfaced with the study of diffusion.134 For diffusive processes, distances grow proportionally to the square root of time, 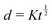 , which is usually written as
, which is usually written as 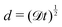 , introducing the ‘diffusivity’ or ‘diffusion coefficient’
, introducing the ‘diffusivity’ or ‘diffusion coefficient’ 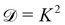 , with
, with 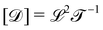 .69,70,135 For reasons beyond our scope, kinematic quantities just like standard mechanical quantities are usually defined in such a way as to have integer exponents.
.69,70,135 For reasons beyond our scope, kinematic quantities just like standard mechanical quantities are usually defined in such a way as to have integer exponents.
Motions characterized by constant speed, constant acceleration, or “diffusive behavior” are the three most historically significant examples of motion, but they are in no way more fundamental than other types of motions discovered subsequently. For instance, following Taylor,7,8 we have seen that the blast of an explosion may advance according to a ‘power law’ 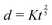 . In this case, the kinematic parameter K is neither a speed, nor acceleration, nor given by a diffusivity. The kinematic quantity
. In this case, the kinematic parameter K is neither a speed, nor acceleration, nor given by a diffusivity. The kinematic quantity 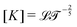 (or
(or 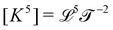 if we prefer integer exponents) is a more unusual combination of time and space. Like most kinematic quantities, K5 does not have a standard name, but it deserves a specific designation. In our https://www.youtube.com/playlist?list=PLbMiQs7eX-bbNTc-7HwdWzohUs8yPw300, we took the liberty of calling it explosivity. An explosion as the one studied by Taylor corresponds to a motion at constant explosivity. Just like speed, acceleration, or diffusivity, explosivity can be understood as a ratio of a pair of mechanical quantities. In Taylor's analysis, K5 ≃ E/ρ, where E is the energy of the bomb and ρ the density of the ambient medium. This relationship is a direct consequence of eqn (26). Indeed, since energy and density are five columns and two lines apart in Table 1, we have:
if we prefer integer exponents) is a more unusual combination of time and space. Like most kinematic quantities, K5 does not have a standard name, but it deserves a specific designation. In our https://www.youtube.com/playlist?list=PLbMiQs7eX-bbNTc-7HwdWzohUs8yPw300, we took the liberty of calling it explosivity. An explosion as the one studied by Taylor corresponds to a motion at constant explosivity. Just like speed, acceleration, or diffusivity, explosivity can be understood as a ratio of a pair of mechanical quantities. In Taylor's analysis, K5 ≃ E/ρ, where E is the energy of the bomb and ρ the density of the ambient medium. This relationship is a direct consequence of eqn (26). Indeed, since energy and density are five columns and two lines apart in Table 1, we have:
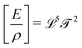 | (34) |
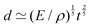 , as being the natural expression of the ratio E/ρ when time is measured by t and space by d. We shall use these two symbols, t and d, instead of τ and
, as being the natural expression of the ratio E/ρ when time is measured by t and space by d. We shall use these two symbols, t and d, instead of τ and ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , in order to underline the fact that the length and time are variable here.
, in order to underline the fact that the length and time are variable here.
This way of representing kinematics as an evolution law for a size d(t) is pretty visual, so we will adopt it in this entire subsection, but as we shall see in Section 5 it is by no means the only perspective on kinematics. From this length versus time perspective the general formula in eqn (26) can be expressed as a ‘scaling law’ or regime:
| Q1/Q2 ≃ dx1−x2ty1−y2 | (35) |
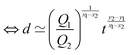 | (36) |
Note the use of the approximate equality ‘≃’ in eqn (36), which underlines the fact that this relationship may not be exact, depending on the precise definitions of the mechanical parameters (Q1 and Q2) and kinematic variables (d and t). For now, we will consider that the mechanical quantities Q1 and Q2 provide a satisfying model of the dynamics if the two sides of eqn (36) only differ by numerical factors ‘of order 1’. We will return to this point in Section 6.3.2.
Note also that eqn (36) includes growing regimes, where (y2 − y1)/(x1 − x2) > 0, and shrinking regimes, where (y2 − y1)/(x1 − x2) < 0. In these shrinking regimes, the length d diverges at initial time, and it only converges to zero for t → ∞. In this review, we will focus on growing regimes. We differ a discussion of shrinking regimes to a future publication.
In Table 1, we have defined 25 widely used mechanical quantities. Considering all pairs, would generate more than 300 regimes. If we remove the simple lengths, times, and speeds, and if we only focus on dynamics where the size grows over time (d ∼ tα with α > 0), there are still more than 100 possible regimes. This large number reflects the great diversity of “physics” that can be at play in different situations. In the following sub-sections, we will evidently not discuss all possibilities, but we will show that regimes of all sorts have already been used to describe dynamics across scales and disciplines.
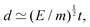 and Taylor's regime of explosions,
and Taylor's regime of explosions, 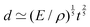 . Taylor's regime is depicted in Fig. 2b, and the scaling is plotted in Fig. 3a.
. Taylor's regime is depicted in Fig. 2b, and the scaling is plotted in Fig. 3a.
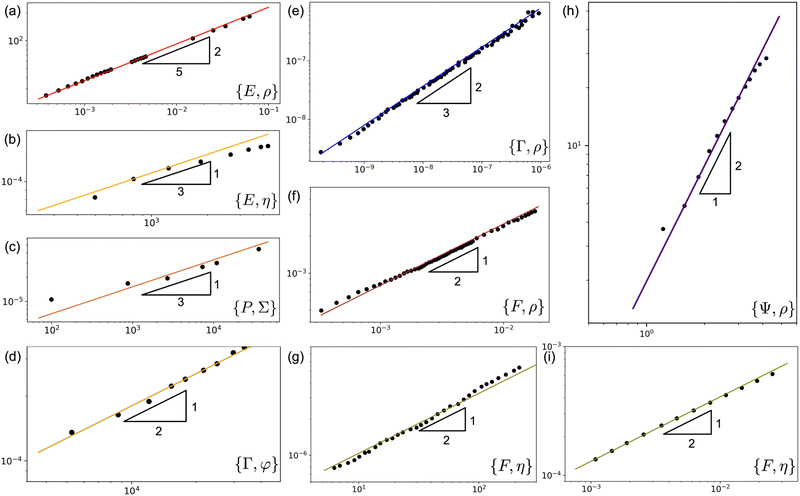 | ||
| Fig. 3 Growth of a length d over time t for regimes observed in dynamics from atomic explosions to living cells. The lengths and times are respectively measured by the vertical and horizontal axes, in meters and seconds for all plots. All plots show power laws of the form d = Ktα. On each plot the value of α is represented by a triangle giving the slope of the line. The pair of mechanical quantities invoked to rationalize the slopes are given at the bottom-right corner. Note that in some cases the data have been truncated to isolate the range of validity of the regime in focus, undisturbed by the effect of additional mechanical quantities (see the conclusion of the review for a discussion of this point). (a) Blast radius of the Trinity explosion.8 (b) Initial spreading of a spherical aggregate of cells under ‘partial wetting’ conditions.126 (c) Grain size growth for the ‘Ostwald ripening’ regime of ‘sintering’.136 (d) Spreading of the ‘precursor film’ of motile cells for the ‘complete wetting’ of a spherical aggregate of cells.126 (e) Inertio-capillary pinching of a bridge of liquid mercury.137 In this case, the time t is the duration before pinch-off, so the “actual time” runs from right to left. (f) Spreading of a water droplet.138 (g) Spreading of a single cell.98 (h) Distance traveled by a debris flow down an incline.139 (i) Radius of the neck for the coalescence of two air bubbles in silicone oil.140 | ||
If we seek additional regimes impelled by energy, quantities on the same line or column as E cannot be included since they yield simple lengths and times. Quantities represented on the line with an index y = −3 cannot be included because they result in shrinking behavior (d ∼ tα, with α < 0) rather than growth (α > 0). Of the quantities that are left, we have chosen to highlight the 2D density Λ ≡ Q(−2, 0), the mass flux φ ≡ Q(−2, −1) (understood as an interfacial friction Q(−2, −1) = Q(0 − 2, −1)), and the viscosity η ≡ Q(−1, −1):
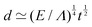 | (37) |
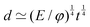 | (38) |
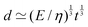 | (39) |
![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , one can define a 2D density as Λ ≃ ρ
, one can define a 2D density as Λ ≃ ρ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , leading to a regime with
, leading to a regime with 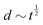 . This regime can be used to describe exploding-bridgewire detonators.141
. This regime can be used to describe exploding-bridgewire detonators.141
The second example can be used to describe the dynamics of the radius of contact of ‘spin-coated’ drops, as illustrated in Fig. 2c.125 In this case, the spinning is associated with a centrifugal energy E = ρ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 5ω2, where
5ω2, where ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 3 is the volume of a drop and ω is the rotational frequency. One can then define a form of ‘interfacial friction’ from the fluid viscosity as φ = η/
3 is the volume of a drop and ω is the rotational frequency. One can then define a form of ‘interfacial friction’ from the fluid viscosity as φ = η/![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) . With these definitions, the spreading of the spun drop follows eqn (38). Note that quantities with the same dimensions have been used to study the “friction” of fluids, polymers and elastomers on solid boundaries.142,143 In this context, one can also define the so-called Navier slip length,
. With these definitions, the spreading of the spun drop follows eqn (38). Note that quantities with the same dimensions have been used to study the “friction” of fluids, polymers and elastomers on solid boundaries.142,143 In this context, one can also define the so-called Navier slip length, ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ≃ η/φ, a form of simple length measured by extrapolating the velocity profile beyond the boundary.144 We will see another use of this kind of ‘interfacial friction’ φ in eqn (47).
≃ η/φ, a form of simple length measured by extrapolating the velocity profile beyond the boundary.144 We will see another use of this kind of ‘interfacial friction’ φ in eqn (47).
The third example in eqn (39) describes a regime where viscosity prevents the spreading of a source of energy. This regime could be relevant for point-like inputs of energy in very viscous fluids. This input could, for instance, come from explosions, lasers,145 or ultrasounds.146,147 Interestingly, this regime has also been applied to contexts far from explosions, to describe the spreading of aggregates of cells, as illustrated in Fig. 2e.126 In this context, a ball of cells comes in contact with a substrate, on which it starts spreading through cell migration. The cell–substrate adhesion Γ and the size ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) of the ball can be used to define an adhesion energy E ≃ Γ
of the ball can be used to define an adhesion energy E ≃ Γ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) 2, such that the spreading abides eqn (39), where η is the aggregate viscosity. A comparison between this mechanical model and the data is shown in Fig. 3b.
2, such that the spreading abides eqn (39), where η is the aggregate viscosity. A comparison between this mechanical model and the data is shown in Fig. 3b.
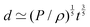 | (40) |
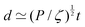 | (41) |
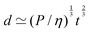 | (42) |
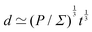 | (43) |
Eqn (41) gives another example of a regime based on power. This equation defines the simple speed 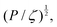 so we could have put it in Section 4.2. This speed is relevant to dynamics characterized by a constant friction ζ. In many situations, the friction ζ is not constant and depends on speed. In general one can define ζ from the friction force F as ζ = F/u, where u is the speed. In the ‘inertial regime’, the friction force is proportional to the square of the speed, such that ζ ≃ ρud2. Using u ≃ d/t this definition of the friction would lead back to eqn (40). In contrast, in the ‘viscous regime’, the friction force is proportional to speed and given by ζ ≃ ηd.100 If d is the variable distance, this leads to the regime given in eqn (42). However, in some cases d is a constant length, for instance, connected to the size of a vehicle. The quantity ζ would then be a constant parameter and eqn (41) may apply.
so we could have put it in Section 4.2. This speed is relevant to dynamics characterized by a constant friction ζ. In many situations, the friction ζ is not constant and depends on speed. In general one can define ζ from the friction force F as ζ = F/u, where u is the speed. In the ‘inertial regime’, the friction force is proportional to the square of the speed, such that ζ ≃ ρud2. Using u ≃ d/t this definition of the friction would lead back to eqn (40). In contrast, in the ‘viscous regime’, the friction force is proportional to speed and given by ζ ≃ ηd.100 If d is the variable distance, this leads to the regime given in eqn (42). However, in some cases d is a constant length, for instance, connected to the size of a vehicle. The quantity ζ would then be a constant parameter and eqn (41) may apply.
Eqn (43) gives yet another example of a regime driven by power, where the impeding quantity is stress. This equation may be applicable to ‘sintering’.136 In this process, the typical size of grains grows as 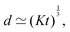 where in the so-called ‘Ostwald ripening’ regime, the grain growth rate can be written as
where in the so-called ‘Ostwald ripening’ regime, the grain growth rate can be written as 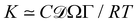 . The parameter R is the ideal gas constant, and Ω is the molar volume, such that Σ ≡ RT/Ω is a characteristic thermal stress. The constant C is a dimensionless solubility, Γ is the solid–liquid surface energy and
. The parameter R is the ideal gas constant, and Ω is the molar volume, such that Σ ≡ RT/Ω is a characteristic thermal stress. The constant C is a dimensionless solubility, Γ is the solid–liquid surface energy and 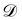 is the solid diffusivity in the liquid. Thus, one can define the power associated with an increase in the size of the grains as
is the solid diffusivity in the liquid. Thus, one can define the power associated with an increase in the size of the grains as 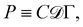 such that the sintering equation becomes an example of eqn (43). An example of this regime is given in Fig. 3c.136
such that the sintering equation becomes an example of eqn (43). An example of this regime is given in Fig. 3c.136
| d ≃ (Ψ/ρ)t2 | (44) |
Note that eqn (44) may also describe rises rather than falls, in the context of buoyancy. In this case, the force–density takes into account a difference in density between two materials Ψ ≃ |ρf − ρ|g. This version of eqn (44) would, for instance, be useful for understanding the initial rise of a mushroom cloud after a nuclear explosion such as Trinity.8 Indeed, the blast generates a zone of very low density, which acts as a bubble inside the comparatively denser air.
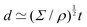 . Let us also mention the following ‘diffusive’ regime:
. Let us also mention the following ‘diffusive’ regime: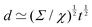 | (45) |
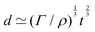 | (46) |
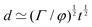 | (47) |
| d ≃ (Γ/Φ)t2 | (48) |
The regime given in eqn (47) describes dynamics driven by a surface energy Γ, but impaired by a mass flux or momentum density φ. An example of this regime is shown in Fig. 3d.126 In this example, the dynamics describe the spreading of the ‘precursor film’ composed of motile cells moving away in 2D after the contact of a spherical aggregate. In this context, the surface energy arises from the adhesion of cells with the substrate, and the parameter φ is understood as a form of “friction”.126
In eqn (48) the term ‘1D density’ Φ can be interpreted as a ‘normal stress coefficient’, as mentioned in Section 3.2. The ratio Γ/Φ yields acceleration, which has some relevance to the free-surface flows of visco-elastic liquids. In particular, eqn (48) has been discussed in the context of the pinching of the so-called ‘second order fluids’.14
| d ≃ (F/m)t2 | (49) |
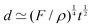 | (50) |
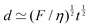 | (51) |
In eqn (50), the force is balanced by density rather than mass. This regime applies to spreading, coalescence and pinching of drops when the whole size of the drop has a substantial impact.18,106,107,151–153 In this context, the force can be expressed as F ≃ Γ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , where Γ is the surface-tension and
, where Γ is the surface-tension and ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) is the size of the drop. An example of this regime is given in Fig. 3f for the spreading of a water drop.138
is the size of the drop. An example of this regime is given in Fig. 3f for the spreading of a water drop.138
In eqn (51), the force is balanced by viscosity. This regime has been observed in a few different contexts. For instance, this regime describes the coalescence of drops in a viscous outer fluid, where η is the viscosity of the outer fluid, as shown in Fig. 3i.140 A similar regime can be used to describe the spreading of thin films, where F/η is understood as an effective diffusivity, with F ≃ E/![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , where E is the Hamaker constant and
, where E is the Hamaker constant and ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) the film thickness.19,159 Since the Hamaker constant is usually on the order of the thermal energy, the effective diffusivity matches with Stokes diffusivity. The same equation was also used to describe the early spreading of single cells, as illustrated in Fig. 2d.99 In this case, the viscosity corresponds to that of the cell and the force can be attributed either to the stiffness of the cytoskeleton, or to a ‘protrusion force’ originating from the polymerization of ‘actin’.98,99 An example of such scaling in the early spreading of cells is given in Fig. 3g.98
the film thickness.19,159 Since the Hamaker constant is usually on the order of the thermal energy, the effective diffusivity matches with Stokes diffusivity. The same equation was also used to describe the early spreading of single cells, as illustrated in Fig. 2d.99 In this case, the viscosity corresponds to that of the cell and the force can be attributed either to the stiffness of the cytoskeleton, or to a ‘protrusion force’ originating from the polymerization of ‘actin’.98,99 An example of such scaling in the early spreading of cells is given in Fig. 3g.98
4.4 Impelling and impeding
https://youtu.be/J5tm6IXt_fU?si=bPbBS5hD6SkaO3wmIf two mechanical quantities are given, there is a unique regime associated with them. For this reason, we may use the notation {Q1, Q2} to denote the regime associated with the pair of mechanical quantities Q1 and Q2. For instance, Taylor's regime is {E, ρ}, or {ρ, E}. The order of the quantities between brackets does not matter. In the pair {E, ρ}, the energy will always be the motor, or impelling factor, in accordance with eqn (36). Let us recall this equation here so there is no need to turn the pages:
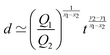 | (52) |
Although in a pair, one quantity is always driving; what is driving in one situation can be resisting in another and vice versa. This fact was clearly not understood when the first few mechanical quantities were defined, and to this day it remains the source of a lot of confusion. We only need to look at the historical names of the quantities in Table 1 to see that some of them are quite heavily connoted. On the right of the table, where these quantities are more likely to be impelling motion, the names are markedly positive, like ‘action’, ‘energy’, ‘force’, or ‘power’. All these “macho” terms in the English language are here to remind us that these quantities were thought as movers. For a lot of early scientists they were nothing short of the hand of God in the physical world. In contrast, terms like ‘friction’, ‘mass’, or ‘viscosity’, were initially thought of as sticky, gooey, resisting or at most inert rather than active. In fact, these quantities can be active; they can be impelling motion rather than impeding it, under the right circumstances. We shall illustrate this versatility on two historically significant examples.
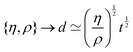 | (53) |
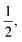 this motion can be said to be ‘diffusive’, and the mechanical ratio ν ≡ η/ρ can be interpreted as (‘momentum’) diffusivity. It is also called ‘kinematic viscosity’. The greater the viscosity the greater the diffusivity, so indeed, viscosity is here impelling motion! It is surprising that eqn (53) does not involve the speed of the moving boundary. The dynamics of the boundary layer are actually independent of this speed only up to a point, where the flow becomes turbulent. So eqn (53) only refers to ‘laminar boundary layers’.101
this motion can be said to be ‘diffusive’, and the mechanical ratio ν ≡ η/ρ can be interpreted as (‘momentum’) diffusivity. It is also called ‘kinematic viscosity’. The greater the viscosity the greater the diffusivity, so indeed, viscosity is here impelling motion! It is surprising that eqn (53) does not involve the speed of the moving boundary. The dynamics of the boundary layer are actually independent of this speed only up to a point, where the flow becomes turbulent. So eqn (53) only refers to ‘laminar boundary layers’.101
At the end of the 16th century, building on the observations of his master Tycho Brahe, Kepler had established his now famous ‘third law of planetary motion’: t2 ∼ d3. The square of a planet's orbital period, t, is proportional to the cube of the length of the semi-major axis of its orbit, d.160 If we want we can of course express this law in the following fashion: 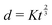 , where the unknown kinematic prefactor K must have dimensions
, where the unknown kinematic prefactor K must have dimensions  , so
, so 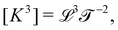 a rather odd kinematic quantity that is nowadays called the ‘gravitational parameter’. In the case of the solar system the data available to Kepler established that K3 ≃ 3 × 1018 m3 s−2. The mechanical interpretation of this kinematic quantity had to wait for one more generation, with the work of Newton.
a rather odd kinematic quantity that is nowadays called the ‘gravitational parameter’. In the case of the solar system the data available to Kepler established that K3 ≃ 3 × 1018 m3 s−2. The mechanical interpretation of this kinematic quantity had to wait for one more generation, with the work of Newton.
Fig. 4a demonstrates the validity of Kepler's law for the orbits of the planets of the solar system. Also included are the characteristics (periods and semi-major axes) of the orbits of the satellites of the various planets of the solar system. Concomitantly with Kepler's investigations, Galileo had discovered the four largest moons of Jupiter,161 now called the ‘Galilean moons’. As evident in Fig. 4a, the Galilean moons, as well as the moons of other planets also follow power laws of the form 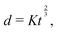 but the value of the kinematic prefactor K changes from one system to another.
but the value of the kinematic prefactor K changes from one system to another.
It was Newton who first realized that the prefactor K is not a “universal” constant, but that it depends on the “world” under study.162 Newton then set out to find a decomposition of the kinematic quantity K into mechanical quantities. Using the notation introduced in this review, we could say that Newton sought a pair of mechanical quantities Q1(x1, y1) and Q2(x2, y2) such that 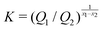 . As shown in Fig. 4b, Newton realized that K3 ∼ m, with the impelling mechanical quantity being the central mass of the orbit, Q1 = m, where m is for instance the mass of the Sun for the solar system.162
. As shown in Fig. 4b, Newton realized that K3 ∼ m, with the impelling mechanical quantity being the central mass of the orbit, Q1 = m, where m is for instance the mass of the Sun for the solar system.162
Since 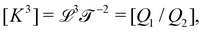 and
and 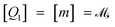 Newton's second mechanical quantity had to have the following dimensions:
Newton's second mechanical quantity had to have the following dimensions: 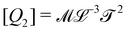 . Thus, Q2 = Q(−3, 2). Looking back at Table 1, we see that this mechanical quantity occupies an odd position in comparison to all the others, which probably explains its peculiar fate. A quantity Q(−3, 2) is what we refer to as levity, denoted by the symbol
. Thus, Q2 = Q(−3, 2). Looking back at Table 1, we see that this mechanical quantity occupies an odd position in comparison to all the others, which probably explains its peculiar fate. A quantity Q(−3, 2) is what we refer to as levity, denoted by the symbol  . Since this mysterious quantity is on the left of the mass (in the table), it is impeding motion, whereas mass is impelling it:
. Since this mysterious quantity is on the left of the mass (in the table), it is impeding motion, whereas mass is impelling it:
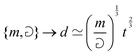 | (54) |
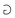 with the inverse of the so-called ‘universal constant of gravity’ G. More precisely,
with the inverse of the so-called ‘universal constant of gravity’ G. More precisely, 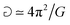 (assuming the central mass is much more massive than the orbiting objects).
(assuming the central mass is much more massive than the orbiting objects).
The purpose of defining such “levity” is to stress that Kepler's law—like any other regime—can be expressed from a pair of standard mechanical quantities with dimensions of the form 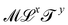 . In contrast, the universal constant of gravity has dimensions
. In contrast, the universal constant of gravity has dimensions 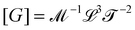 . Other mechanical quantities with similar negative power of the mass have been occasionally used, like the ‘fluidity’ with dimensions
. Other mechanical quantities with similar negative power of the mass have been occasionally used, like the ‘fluidity’ with dimensions 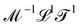 .164 These quantities are absolutely valid, but they are unnecessary, since they can be reduced to standard quantities through inversion. We do believe that the “levity” deserves its place in Table 1 and should be free to interact with all the other quantities. As we saw, pairing levity and mass leads to Kepler's law for orbital motion, but we invite the reader to try different pairings. For instance,
.164 These quantities are absolutely valid, but they are unnecessary, since they can be reduced to standard quantities through inversion. We do believe that the “levity” deserves its place in Table 1 and should be free to interact with all the other quantities. As we saw, pairing levity and mass leads to Kepler's law for orbital motion, but we invite the reader to try different pairings. For instance, 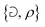 gives rise to a simple time
gives rise to a simple time 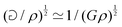 , which is called the ‘free-fall time’, with important applications in astrophysics.73
, which is called the ‘free-fall time’, with important applications in astrophysics.73
5 Perspectives on motion
https://youtu.be/lVLjflQI41w?si=m3h0d3Kvm6hI_2e6Since eqn (36), we have presented mechanics from a particular kinematic perspective. The assumption was that we observed some kind of motion, like an explosion, or the pinching of a water droplet, or the fall of an object, or the orbit of a planet. With these examples, we represented motion by plotting length d versus time t, as in Fig. 3. That length could be a size or a distance, and the time could be an indefinite duration, or a recurring period. We have seen that the interplay of a pair of mechanical quantities Q1 and Q2 produces a single kinematic power law, d = Ktα, where the kinematic prefactor K and the dimensionless exponent α are obtained from the underlying mechanical quantities. As soon as we know the relative dimensions of the two mechanical quantities Q1 and Q2, we know the scaling connecting kinematics and mechanics.
Describing motion as the time series of a distance d(t) is quite visual; however, not all dynamics can easily be understood in this fashion, and it is sometimes much more practical to use different pairs of kinematic variables. Each pair of kinematic variables corresponds to a different perspective on the dynamics. In some situations, it may even be useful to manipulate mechanical rather than kinematic variables. These different kinematic and mechanical perspectives provide complementary approaches on the same physics. The “physics” is set by the quantities Q1 and Q2, and the perspectives are set by the choice of variables. The exact forms of the scaling differ from one perspective to another, but they are always the direct consequence of dimensional analysis.
5.1 Kinematic perspectives
For a given pair of mechanical quantities, we know since eqn (26) that the dimensions of their ratio can be expressed as a combination of space and time: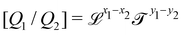 . So far, we have considered cases where the spatial dimension could be associated with a variable size d, and the time dimension could be associated with a variable time t. However, in some situations the more obvious variables are different kinematic combinations.
. So far, we have considered cases where the spatial dimension could be associated with a variable size d, and the time dimension could be associated with a variable time t. However, in some situations the more obvious variables are different kinematic combinations.
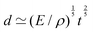 . This perspective on the dynamics is convenient, because the validity of this regime can simply be checked by measuring the radius of the blast on pictures captured at different instants,8,165 as those reproduced in Fig. 2b. However, one may rather choose to record this explosion by measuring the speed of the blast, v, as it passes by detectors placed at different distances d from ground zero. What should we expect for v(d)? If v is defined as the instantaneous speed of the front, then v ≡ ∂d/∂t = 2d/5t, which can be expressed solely from the variable length d, as
. This perspective on the dynamics is convenient, because the validity of this regime can simply be checked by measuring the radius of the blast on pictures captured at different instants,8,165 as those reproduced in Fig. 2b. However, one may rather choose to record this explosion by measuring the speed of the blast, v, as it passes by detectors placed at different distances d from ground zero. What should we expect for v(d)? If v is defined as the instantaneous speed of the front, then v ≡ ∂d/∂t = 2d/5t, which can be expressed solely from the variable length d, as 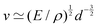 (where numerical factors are absorbed by the approximate equality). This equation still displays the same physics combining energy and density, but it is expressed from the perspective of speed versus distance, rather than distance versus time. We may write this shift in perspective by adding indices to the pair of mechanical quantities underlining this type of motion:
(where numerical factors are absorbed by the approximate equality). This equation still displays the same physics combining energy and density, but it is expressed from the perspective of speed versus distance, rather than distance versus time. We may write this shift in perspective by adding indices to the pair of mechanical quantities underlining this type of motion: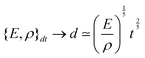 | (55) |
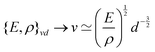 | (56) |
We have seen in eqn (22) that combining a viscosity and a stress yields a simple time 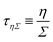 . In the case of a Newtonian fluid like water under normal conditions, this simple time can be understood as the inverse of the deformation rate on a fluid with viscosity η, upon applying a shear stress Σ at the boundary. The deformation rate can in turn be interpreted as a ‘velocity gradient’, since
. In the case of a Newtonian fluid like water under normal conditions, this simple time can be understood as the inverse of the deformation rate on a fluid with viscosity η, upon applying a shear stress Σ at the boundary. The deformation rate can in turn be interpreted as a ‘velocity gradient’, since 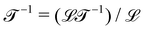 . So the interplay between viscosity and stress can also be written as v/d ≃ Σ/η, or as:
. So the interplay between viscosity and stress can also be written as v/d ≃ Σ/η, or as:
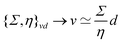 | (57) |
Another very useful flow is the pipe flow. Here, the profile is not linear but parabolic. Why? In this situation, the viscosity of the fluid is still relevant, but the flow is not driven by a stress at the boundary, but by a difference in pressure along the conduit. The impelling factor behind these dynamics is the gradient of pressure, which is also a force–density Ψ, since 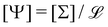 . Then, indeed, the combination of a force–density and a viscosity leads to a parabolic profile:
. Then, indeed, the combination of a force–density and a viscosity leads to a parabolic profile:
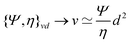 | (58) |
The scalings {Ψ,η}vd, {Σ,η}vd and {E,ρ}vd give three examples of velocity profiles, illustrating a general principle that can be applied to any pair of mechanical quantities.
 to the inverse of length
to the inverse of length  . We will give two famous examples related to the waves on the surface of liquids like water.
. We will give two famous examples related to the waves on the surface of liquids like water.
For small waves, also called ‘ripples’,166 the relevant mechanical quantities are surface tension Γ, and density ρ. We have seen this pair already, in eqn (46), in the context of pinching, coalescence and spreading of low viscosity fluids. In these cases, we saw that the interplay of the pair of mechanical quantities could be expressed as a power law of the form 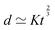 , where the length d was the radius of the neck for pinching and coalescence, or the radius of contact for spreading. The time t was either the time elapsed since contact for spreading and coalescence, or the time remaining before pinch-off. For ripples, the size d ≃ k−1 and the time t ≃ ω−1 are understood as the wavelength and period of the waves, so the perspective we started with, {Γ, ρ}dt, can be translated to a dispersion relation {Γ,ρ}ωk:
, where the length d was the radius of the neck for pinching and coalescence, or the radius of contact for spreading. The time t was either the time elapsed since contact for spreading and coalescence, or the time remaining before pinch-off. For ripples, the size d ≃ k−1 and the time t ≃ ω−1 are understood as the wavelength and period of the waves, so the perspective we started with, {Γ, ρ}dt, can be translated to a dispersion relation {Γ,ρ}ωk:
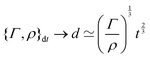 | (59) |
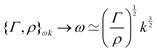 | (60) |
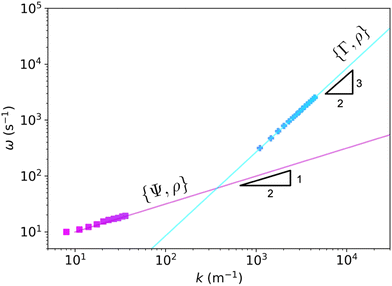 | ||
Fig. 5 Examples of dispersion relations of ‘capillary ripples’168 and ‘gravity waves’169 on the surface of water. The capillary ripples correspond to the pair 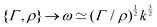 . The gravity waves correspond to . The gravity waves correspond to 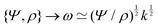 . . | ||
For Newtonian liquids like water, the dynamics of waves change when their wavelength becomes substantially larger than the capillary length 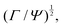 which we introduced in eqn (9). In this regime, the dominant impelling factor becomes the weight density Ψ ≃ ρg, while the impeding factor remains the ‘inertia’ of the fluid encapsulated in its density ρ. Thus, the struggle is between Ψ and ρ, and we have seen this balance before in eqn (44). Just as we did for ripples, we can compare the initial scaling with how the dynamics look when given as a dispersion relation:
which we introduced in eqn (9). In this regime, the dominant impelling factor becomes the weight density Ψ ≃ ρg, while the impeding factor remains the ‘inertia’ of the fluid encapsulated in its density ρ. Thus, the struggle is between Ψ and ρ, and we have seen this balance before in eqn (44). Just as we did for ripples, we can compare the initial scaling with how the dynamics look when given as a dispersion relation:
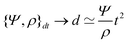 | (61) |
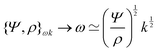 | (62) |
The cases discussed here provide two historically important examples of dispersion relations. Nevertheless, here again the procedure is general, and any regime can be expressed from such perspective if need be. We invite the reader to select any pair from Table 1, to derive the associated dispersion relation and to investigate if such a relation has been observed.
In the context of turbulence, one often measures what is routinely called an ‘energy spectrum’ 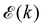 . The first variable, k, is a wavenumber, as in the case of dispersion relations, so with
. The first variable, k, is a wavenumber, as in the case of dispersion relations, so with 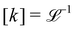 . The misleadingly called “energy”
. The misleadingly called “energy” 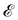 is actually a kinematic quantity, with
is actually a kinematic quantity, with 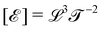 ,170 so it could be thought of as a specific strength (strength over mass). Fig. 6a gives a famous example of such an energy spectrum measured in the case of turbulence in a tidal channel.171 These measurements were among the first to validate a prediction from Kolmogorov,170 about the spectrum to be expected in the case where the turbulence is impelled by a power per unit volume Π and impaired by the density of the fluid ρ:
,170 so it could be thought of as a specific strength (strength over mass). Fig. 6a gives a famous example of such an energy spectrum measured in the case of turbulence in a tidal channel.171 These measurements were among the first to validate a prediction from Kolmogorov,170 about the spectrum to be expected in the case where the turbulence is impelled by a power per unit volume Π and impaired by the density of the fluid ρ:
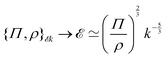 | (63) |
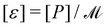 .170 This kinematic notation unfortunately obscures the underlying mechanical quantities, so we shall not adopt it. As we will see now, the spectrum given in eqn (63) provides a great exercise in translation between different perspectives.
.170 This kinematic notation unfortunately obscures the underlying mechanical quantities, so we shall not adopt it. As we will see now, the spectrum given in eqn (63) provides a great exercise in translation between different perspectives.
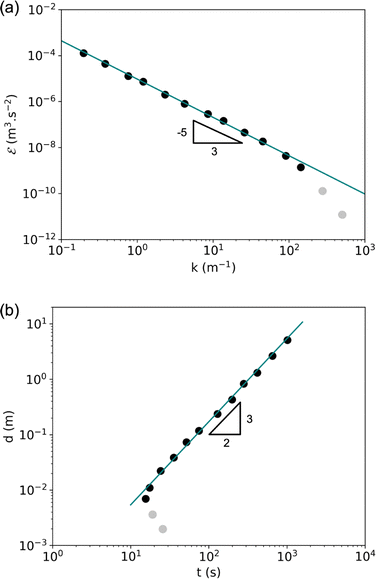 | ||
Fig. 6 (a) Kolmogorov's spectrum of ‘inertial turbulence’ {Π, ρ}![[scr E, script letter E]](https://www.rsc.org/images/entities/char_e140.gif) k (eqn (63)), observed in a tidal channel by Grant et al.171 (b) Translation of the spectrum into the evolution law for the size of strained “blobs” {Π, ρ}dt, using d ≡ k−1 and k (eqn (63)), observed in a tidal channel by Grant et al.171 (b) Translation of the spectrum into the evolution law for the size of strained “blobs” {Π, ρ}dt, using d ≡ k−1 and 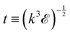 . Note that since the translation formulas neglect numerical factors, the resulting plot is only qualitative. In both (a) and (b) the grey data points deviate from Kolmogorov's scaling due to the effect of a third mechanical quantity: the viscosity of the fluid. We refer the reader to the https://youtu.be/1WP7k1VUf3Q?si=Muusfb3EDpNHB2I2 of our Mechanics series for a discussion on this crossover. It is beyond the scope of this review. . Note that since the translation formulas neglect numerical factors, the resulting plot is only qualitative. In both (a) and (b) the grey data points deviate from Kolmogorov's scaling due to the effect of a third mechanical quantity: the viscosity of the fluid. We refer the reader to the https://youtu.be/1WP7k1VUf3Q?si=Muusfb3EDpNHB2I2 of our Mechanics series for a discussion on this crossover. It is beyond the scope of this review. | ||
The variable 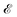 is called “energy” because its integral over all wavenumbers is defined by the variance of the flow speed, also understood as ‘specific energy’:
is called “energy” because its integral over all wavenumbers is defined by the variance of the flow speed, also understood as ‘specific energy’: 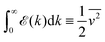 . If we maintain our neglect for numerical factors, all the subtleties of this integration should not concern us, and we can simply write
. If we maintain our neglect for numerical factors, all the subtleties of this integration should not concern us, and we can simply write 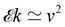 . The variable
. The variable 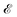 is connected to the magnitude v of velocity fluctuations for each wavenumber k. Larger wavenumbers are in turn related to smaller distances, with k ≃ 1/d. Thus, we can use Kolmogorov's spectrum given in eqn (63), and
is connected to the magnitude v of velocity fluctuations for each wavenumber k. Larger wavenumbers are in turn related to smaller distances, with k ≃ 1/d. Thus, we can use Kolmogorov's spectrum given in eqn (63), and 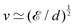 to express the interplay of power-density and mass-density from a relationship between the amplitude of velocity fluctuations and the size of these fluctuations:
to express the interplay of power-density and mass-density from a relationship between the amplitude of velocity fluctuations and the size of these fluctuations:
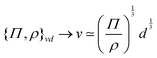 | (64) |
 but it provides a slightly different perspective on the pair {Π, ρ}. From this alternate vantage point, we can more easily translate the dynamics to the perspective of a length versus a time, which we adopted for most of this review. Let us do this with an intermediate step in between:
but it provides a slightly different perspective on the pair {Π, ρ}. From this alternate vantage point, we can more easily translate the dynamics to the perspective of a length versus a time, which we adopted for most of this review. Let us do this with an intermediate step in between: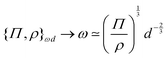 | (65) |
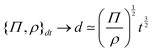 | (66) |
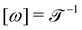 .101 This variable ω is also connected to the zones between vortices. Here, ω is the extension rate, when the vortices rotate in different directions, or the shear rate where locally the vortices rotate in the same direction. Through shear, extension, or rotation, ω is connected to the strain rate. So eqn (65) can be interpreted as relating the strain rate to the characteristic size of the strained region, whether it is a ‘vortex filament’, or a ‘shear layer’, or some more complicated structure.172–174
.101 This variable ω is also connected to the zones between vortices. Here, ω is the extension rate, when the vortices rotate in different directions, or the shear rate where locally the vortices rotate in the same direction. Through shear, extension, or rotation, ω is connected to the strain rate. So eqn (65) can be interpreted as relating the strain rate to the characteristic size of the strained region, whether it is a ‘vortex filament’, or a ‘shear layer’, or some more complicated structure.172–174
To conclude, we can mention the perspective given in eqn (66), size d versus time t, which is the initial approach of this review. In Table 1 the mass–density ρ and power–density Π are two columns apart and three lines apart so we get a size growing like time with a power  . We know that one way to think about the variable d is as the size of a strained region. We know that we can think of t as the period of velocity fluctuations at this length scale, but also as the inverse of the strain rate ω on a fluid region of size d. Yet another way to think about it is following the evolution of a strained “blob” of varying size d. We do not know a priori if the blobs are getting bigger or smaller, pinching like droplets. Some blobs might grow while other deflate, but in any case dimensional analysis gives us the power law relating size and time. Although tracking this process for a single blob entangled in a sea of countless others may be challenging, we can actually estimate what we would get by translating the measured energy spectrum, as shown in Fig. 6. For each measured values of
. We know that one way to think about the variable d is as the size of a strained region. We know that we can think of t as the period of velocity fluctuations at this length scale, but also as the inverse of the strain rate ω on a fluid region of size d. Yet another way to think about it is following the evolution of a strained “blob” of varying size d. We do not know a priori if the blobs are getting bigger or smaller, pinching like droplets. Some blobs might grow while other deflate, but in any case dimensional analysis gives us the power law relating size and time. Although tracking this process for a single blob entangled in a sea of countless others may be challenging, we can actually estimate what we would get by translating the measured energy spectrum, as shown in Fig. 6. For each measured values of ![[scr E, script letter E]](https://www.rsc.org/images/entities/char_e140.gif) and k, we can compute the associated values of d ≃ k−1 and
and k, we can compute the associated values of d ≃ k−1 and 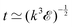 . Of course, since these translation formulas neglect all numerical factors, the resulting plot in Fig. 6b can only be regarded as qualitative, but the scaling is ensured.
. Of course, since these translation formulas neglect all numerical factors, the resulting plot in Fig. 6b can only be regarded as qualitative, but the scaling is ensured.
The four perspectives on turbulence given in eqn (63)–(66) – and any others we may like to adopt – are formally equivalent, but they collectively contribute to a richer and finer appreciation of the interplay between power–density and mass–density, the mechanical pair behind ‘inertial turbulence’. What we see is influenced by the perspective we have taken, and greater insight is reached by comparing what is perceived from different perspectives. We have seen this in some detail for the case of ‘inertial turbulence’ but the lesson is again general. In particular, we invite the reader to investigate the scalings associated with the growing number of ‘non-inertial’ types of turbulence, from visco-elastic fluids175 to active matter.176 Although these more exotic turbulent flows might be represented as energy spectra, ![[scr E, script letter E]](https://www.rsc.org/images/entities/char_e140.gif) ∼ kα, in these cases one finds that
∼ kα, in these cases one finds that 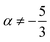 , because mechanical pairs beyond {Π, ρ} are involved—finding which pair is the subject of current research.
, because mechanical pairs beyond {Π, ρ} are involved—finding which pair is the subject of current research.
5.2 Mechanical perspectives
We have seen in the previous sub-section that the effects of any one pair of mechanical quantities can be expressed from different perspectives. So far, all the perspectives we considered were kinematic, which is to say that the pair of chosen variables always had dimensions of the form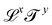 (allowing x or y to be zero). For instance, we saw that Taylor's regime of explosions,
(allowing x or y to be zero). For instance, we saw that Taylor's regime of explosions, 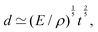 could also be expressed as a velocity profile,
could also be expressed as a velocity profile, 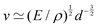 . In the first perspective the variables are d and t, and in the second, v and d. In both cases they are kinematic quantities in a broad sense (i.e. including geometric and chronometric quantities). In some circumstances one may rather prefer to focus on mechanical variables.
. In the first perspective the variables are d and t, and in the second, v and d. In both cases they are kinematic quantities in a broad sense (i.e. including geometric and chronometric quantities). In some circumstances one may rather prefer to focus on mechanical variables.
A proper discussion of mechanical perspectives would lead us astray from our purpose, but let us say a few words about it here, keeping explosions as our example. To illustrate our purpose, we shall consider the following equations, all equivalent formulations of Taylor's regime:
| E ≃ (ρd3)(d/t)2 | (67) |
| E/d ≃ (ρd3)(d/t2) | (68) |
| E/d3 ≃ ρ(d/t)2 | (69) |
| E/t ≃ (ρd3)(d/t2)(d/t) | (70) |
Let us first consider eqn (67). There, the dynamics of the explosion are presented as an energy balance. On the left-hand side is the constant input of energy E coming from the bomb. On the right-hand side, the variables are grouped in such a way as to compensate each other and yield a constant result. The combination 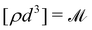 can be understood as a mechanical variable, representing the variable mass
can be understood as a mechanical variable, representing the variable mass ![[m with combining tilde]](https://www.rsc.org/images/entities/i_char_006d_0303.gif) of ambient air swept-away by the blast (we use tildes for mechanical variables to differentiate them from the constant mechanical parameters of the regimes).177 The entirety of the right-hand side can then be understood as the kinetic energy of the swept-up air, Ẽ ≃
of ambient air swept-away by the blast (we use tildes for mechanical variables to differentiate them from the constant mechanical parameters of the regimes).177 The entirety of the right-hand side can then be understood as the kinetic energy of the swept-up air, Ẽ ≃ ![[m with combining tilde]](https://www.rsc.org/images/entities/i_char_006d_0303.gif) v2, and Taylor's regime as E ≃ Ẽ, which can be interpreted as saying that the energy of the bomb is converted into the kinetic energy of the blast front.
v2, and Taylor's regime as E ≃ Ẽ, which can be interpreted as saying that the energy of the bomb is converted into the kinetic energy of the blast front.
If we now consider eqn (68), we can recover the Newtonian perspective on mechanics, a viewpoint cherished in so many textbooks. The terms of this equation are now forces, since [E/d] = [F]. We can introduce the variable “explosion force” ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) ≃ E/d. The term ρd3 ≃
≃ E/d. The term ρd3 ≃ ![[m with combining tilde]](https://www.rsc.org/images/entities/i_char_006d_0303.gif) is still the variable swept-up mass, and d/t2 ≃ a is the acceleration of the explosion front. Overall eqn (68) can be written as a force balance, similar to Newton's second law:
is still the variable swept-up mass, and d/t2 ≃ a is the acceleration of the explosion front. Overall eqn (68) can be written as a force balance, similar to Newton's second law: ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) ≃
≃ ![[m with combining tilde]](https://www.rsc.org/images/entities/i_char_006d_0303.gif) a.
a.
In eqn (69), the dynamics are now described as a balance of pressures. The left-hand side, 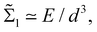 is the energy density of the explosion. The right-hand side,
is the energy density of the explosion. The right-hand side, 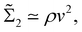 is the dynamic pressure emerging from the speed of the front, v ≃ d/t. Taylor's regime corresponds to a front moving in such a way that the varying energy density equals the dynamic pressure.
is the dynamic pressure emerging from the speed of the front, v ≃ d/t. Taylor's regime corresponds to a front moving in such a way that the varying energy density equals the dynamic pressure.
In eqn (70), the dynamics are described as a balance of power. The power of the explosion ![[P with combining tilde]](https://www.rsc.org/images/entities/i_char_0050_0303.gif) ≃ E/t is countered by
≃ E/t is countered by ![[m with combining tilde]](https://www.rsc.org/images/entities/i_char_006d_0303.gif) av.
av.
In these four examples we illustrated how a given regime can be interpreted in terms of mechanical variables. Here these mechanical variables are just built from algebraic manipulations, but in some cases they can be measured using instruments designed for this purpose. For instance, in the context of explosions one may perform pressure measurements at various distances from ground zero. Many such measurements were, for instance, performed for the Trinity test, enough to confirm the validity of eqn (69) from this perspective (cf. Barschall (1945) LA-352, Graves (1945) LA-354, Manley (1945) LA-360, Bright (1945) LA-366, Marley (1945) LA-431; all these reports are available in the Los Alamos National Laboratory https://sgp.fas.org/othergov/doe/lanl/).
For more information on this topic, we invite the reader to watch our lecture series on explosions, and more particularly the episode dedicated to kinematic and mechanical perspectives (https://youtu.be/bvVCvdB5Uzk?si=7BpPAD6HsvfYjpUe).
6 Simple dimensionless numbers
https://youtu.be/aun0CHanq94?si=w6CXSKyvzqmlqrqkWe have seen in the previous section that different perspectives provide complementary approaches on the same “physics”. In the framework of this review, these “physics” are conveniently expressed by a pair of mechanical quantities {Q1, Q2}, taken from Table 1, and these mechanical parameters manifest themselves as regimes. The exact forms of the regimes or ‘scalings’ differ from one perspective to another, but they are always the direct consequence of dimensional analysis. They can be re-derived with a few lines of algebra. When we briefly considered mechanical variables, we observed that the kinematic components of a scaling could be combined in such a particular way as to yield a constant outcome. For instance, in the case of Taylor's regime, we saw that E ≃ (ρd3)(d/t)2, and since the energy E and density ρ were constant, this meant that d3(d/t)2 was also constant, despite being constructed from variables, d and t. Even if we restrict ourselves to kinematic variables, for any pair of mechanical parameters {Q1, Q2}, there is always a perspective that can be built to exploit this property. Kinematic variables and mechanical parameters can combine to form mechanical variables, but they can also combine in such a way as to provide a so-called ‘dimensionless number’, which provides a useful angle on the dynamics.
6.1 Constant variables
Let us consider the free fall as an example. In this context an object—like an apple—falls down to the ground. Galileo had discovered that before any significant friction can set in, the trajectory is characterized by a constant acceleration. Because this fact is now taught in pretty much all schools throughout the world, it is easy to overlook that a “constant acceleration” would have sounded like a complete oxymoron back then. Indeed “acceleration” is derived from the Latin word “accelerare”, meaning “to hasten, quicken”, whereas “constant” comes from “constantem”, meaning “standing firm”. What did Galileo mean? The distance traveled since the object was dropped increases as the square of the elapsed time, d = Kt2. If instead of following the distance over time we track the speed over time, we would find that it increases linearly, v ≃ d/t ≃ Kt. Now, if we track acceleration over time, we find that it is indeed independent of time, since a ≃ v/t ≃ K. All these considerations may sound trivial if we solely consider this one example of the free fall. However, the remarks made in this particular case can be generalized to any sort of regime.We just saw the famous example of constant acceleration, but there are “constant variables” of all kinds. If we agree to say “constant acceleration”, we should welcome this apparent contradiction as well. For any pair of mechanical quantities, for any regime, there is always a way to combine the initial variables to define a new variable, which will remain constant in the range of validity of the regime. The recipe to build such combination is simple. If we have two mechanical quantities Q1(x1, y1) and Q2(x2, y2), then assuming that the kinematics can be described by a variable length d and time t, we know from eqn (35) that Q1/Q2 ≃ dx1−x2ty1−y2. Since both Q1 and Q2 are constant parameters, the combination of variables κ ≡ dx1−x2ty1−y2 is also constant. The new kinematic variable κ is the constant variable of this regime.
In the case of Taylor's regime, the constant variable is thus κ ≡ d5t−2, which we called “explosivity”. As shown in Fig. 7, if we plot the values of this variable for the Trinity test, we find that it remains roughly constant over time. In fact, this “constant variable” is not only constant over time, but also for all sizes d, all speeds v ≃ d/t, etc. In Fig. 7, changing the horizontal axis does not change the shape of the plot. For the x-axis, we could actually choose any kinematic quantity as long as it is not a power of explosivity, which would just replicate the y-axis. The horizontal axis is largely arbitrary. The dimensions of the selected variable are inconsequential, as are its units. In contrast, for the vertical axis, the mechanics underlying this motion suggest a particular unit for explosivity: κ0 ≡ E/ρ, with [κ0] = [κ], the ratio of energy and density is an explosivity. So we can use the ratio of energy and density as our “natural” or “objective” unit of explosivity (a term we shall specify in Section 6.3). Another way to say this is that NEρ ≡ κ/κ0 ≡ ρd5/Et2 provides a ‘dimensionless number’ for the pair {E, ρ}. Indeed, 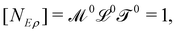 which is to say that the quantity NEρ has no dimensions. As we will see now these numbers are quite useful.
which is to say that the quantity NEρ has no dimensions. As we will see now these numbers are quite useful.
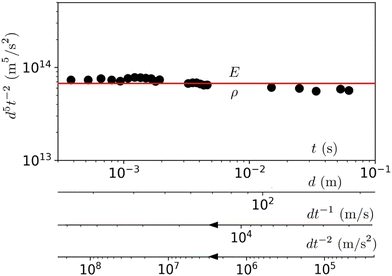 | ||
| Fig. 7 The “constant variable” of the Trinity explosion, i.e., “explosivity” κ ≡ d5t−2 plotted against the time since detonation t, the blast size d, the average front speed d/t, or the average front acceleration d/t2. Note that the axes for speed and acceleration run from right to left, since the speed and acceleration are initially greater. The horizontal red line is the “unit of explosivity” κ0 ≡ E/ρ. The data correspond to the set used by Taylor,8 and are shown as d(t) in Fig. 3a. | ||
6.2 Dimensionless numbers
In their simplest expression, dimensionless numbers provide a privileged perspective on each particular regime. Nevertheless, the term “dimensionless number” is also used to describe more complex combinations of kinematic and mechanical quantities, with no overall dimensions of space, time or mass. In this review, we will focus on simple dimensionless numbers. For any pair of mechanical quantities {Q1, Q2}, we assume an initial perspective with two variables, d and t, and we define the simple dimensionless number from eqn (35) as follows: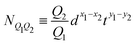 | (71) |
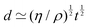 .101 The dimensionless number associated with this regime is the ‘Reynolds number’ Re, which is probably one of the most famous of its kind.178
.101 The dimensionless number associated with this regime is the ‘Reynolds number’ Re, which is probably one of the most famous of its kind.178
According to eqn (71) the dimensionless number associated with the pair {η, ρ} is:
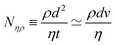 | (72) |
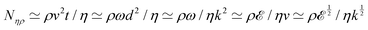 , etc. The form used depend on the choice of kinematic variables, and so on the context.
, etc. The form used depend on the choice of kinematic variables, and so on the context.
All too often, the Reynolds number is narrowly defined from one particular perspective. For instance, Wikipedia states that “The Reynolds number is the ratio of inertial forces to viscous forces”. In fact, because dimensionless numbers are dimensionless, they can be understood as ratios of any pair of quantities with the same dimensions. So the Reynolds number can indeed be defined as a ratio of (variable) forces, but it can also as a ratio of a number of other quantities. Here are some of the different ways to interpret the Reynolds number:
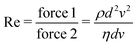 | (73) |
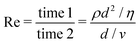 | (74) |
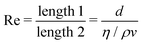 | (75) |
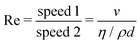 | (76) |
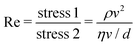 | (77) |
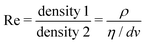 | (78) |
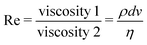 | (79) |
Among the various ways of interpreting the Reynolds number, one has a stronger standing than the others. As we saw in Section 4.4.1, the ratio of viscosity and density gives rise to ‘diffusivity’ (ν ≡ η/ρ, with 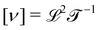 ). The most natural way to express the Reynolds number is as a ratio of diffusivities:
). The most natural way to express the Reynolds number is as a ratio of diffusivities:
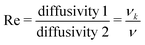 | (80) |
According to eqn (71), the dimensionless number associated with the pair {E, ρ} is:
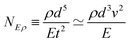 | (81) |
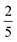 regime correspond to a constant Taylor–Sedov number, NEρ ≃ 1. Contrary to the Reynolds number, this dimensionless number is not naturally expressed as a ratio of diffusivities, but as a ratio of explosivities.
regime correspond to a constant Taylor–Sedov number, NEρ ≃ 1. Contrary to the Reynolds number, this dimensionless number is not naturally expressed as a ratio of diffusivities, but as a ratio of explosivities.
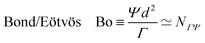 | (82) |
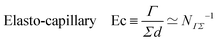 | (83) |
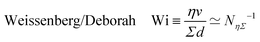 | (84) |
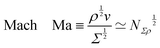 | (85) |
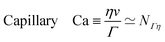 | (86) |
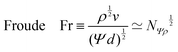 | (87) |
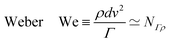 | (88) |
 .
.
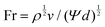 (Froude number),
(Froude number), 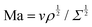 (Mach number), Wi = ηv/(Σd) (Weissenberg number), Ca = ηv/Γ (Capillary number), Re = ρdv/η (Reynolds number), We = ρv2d/Γ (Weber number), Ne = P/(ρv3d2) (Newton or Power number), Se = ρd3v2/E (Taylor–Sedov), C1 = F/(ρv2d2) (inertial drag coefficient), C2 = F/(ηvd) (viscous drag coefficient). All numbers shown are standard, except for the Taylor–Sedov, which we defined in this review. The notation ‘Se’ is here to imitate the traditional style, but as mentioned in the text we recommend the standard notation introduced in eqn (71). The structure constant α0 = S/Hv is usually defined in the special case where H = ℏ, v = c and S = S0 = kCe2, and in this case it is called the ‘fine structure constant.’
(Mach number), Wi = ηv/(Σd) (Weissenberg number), Ca = ηv/Γ (Capillary number), Re = ρdv/η (Reynolds number), We = ρv2d/Γ (Weber number), Ne = P/(ρv3d2) (Newton or Power number), Se = ρd3v2/E (Taylor–Sedov), C1 = F/(ρv2d2) (inertial drag coefficient), C2 = F/(ηvd) (viscous drag coefficient). All numbers shown are standard, except for the Taylor–Sedov, which we defined in this review. The notation ‘Se’ is here to imitate the traditional style, but as mentioned in the text we recommend the standard notation introduced in eqn (71). The structure constant α0 = S/Hv is usually defined in the special case where H = ℏ, v = c and S = S0 = kCe2, and in this case it is called the ‘fine structure constant.’
As we first observed with the Reynolds number, Re, dimensionless numbers are often referred to using the first two letters of the person most often associated with that number. This is true for the Bond, Mach, Froude or Weber numbers. This is almost true for the Weissenberg number, which uses Wi, instead of We, to avoid confusion with the Weber number. Some numbers, like elasto-capillary or capillary numbers imitate the style of one capitalized letter followed by a lower case letter, although no surnames are attached to it. This odd nomenclature can become quite unpractical when dealing with an increasing number of regimes, and can be the source of bitter priority disputes. It is one of the reasons why we advocate the more neutral and less reverent notation NQ1Q2, as defined in eqn (71). Note also that in some cases the name used for a particular dimensionless number depends on the choice of kinematic variables. For instance, one usually refers to the ‘Weissenberg number’ to describe Wi ≡ ηv/Σd, but to the ‘Deborah number’ for De ≡ η/Σt.180,181
As is apparent, for a given pair {Q1,Q2}, the traditional dimensionless numbers usually coincide with the definitions of NQ1Q2. However, in certain cases they differ by some power, which is a vestige of historical circumstances. Indeed, dimensionless numbers are defined modulo an overall power, meaning that if a combination of kinematic variables and mechanical parameters is dimensionless, then any power of that combination is also necessarily dimensionless. So for a dimensionless number N, Nα will also be dimensionless for all values of α. Underneath this trivial fact, if κ is a constant variable for a regime, the κα will also be constant.
The general definition provided in eqn (71) ensures that all dimensionless numbers constructed in that way are linear in the underlying mechanical quantities, unlike Ma or Fr. In addition, since both N and N−1 can be equally valid definitions, eqn (71) ensures that the impelling and impeding factors always occupy the same place from one number to another. For instance, historically, the ‘elasto-capillary number’ Ec = Γ/(Σd)14 has been defined in contrast to the Bond number, such that Ec < 1 means that d > ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ΓΣ, whereas Bo < 1 means that d <
ΓΣ, whereas Bo < 1 means that d < ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) ΓΨ. When we use traditional definitions, all these little discrepancies pile up and end up seriously obstructing the use of these dimensionless quantities. This is another reason to prefer the notation introduced in eqn (71).
ΓΨ. When we use traditional definitions, all these little discrepancies pile up and end up seriously obstructing the use of these dimensionless quantities. This is another reason to prefer the notation introduced in eqn (71).
6.3 Numbers and units
We have seen that given a pair of mechanical quantities we can derive its kinematic outcome and express it from a number of perspectives. One of these perspectives combines all variables and parameters into a dimensionless number. How is this last viewpoint special? How to use dimensionless numbers to further our understanding of the relationship between mechanics and kinematics?As shown in Fig. 8a, the visco-capillary regime has been observed in a number of situations where viscous fluids are driven by surface tension.107 As mentioned in Section 4.2, this regime is, for instance, observed for the pinching of viscous liquid bridges.124 In that case, t is the duration before pinch-off, and so the “actual time” runs from right to left. This pinching configuration is just one out of many possible set-ups exhibiting the regime {Γ, η}. Similar visco-capillary dynamics can also occur with rising bubbles pinching-off.182,183 In that case, the viscosity corresponds to that of the outer fluid. This regime has also been found in a number of slightly different configurations of droplet coalescence.184–188 For these examples, the neck between the drop grows, indicating the passage of time from left to right. As we will see shortly, the differences in setups actually have a marginal impact. The visco-capillary regime is observed during pinching, coalescence, and spreading of drops onto substrates.157 For spreading the size d is the radius of contact.
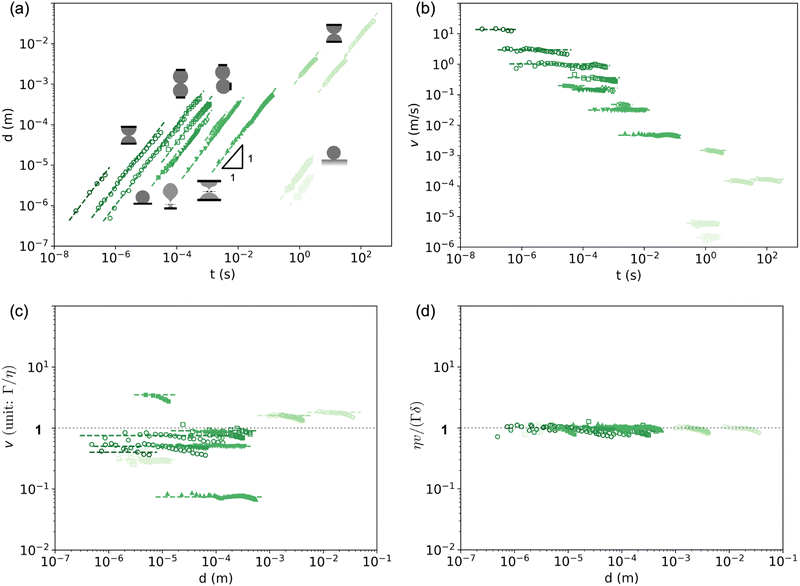 | ||
Fig. 8 Illustration of “rescaling” on a set of data exhibiting the visco-capillary regime {Γ, η}. The data include various configurations of pinching,124,182,183 coalescence184–188 and spreading.157 The data sets are available in the supplementary files of our recent review of this subject.107 (a) The visco-capillary regime seen from the “canonical perspective” of a length versus a time, {Γ, η}dt. The length d is the radius of the neck or contact area. The time t is the duration since contact for spreading and coalescence, and the duration before pinch-off for pinching. (b) The speed of the neck or of the edge of the contact area is plotted against time, {Γ, η}vt. Since the speed is the “constant variable” of this regime, the data sets fall on plateaus. (c) Upon using the mechanical ratio Γ/η as an “objective unit” of speed, the data sets are all found to be close to 1, ηv/Γ ≃ 1. The vertical axis can also be interpreted as giving the value of the simple dimensionless number of the regime, NΓη = Ca. The actual ordinates of each plateau give the value of the scaling constant δΓη in each experiment. (d) When the scaling constant is included in the definition of the dimensionless number 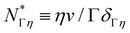 , all data sets naturally collapse on a single plateau given by , all data sets naturally collapse on a single plateau given by 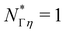 . Note that in (c) and (d), the size d is used as the horizontal axis, but any variable other than the speed v or powers of it could have been used (cf.Fig. 7). . Note that in (c) and (d), the size d is used as the horizontal axis, but any variable other than the speed v or powers of it could have been used (cf.Fig. 7). | ||
All lines in Fig. 8a have the same slope but the intercepts are different, because the values of viscosity and surface tension are different in each case, giving rise to visco-capillary speeds ranging from over 10 m s−1 for water with a bit of glycerol, in dark green,187 to slightly over 1 μm s−1 for colloid–polymer mixtures, in faint green.186 What we observe in Fig. 8a is how all these experiments look like from the perspective of length versus time, the “canonical perspective” of this review. But we are free to adopt a different perspective, particularly a perspective where one of the axis is the “constant variable”, which in this case is the speed v ≃ d/t. In Fig. 8b we have kept the time t as the other variable, but we could have chosen the size d, or anything we want other than speed (or powers of the speed). As we have observed already in Fig. 7 the second variable is largely irrelevant. The data sets would have still looked the same: horizontal lines. Since {Γ, η} → v ≃ Γ/η, in Fig. 8b, the different ordinates of the horizontal lines associated with each experiment reflect the different values of Γ/η. Experiments found higher on the plot correspond to higher values of surface-tension or lower values of viscosity.
In Fig. 8b, regardless of the fluid and set-up, the speeds are measured in meters per second. Of course, we could have used any arbitrary unit we want like cm/min or feet/hour. The choice of unit is completely subjective. We can—if we want—measure the speed of these pinching, coalescing and spreading droplets in relation to the length of our feet (ft), and to a fraction of the rotation period of our planet (hour). We are allowed to do this, but we should recognize how presumptuous we are to expect that the dynamics of droplets would be best described by such provincial choices. When, for instance, we say that the pinching of glycerol happens at a speed around 54 feet per hour, the number we get, 54, is due to two different things. First, and hopefully, it is related to an actual natural phenomenon, which was recorded sometime around the turn of the millennium.124 Second, the number 54 is connected to the choice of unit. If we choose different units, we get a different number, like 27.6 cm min−1. So these two concepts of units and numbers are obviously related. That is not really contentious. Whether it is 54 or 27.6, these numbers are a bit random, much like our choice of units.
Can we instead find a way to define more “objective” or “natural” units, less bound to our preferences? Units that would be set by the mechanics at play? Yes we can, this unit of speed is given by the ratio of surface tension and viscosity, v0 ≡ Γ/η. For each experiment we know the surface tension, and viscosity, so we can compute their ratio and use it directly as our unit. This unit is more objective than any of our choices because it is directly determined by the mechanical quantities dictating the dynamics.
In contrast to subjective units like meters per second, the value of the objective unit changes from one experiment to another. Once we have our objective units we can then plot all curves together, as shown in Fig. 8c. The curves now start to overlap, revealing their inherent similarity. With these objective units, all speeds are reasonably “close to 1”. Also note that plotting the speed v “in units of Γ/η” is the same as plotting the dimensionless number of the regime, which in this case is the capillary number Ca = NΓη. In Fig. 8c, it is often said that the dynamics from Fig. 8a or b have been ‘rescaled’.
Nevertheless, the overlap between the different curves in Fig. 8c is not perfect. For all curves we may say that NΓη ≃ 1, but not that NΓη = 1. Now that we are reaching the end of our exploration of scalings based on pairs of mechanical quantities, it is time to come back to the difference between the approximate equality, ‘≃’, which we have been relying on, and a stricter kind of equality, ‘=’.
A dimensional equation like [E/ρ] = [d]5[t]−2 is unimpeachable and exact regardless of the values of energy, density, size and time. This is why we use the sign ‘=’. When the brackets are dropped, E/ρ ≃ d5t−2, the approximate equality ‘≃’ is not so much an expression of imprecision as it is a requirement for adequacy between the chosen kinematic variables d and t, and the values of the mechanical quantities E and ρ. One may say that if the dynamics of an explosion blast are due to the interplay of energy and density, then E/ρ and d5t−2 must indeed have the same order of magnitude. More generally, assuming for simplicity that we have identified a variable length d and a variable time t to describe the kinematics, if we have Q1/Q2 ≃ dx1−x2ty1−y2, we can safely assume that the dynamics are impelled by Q1 and impeded by Q2. However, the agreement between the two sides may not be exact. We can write this from the “canonical perspective” of length versus time:
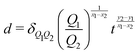 | (89) |
The scaling constant δQ1Q2 of a regime is a black box, containing all sorts of influences beyond that of the mechanical pair underlying the dynamics. For instance, consider Archimedes’ simple length 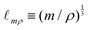 . Suppose we are dealing with a sphere of diameter d, so its volume is Ω = (π/6)d3, and its mass is m = ρΩ, and so
. Suppose we are dealing with a sphere of diameter d, so its volume is Ω = (π/6)d3, and its mass is m = ρΩ, and so 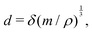 with
with 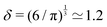 . In this case, the scaling constant δ includes shape effects. Its value would be different if we were dealing with a cube or a pyramid, or some more complicated figure. The description of shapes may require more than a single length (height, width, etc.) and would then involve more than a pair of mechanical quantities.
. In this case, the scaling constant δ includes shape effects. Its value would be different if we were dealing with a cube or a pyramid, or some more complicated figure. The description of shapes may require more than a single length (height, width, etc.) and would then involve more than a pair of mechanical quantities.
When we discussed the Hooke–Rayleigh time τmΓ, we observed a similar effect in the time rather than the space dimension. The period of oscillation of a spring and mass system is 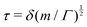 , with δ = 2π ≃ 6.3. Such correction can very well be absorbed by redefining the variables. For instance here, we can use the angular frequency ω ≡ 2π/τ to reach
, with δ = 2π ≃ 6.3. Such correction can very well be absorbed by redefining the variables. For instance here, we can use the angular frequency ω ≡ 2π/τ to reach 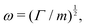 where there is no more correction factor.
where there is no more correction factor.
In some cases, as in previous two examples, the scaling constant can be disposed off by an appropriate redefinition of the variables. In other cases, a redefinition of the mechanical quantities may also be helpful. For instance, if the standard way for measuring the energy (or ‘yield’) of an explosion involved fitting the dynamics of the blast by a power law 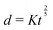 and then to set E ≡ ρK5, then obviously we would have
and then to set E ≡ ρK5, then obviously we would have 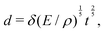 with δ = 1, as long as our measurement of the air density ρ is correct. Whenever we encounter a regime with a scaling constant that conveniently reduces to one, it is probably because the mechanical quantities of this regime are actually defined in that context. However, this is rarely the case, since mechanical quantities are free to interact with so many partners, like all those given in Table 1. The difficulties in understanding the value of the scaling constant δ then lie in the fact that δ usually connects a given regime to other manifestations of its mechanical factors, beyond the range of validity of the regime. For instance, for explosions, the energy may be defined from measurements of the final blast radius
with δ = 1, as long as our measurement of the air density ρ is correct. Whenever we encounter a regime with a scaling constant that conveniently reduces to one, it is probably because the mechanical quantities of this regime are actually defined in that context. However, this is rarely the case, since mechanical quantities are free to interact with so many partners, like all those given in Table 1. The difficulties in understanding the value of the scaling constant δ then lie in the fact that δ usually connects a given regime to other manifestations of its mechanical factors, beyond the range of validity of the regime. For instance, for explosions, the energy may be defined from measurements of the final blast radius ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , as
, as 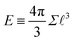 , where Σ is the bulk modulus of the air (according to eqn (6)), or from the initial speed u, as
, where Σ is the bulk modulus of the air (according to eqn (6)), or from the initial speed u, as 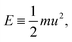 where m is the ejected mass (according to eqn (29)). The numerical factors used in these definitions (
where m is the ejected mass (according to eqn (29)). The numerical factors used in these definitions ( , or
, or 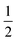 ) get carried over from one equation to another and end up pilling up in the scaling constant δ.
) get carried over from one equation to another and end up pilling up in the scaling constant δ.
In the case of the pinching, coalescing and spreading fluids in Fig. 8c, the ordinates of the plateaus in each data set give an average value of the scaling constant δΓη for each experiment. We refer the reader to our recent meta-analysis of this subject, where values of the constant are listed for all experiments shown in Fig. 8c.107 The constants are all reasonably “close to 1”, but they are influenced by the geometric details of each set-up. For instance, in the case of the pinching of a liquid thread between two plates, theoretical analyses proposed slightly different values of δΓη depending on subtle differences: δΓη = 0.1666,124δΓη = 0.0709,128δΓη = 0.0304,189,190 and δΓη = 0.0108.190 The experiment on the pinching of glycerol reproduced in Fig. 8 (▲) seemed to favor Papageorgiou's value.124
Ultimately, one may decide to include the scaling constants into the definition of the objective unit or constant variable, that is, into the definition of the simple dimensionless number, in order to achieve a more satisfying overlap of the data, as shown in Fig. 8d for the visco-capillary regime. If the scaling constant is defined from the canonical perspective in eqn (89), then amending eqn (71) we may write:
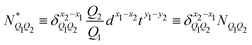 | (90) |
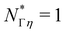 . Systematic deviations from this plateau may indicate the growing effect of mechanical quantities beyond the initial pair, for instance, the effect of the density of the fluid ρ, or the ‘Laplace force’ F ≃ Γ
. Systematic deviations from this plateau may indicate the growing effect of mechanical quantities beyond the initial pair, for instance, the effect of the density of the fluid ρ, or the ‘Laplace force’ F ≃ Γ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) , when approaching the size
, when approaching the size ![[small script l]](https://www.rsc.org/images/entities/char_e146.gif) of the whole drop.107 Since no regime extends forever, such deviations are bound to happen; however, this topic is beyond the scope of this review.
of the whole drop.107 Since no regime extends forever, such deviations are bound to happen; however, this topic is beyond the scope of this review.
7 Conclusion
With Archimedes, Newton, Taylor, and all those who sought to explore the mechanical underpinning of space and time, this review has demonstrated the parsimonious efficiency of dimensional analysis. Whether we want to explain the magnitude of a particular volume, acceleration, or the more exotic motion of an explosion blast, the mechanical philosophy suggests a bold idea: to invoke an extra dimension, the dimension of mass![[scr M, script letter M]](https://www.rsc.org/images/entities/char_e145.gif) , beyond the visible dimensions of space,
, beyond the visible dimensions of space,  and time,
and time, 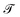 . The scope of this simple idea is immense.
. The scope of this simple idea is immense.
What we see as motion, size, or duration is understood as a form of shadow, cast on a plane by a much broader play. The players are the mechanical quantities, inferred by generations of researchers from their effects. The table of mechanical quantities that we built for this review provides an initial map, drawn from centuries of exploration of this mechanical pantheon. We hope that this table will incite historians of science to trace back the steps of past thinkers from one spot on the map to another, to define pressure, energy, power, action, etc. Why did so many great minds sailed these waters? How does this mechanical chart relate to the world we live in? We wish to do our part in answering these questions, and this review is a first step. In this review, we have shown what emerges from the interplay of pairs of mechanical quantities. This type of pair interaction is the elementary building block of a dimensional analysis of mechanics, to which we can refer to more succinctly as dimensional mechanics.
As we observed, numerous experiments have shown that a range of complex motions can be cast as single lines on a logarithmic scale. These straight regimes are drawn on a plane of space and time, yet we find that they can be more deeply understood as a “reflection”, or “projection”, or “shadow” of phenomena occurring at a higher dimension. What is going on is a “struggle”, or “balance”, between “competing” mechanical factors. Dimensional mechanics help us in formalizing all these colloquialisms and help us find the “causes” behind these scalings. These causes are embodied by the mechanical quantities. Any pair of mechanical quantities is associated with a regime, represented as a line in the kinematic plane. The slope of this line is given by the dimensions of the mechanical ratio. Because the standard mechanical quantities all share a dimension of mass ![[scr M, script letter M]](https://www.rsc.org/images/entities/char_e145.gif) , such mass disappears from their ratio:
, such mass disappears from their ratio:
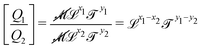 | (91) |
The type of scaling observed in a particular context depends on the dimensions of the underlying mechanical parameters. Some experiments may evidence characteristic lengths, or times, or speeds, but others may record more intricate relationships between space and time. For instance, from the “canonical perspective” of a variable length measured relative to a variable time one may see scalings of the form d ∼ tα, with 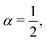 or
or 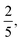 or
or 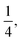 or
or 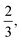 or
or 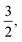 etc. These exponents are not fundamentally stranger than the more traditional α = 1 of uniform motion, or α = 2 of uniformly accelerated motions. We just had less time to get used to them. In this review, we have tried to find a diverse array of examples from different fields, but the lists of scalings that we compiled represent only a fraction of what could be gathered from a more thorough investigation. We invite readers to participate in this encyclopedic enterprise, and we will welcome any correspondence to that end.
etc. These exponents are not fundamentally stranger than the more traditional α = 1 of uniform motion, or α = 2 of uniformly accelerated motions. We just had less time to get used to them. In this review, we have tried to find a diverse array of examples from different fields, but the lists of scalings that we compiled represent only a fraction of what could be gathered from a more thorough investigation. We invite readers to participate in this encyclopedic enterprise, and we will welcome any correspondence to that end.
Throughout this review, we have assumed that the “relevant” mechanical quantities were known in each particular context, and so from these parameters the regimes could be derived through dimensional analysis. We understood the “play” so that we could make sense of its “shadow”, as observed from different perspectives. For instance, in the case of the Trinity explosion, knowing the energy E of the bomb and the density ρ of the air to be the relevant parameters, Taylor could derive that 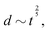 or
or 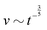 . We have shown how a knowledge of mechanics implies the kinematics. But how is such knowledge gained in the first place? Mechanics implies kinematics, but the reverse is not so simple. Assuming a “canonical perspective”, we can symbolize this as follows:
. We have shown how a knowledge of mechanics implies the kinematics. But how is such knowledge gained in the first place? Mechanics implies kinematics, but the reverse is not so simple. Assuming a “canonical perspective”, we can symbolize this as follows:
Mechanical  Kinematic {Q1, Q2} Kinematic {Q1, Q2}  d ≃ Ktα d ≃ Ktα | (92) |
Kinematics have a lower dimension than mechanics. What we perceive as motion is only a projection of what goes on, and information is lost in this shadow play. We might see a dog or a duck on the screen, but it may actually be the hand of the puppeteer. For a given pair of mechanical quantities, Q1(x1, y1) and Q2(x2, y2), the resulting regime is d ≃ Ktα, with α = (y2 − y1)/(x1 − x2). Only the relative dimensions of the mechanical quantities matter, i.e., the differences y2 − y1 and x1 − x2. Thus, a given exponent α may actually stem from an array of mechanical pairs. For instance, we saw diffusive regimes due to {E, Λ}, {Σ, χ}, {Γ, φ}, {F, ρ}, {F, η}, or {η, ρ}. Reviewing the table of mechanical quantities will confirm that these pairs have the same relative placement. So, if we do observe 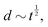 how can we know which mechanical pair is behind this motion? We cannot, that is, if we only observe a single regime…
how can we know which mechanical pair is behind this motion? We cannot, that is, if we only observe a single regime…
On paper, power laws such as d ≃ Ktα are ‘self-similar’;6 they seemingly extend to arbitrarily small or large scales. In practice, no single regime extends indefinitely. For instance, Taylor showed that the Trinity explosion of July 1945 in New Mexico followed 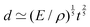 . As shown in Fig. 3a, the data do support this model, but only for the selected time range, from a fraction of a millisecond to a fraction of a second after detonation. Taylor knew very well that this regime was only transient.7,8 If extended indefinitely in the future, the blast would have reached New York by now! And if extended to the instant of detonation, this regime would predict a diverging front speed, since
. As shown in Fig. 3a, the data do support this model, but only for the selected time range, from a fraction of a millisecond to a fraction of a second after detonation. Taylor knew very well that this regime was only transient.7,8 If extended indefinitely in the future, the blast would have reached New York by now! And if extended to the instant of detonation, this regime would predict a diverging front speed, since 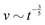 . Evidently, the balance between energy and density cannot account for the whole dynamics of the explosion. We actually know what we should expect. At small time scales, the mass of the bomb will have an effect and we would get a constant initial speed from {E, m}, according to eqn (29). At a large scale, we would reach the final blast radius set by the ambient pressure, {E, Σ}, according to eqn (6). Dynamics are never truly ‘self-similar’, because the complete play always involves more than two players.
. Evidently, the balance between energy and density cannot account for the whole dynamics of the explosion. We actually know what we should expect. At small time scales, the mass of the bomb will have an effect and we would get a constant initial speed from {E, m}, according to eqn (29). At a large scale, we would reach the final blast radius set by the ambient pressure, {E, Σ}, according to eqn (6). Dynamics are never truly ‘self-similar’, because the complete play always involves more than two players.
We have learned much by focusing on pairs of mechanical quantities, and we would be right to expect that a deeper understanding of dimensional mechanics may emerge by progressively enlarging our set of mechanical parameters. We have already started exploring the effects of trios and quartets, and they greatly extend the reach of dimensional mechanics. Our https://www.youtube.com/playlist?list=PLbMiQs7eX-bbNTc-7HwdWzohUs8yPw300 on explosions documents this progression for this particular example, and starting with https://youtu.be/1WP7k1VUf3Q?si=Muusfb3EDpNHB2I2, the series on mechanics generalizes our findings. We are planning to summarize these investigations in upcoming publications, but we can already mention a few salient points, which may resonate with some of the questions that this review might have raised.
When considering three mechanical quantities, two cases should be distinguished: whether the three quantities are aligned or not. When the quantities are aligned they lead to three parallel regimes. For instance, in the special case where the quantities align on the same line in Table 1, this configuration can be used to rationalize shapes with distinct dimensions along different directions, and more generally to explore the mechanical basis of geometry. Readers wishing to delve deeper may investigate situations characterized by trios such as {E, Σ, Ψ}, which may be useful for cratering,64 or {E, Γ, Ψ}, which may be useful for drop impact.88 When considering trios of mechanical quantities that are not aligned, the three regimes associated with the three pairs of the trio intersect at a single point, a special event where dynamics take a turn, and which can serve as the locus of fully objective units. For instance, looking back at the dispersion relation in Fig. 5, we can easily envision that the regimes of waves {Ψ, ρ} and ripples {Γ, ρ} will intersect at a wavenumber given by the inverse of the capillary length implied by the pair {Γ, ρ}. The coordinates of the point of intersection are fully characterized by the trio {Ψ, Γ, ρ}. Another example involves considering the trio {Γ, ρ, η}, to capture the combined effects of inertia and viscosity on capillary flows. We discussed this case in a recent publication.107
The key insight brought by considering a fourth quantity is even more intriguing. Quartets of mechanical quantities include four connected trios associated with intersections. The six associated regimes connect these turning points to one another, enabling, for instance, the tracking of the sequence of events in an explosion from the initial dynamics driven by the energy of the bomb E and impaired by the ejected mass m, to the gradual transformation of the blast into a simple sound wave, depending solely on the density ρ and pressure Σ of the ambient air. As shown in our https://www.youtube.com/playlist?list=PLbMiQs7eX-bbNTc-7HwdWzohUs8yPw300, a quartet such as {E, m, ρ, Σ} also provides a new kind of dimensionless number, depending solely on the constant mechanical parameters. In this particular instance, this number would be 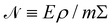 (modulo an overall power). Such dimensionless numbers enable the distinction between two broad classes of dynamics: detonations
(modulo an overall power). Such dimensionless numbers enable the distinction between two broad classes of dynamics: detonations 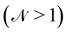 and deflagrations
and deflagrations 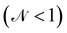 . More profoundly, such dimensionless numbers provides an “objective number base”, to use instead of 10 in logarithmic representations. Base 10 is an international convention for numbers, just like kilograms, meters, and seconds are international conventions for mass, length and time. If dimensional analysis is practiced beyond the mechanical duos we have been focusing on here, these four conventions are replaced with standards set by the physics of the situation under consideration.
. More profoundly, such dimensionless numbers provides an “objective number base”, to use instead of 10 in logarithmic representations. Base 10 is an international convention for numbers, just like kilograms, meters, and seconds are international conventions for mass, length and time. If dimensional analysis is practiced beyond the mechanical duos we have been focusing on here, these four conventions are replaced with standards set by the physics of the situation under consideration.
Although the role of the mass dimension and the associated mechanical quantities is initially to “explain” the kinematics, what dimensional analysis reveals is that the “physics” of a situation also dictate the appropriate ways to represent what is observed. The kind of rescaling displayed in Fig. 8 can be brought to a whole new level by enlarging the experimental range, including the effects of more than two mechanical quantities.
Dimensional analysis is so much more than a trick to circumvent the more “rigorous” use of differential equations. Dimensions provide the fundamental structure of physics, and the dimensions of mechanics are its most robust backbone.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
VS thanks UIC and UChicago students who enrolled in his many courses and provided student perspectives in discussions on dimensions and dimensional analysis, as well as feedback on the supplementary videos. VS also wishes to thank his Soft Matter ODES-lab students. VS acknowledges Matt Tirrell, Juan de Pablo, Paul Nealey, and the Pritzker School of Molecular Engineering at UChicago for financial support during his sabbatical. MH thanks DGAPA-UNAM for funding his sabbatical, which initiated this review. VS, MH and MAF acknowledge the joyful atmosphere of the Ladoux-Mège lab, the stimulating haven that made this project possible. We owe a great debt to our predecessors and mentors, and many in the fluid mechanics, soft matter, and biophysics communities for inspiration and exemplary applications of dimensional analysis.Notes and references
- J. B. J. Fourier, Théorie de la chaleur, 1822 Search PubMed.
- A. K. T. Assis, K. Reich and K. H. Wiederkehr, 21st Century, 2002, 15, 40–48 Search PubMed.
- J. C. Maxwell, A treatise on electricity and magnetism, Clarendon Press, 1873, vol. 1 Search PubMed.
- D. J. Mitchell, Studies in History and Philosophy of Science Part B: Studies in History and Philosophy of Modern Physics, 2017, 58, 63–79 CrossRef.
- E. O. Macagno, J. Franklin Institute, 1971, 292, 391–402 CrossRef.
- G. I. Barenblatt, Scaling, Cambridge University Press, 2003, vol. 34 Search PubMed.
- G. I. Taylor, Proc. R. Soc. London, Ser. A, 1950, 201, 175–186 CAS.
- G. I. Taylor, Proc. R. Soc. London, Ser. A, 1950, 201, 175–186 CAS.
- E. Dussan, Annu. Rev. Fluid Mech., 1979, 11, 371–400 CrossRef.
- P.-G. de Gennes, Rev. Mod. Phys., 1985, 57, 827 CrossRef CAS.
- L. Leger and J. Joanny, Rep. Prog. Phys., 1992, 55, 431 CrossRef CAS.
- S. Middleman, Modeling axisymmetric flows: dynamics of films, jets, and drops, Academic Press, 1995 Search PubMed.
- A. Oron, S. H. Davis and S. G. Bankoff, Rev. Mod. Phys., 1997, 69, 931 CrossRef CAS.
- G. H. McKinley, Rheol. Rev., 2005, 1, 1–48 Search PubMed.
- V. Starov, M. Velarde and C. Radke, Wetting and Spreading Dynamics, CRC Press, 2007 Search PubMed.
- S. Kalliadasis and U. Thiele, Thin films of soft matter, Springer, 2007, vol. 1 Search PubMed.
- R. V. Craster and O. K. Matar, Rev. Mod. Phys., 2009, 81, 1131 CrossRef CAS.
- D. Bonn, J. Eggers, J. Indekeu, J. Meunier and E. Rolley, Rev. Mod. Phys., 2009, 81, 739 CrossRef CAS.
- M. N. Popescu, G. Oshanin, S. Dietrich and A. Cazabat, J. Phys.: Condens. Matter, 2012, 24, 243102 CrossRef CAS PubMed.
- P.-G. de Gennes, F. Brochard-Wyart and D. Quéré, Capillarity and wetting phenomena: drops, bubbles, pearls, waves, Springer Science & Business Media, 2013 Search PubMed.
- J. H. Snoeijer and B. Andreotti, Annu. Rev. Fluid Mech., 2013, 45, 269–292 CrossRef.
- G. Lu, X.-D. Wang and Y.-Y. Duan, Adv. Colloid Interface Sci., 2016, 236, 43–62 CrossRef CAS PubMed.
- J. Bico, É. Reyssat and B. Roman, Annu. Rev. Fluid Mech., 2018, 50, 629–659 CrossRef.
- B. Andreotti and J. H. Snoeijer, Annu. Rev. Fluid Mech., 2020, 52, 285–308 CrossRef.
- D. Lohse and X. Zhang, et al. , Rev. Mod. Phys., 2015, 87, 981 CrossRef CAS.
- D. Lohse and X. Zhang, Nat. Rev. Phys., 2020, 2, 426–443 CrossRef.
- H. A. Bethe, K. Fuchs, J. O. Hirschfelder, J. L. Magee and R. V. Neumann, Blast wave, Los alamos national lab nm technical report, 1958.
- S. Glasstone, P. J. Dolan, et al., The effects of nuclear weapons, US Department of Defense, 1977, vol. 50 Search PubMed.
- L. I. Sedov, Similarity and dimensional methods in mechanics, CRC Press, 1993 Search PubMed.
- P. O. Krehl, History of shock waves, explosions and impact: a chronological and biographical reference, Springer Science & Business Media, 2008 Search PubMed.
- P. S. Westine, F. Dodge and W. Baker, Similarity methods in engineering dynamics: theory and practice of scale modeling, Elsevier, 2012 Search PubMed.
- G. F. Kinney and K. J. Graham, Explosive shocks in air, Springer Science & Business Media, 2013 Search PubMed.
- P. Sachdev, Shock waves & explosions, Chapman and Hall/CRC, 2016 Search PubMed.
- D. Thompson, On Growth and Form, Namaskar Books, 1917 Search PubMed.
- T. Mitchison and L. Cramer, Cell, 1996, 84, 371–379 CrossRef CAS PubMed.
- W. Alt, Dynamics of cell and tissue motion, Springer Science & Business Media, 1997 Search PubMed.
- M. P. Sheetz, Nat. Rev. Mol. Cell Biol., 2001, 2, 392–396 CrossRef CAS PubMed.
- K. Roberts, B. Alberts, A. Johnson, P. Walter and T. Hunt, Molecular biology of the cell, Garland Science, New York, 2002 Search PubMed.
- T. Lecuit and P.-F. Lenne, Nat. Rev. Mol. Cell Biol., 2007, 8, 633–644 CrossRef CAS PubMed.
- C. Le Clainche and M.-F. Carlier, Physiol. Rev., 2008, 88, 489–513 CrossRef CAS PubMed.
- T. D. Pollard and J. A. Cooper, Science, 2009, 326, 1208–1212 CrossRef CAS PubMed.
- R. Phillips, J. Kondev, J. Theriot and H. Garcia, Physical biology of the cell, Garland Science, 2012 Search PubMed.
- M. C. Marchetti, J.-F. Joanny, S. Ramaswamy, T. B. Liverpool, J. Prost, M. Rao and R. A. Simha, Rev. Mod. Phys., 2013, 85, 1143 CrossRef CAS.
- U. S. Schwarz and S. A. Safran, Rev. Mod. Phys., 2013, 85, 1327 CrossRef CAS.
- E. Buckingham, Phys. Rev., 1914, 4, 345 CrossRef.
- L. Rayleigh, Nature, 1915, 95, 202–203 CrossRef.
- P. W. Bridgman, Dimensional analysis, Yale University Press, 1922 Search PubMed.
- G. I. Barenblatt, Scaling, self-similarity, and intermediate asymptotics: dimensional analysis and intermediate asymptotics, Cambridge University Press, 1996 Search PubMed.
- J. G. Santiago, A First Course in Dimensional Analysis: Simplifying Complex Phenomena Using Physical Insight, Mit Press, 2019 Search PubMed.
- A. Mottana, Philosophia Scientiæ. Travaux d'histoire et de philosophie des sciences, 2017, 165–179 Search PubMed.
- B. Hopkinson, The National Archives, Kew, UK, 1915, 11 Search PubMed.
- C. Cranz, Lehrbuch der Ballistik, II Band, 1926 Search PubMed.
- R. G. Sachs, The dependence of blast on ambient pressure and temperature, Army ballistic research lab aberdeen technical report, 1944.
- T. Wei and M. J. Hargather, Shock Waves, 2021, 31, 231–238 CrossRef.
- V. Fokin, Atomic Energy, 2000, 89, 924–927 CrossRef CAS.
- S. P. Reynolds, Annu. Rev. Astron. Astrophys., 2008, 46, 89–126 CrossRef CAS.
- A. I. Asvarov, Astron. Astrophys., 2014, 561, A70 CrossRef.
- (a) A. Einstein, Ann. Phys., 1905, 17, 549 CrossRef CAS; (b) A. Einstein, Ann. Phys., 1905, 17, 132 CrossRef CAS.
- R. Phillips and S. R. Quake, Phys. Today, 2006, 59, 38–43 CrossRef CAS.
- M. Guo, A. F. Pegoraro, A. Mao, E. H. Zhou, P. R. Arany, Y. Han, D. T. Burnette, M. H. Jensen, K. E. Kasza and J. R. Moore, et al. , Proc. Natl. Acad. Sci. U. S. A., 2017, 114, E8618–E8627 CAS.
- K. Xie, Y. Yang and H. Jiang, Biophys. J., 2018, 114, 675–687 CrossRef CAS PubMed.
- R. M. Adar and S. A. Safran, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 5604–5609 CrossRef CAS PubMed.
- K. Housen, R. Schmidt and K. Holsapple, J. Geophys. Res.: Solid Earth, 1983, 88, 2485–2499 CrossRef.
- K. A. Holsapple, Annu. Rev. Earth Planet. Sci., 1993, 21, 333–373 CrossRef.
- H. Katsuragi, et al., Physics of soft impact and cratering, Springer, 2016 Search PubMed.
- J. Uehara, M. Ambroso, R. Ojha and D. J. Durian, Phys. Rev. Lett., 2003, 90, 194301 CrossRef CAS PubMed.
- H. Takita and I. Sumita, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2013, 88, 022203 CrossRef PubMed.
- A. Benusiglio, D. Quéré and C. Clanet, J. Fluid Mech., 2014, 752, 123–139 CrossRef CAS.
- V. Sharma, K. Park and M. Srinivasarao, Mater. Sci. Eng., R, 2009, 65, 1–38 CrossRef.
- J. B. Perrin, Nobel Lectures, 1926, 1, 2 Search PubMed.
- C. Clanet, C. Béguin, D. Richard and D. Quéré, J. Fluid Mech., 2004, 517, 199–208 CrossRef.
- A. R. Choudhuri, Astrophysics for physicists, Cambridge University Press, 2010 Search PubMed.
- R. Kippenhahn, A. Weigert and A. Weiss, Stellar structure and evolution, Springer, 1990, vol. 192 Search PubMed.
- C. Frontali, Phys. Educ., 2013, 48, 484 CrossRef.
- M.-A. Fardin and M. Hautefeuille, Am. J. Phys., 2022, 90, 914–920 CrossRef.
- C. Creton and M. Ciccotti, Rep. Prog. Phys., 2016, 79, 046601 CrossRef PubMed.
- E. R. Jerison, Y. Xu, L. A. Wilen and E. R. Dufresne, Phys. Rev. Lett., 2011, 106, 186103 CrossRef PubMed.
- M. Coux and J. M. Kolinski, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 32285–32292 CrossRef CAS PubMed.
- A. Carré, J.-C. Gastel and M. E. Shanahan, Nature, 1996, 379, 432–434 CrossRef.
- A. Sheludko, Adv. Colloid Interface Sci., 1967, 1, 391–464 CrossRef CAS.
- A. Nikolov and D. Wasan, Adv. Colloid Interface Sci., 2014, 206, 207–221 CrossRef CAS PubMed.
- J. N. Israelachvili, Intermolecular and surface forces, Academic Press, 2015 Search PubMed.
- V. M. Starov and M. G. Velarde, Wetting and spreading dynamics, CRC Press, 2019 Search PubMed.
- Y. Zhang and V. Sharma, Langmuir, 2018, 34, 1208–1217 CrossRef CAS PubMed.
- S. Yilixiati, E. Wojcik, Y. Zhang and V. Sharma, Mol. Syst. Des. Eng., 2019, 4, 626–638 RSC.
- T. Bennett and D. Poulikakos, J. Mater. Sci., 1993, 28, 963–970 CrossRef.
- J. Eggers, M. A. Fontelos, C. Josserand and S. Zaleski, Phys. Fluids, 2010, 22, 062101 CrossRef.
- N. Laan, K. G. de Bruin, D. Bartolo, C. Josserand and D. Bonn, Phys. Rev. Appl., 2014, 2, 044018 CrossRef.
- L. D. Landau, E. M. Lifshitz, et al., Theory of elasticity, Pergamon Press, Oxford New York, 1986, vol. 7 Search PubMed.
- P. A. Dirac, Nature, 1937, 139, 323 CrossRef.
- D. J. Griffiths and D. F. Schroeter, Introduction to quantum mechanics, Cambridge University Press, 2018 Search PubMed.
- M. Muthukumar, Physics of Charged Macromolecules, Cambridge University Press, 2023 Search PubMed.
- R. Robinson and R. Stokes, Electrolyte Solutions, Dover Publications, Incorporated, 2nd edn, 2012 Search PubMed.
- P. C. Clemmow, Electrodynamics of particles and plasmas, CRC Press, 2018 Search PubMed.
- H. Kragh, Archive History Exact Sci., 2003, 57, 395–431 CrossRef.
- L. de Broglie, Ann. Phys., 1925, 22–128 Search PubMed.
- K. Trachenko and V. Brazhkin, Sci. Adv., 2020, 6, eaba3747 CrossRef CAS PubMed.
- D. Cuvelier, M. Théry, Y.-S. Chu, S. Dufour, J.-P. Thiéry, M. Bornens, P. Nassoy and L. Mahadevan, Curr. Biol., 2007, 17, 694–699 CrossRef CAS PubMed.
- M. Fardin, O. Rossier, P. Rangamani, P. Avigan, N. Gauthier, W. Vonnegut, A. Mathur, J. Hone, R. Iyengar and M. Sheetz, Soft Matter, 2010, 6, 4788–4799 RSC.
- G. Stokes, Trans. Camb. Philos. Soc., 1850, 9, 106 Search PubMed.
- L. Landau and E. Lifshitz, Fluid Mechanics, Pergamon, 1959, vol. 61 Search PubMed.
- W. B. Russel, W. Russel, D. A. Saville and W. R. Schowalter, Colloidal dispersions, Cambridge University Press, 1991 Search PubMed.
- P.-G. de Gennes and J. Prost, The physics of liquid crystals, Oxford University Press, 1993 Search PubMed.
- L. Rayleigh, et al. , Proc. R. Soc. London, 1879, 29, 71–97 CrossRef.
- D. Richard, C. Clanet and D. Quéré, Nature, 2002, 417, 811 CrossRef CAS PubMed.
- J. Eggers, Rev. Mod. Phys., 1997, 69, 865 CrossRef CAS.
- M. A. Fardin, M. Hautefeuille and V. Sharma, Soft Matter, 2022, 18, 3291–3303 RSC.
- G. Shaviv, New Astron. Rev., 2008, 51, 803–813 CrossRef.
- C. Kittel, Introduction to solid state physics, John Wiley & Sons, Inc, 2005 Search PubMed.
- M. Z. Bazant, K. Thornton and A. Ajdari, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2004, 70, 021506 CrossRef PubMed.
- R. G. Larson, The structure and rheology of complex fluids, Oxford University Press, New York, 1999, vol. 150 Search PubMed.
- R. B. Bird, R. C. Armstrong and O. Hassager, Dynamics of polymeric liquids, Fluid mechanics, John Wiley and Sons Inc., New York, NY, 1987, vol. 1 Search PubMed.
- M. Fardin, C. Perge and N. Taberlet, Soft Matter, 2014, 10, 3523–3535 RSC.
- M. Doi, S. F. Edwards and S. F. Edwards, The theory of polymer dynamics, Oxford University Press, 1988, vol. 73 Search PubMed.
- T. Divoux, M. A. Fardin, S. Manneville and S. Lerouge, Annu. Rev. Fluid Mech., 2016, 48, 81–103 CrossRef.
- C. Clarke and B. Carswell, Principles of astrophysical fluid dynamics, Cambridge University Press, 2007 Search PubMed.
- G. I. Ogilvie and M. R. Proctor, J. Fluid Mech., 2003, 476, 389–409 CrossRef.
- J. Dinic and V. Sharma, Macromolecules, 2020, 53, 4821–4835 CrossRef CAS.
- B. J. Hamrock, B. J. Schmid and B. O. Jacobson, Fundamentals of fluid film lubrication, CRC Press, 2004, vol. 169 Search PubMed.
- L. G. Leal, Advanced transport phenomena: fluid mechanics and convective transport processes, Cambridge University Press, 2007, vol. 7 Search PubMed.
- C. Pérez-González, R. Alert, C. Blanch-Mercader, M. Gómez-González, T. Kolodziej, E. Bazellieres, J. Casademunt and X. Trepat, Nat. Phys., 2019, 15, 79–88 Search PubMed.
- A. Puliafito, L. Hufnagel, P. Neveu, S. Streichan, A. Sigal, D. K. Fygenson and B. I. Shraiman, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 739–744 CrossRef CAS PubMed.
- N. J. Mlot, C. A. Tovey and D. L. Hu, Proc. Natl. Acad. Sci. U. S. A., 2011, 108, 7669–7673 CrossRef CAS PubMed.
- G. H. McKinley and A. Tripathi, J. Rheol., 2000, 44, 653–670 CrossRef CAS.
- F. Melo, J. Joanny and S. Fauve, Phys. Rev. Lett., 1989, 63, 1958 CrossRef CAS PubMed.
- S. Douezan, K. Guevorkian, R. Naouar, S. Dufour, D. Cuvelier and F. Brochard-Wyart, Proc. Natl. Acad. Sci. U. S. A., 2011, 108, 7315–7320 CrossRef CAS PubMed.
- S. Chandrasekhar, Hydrodynamic and hydromagnetic stability, Courier Corporation, 2013 Search PubMed.
- D. T. Papageorgiou, J. Fluid Mech., 1995, 301, 109–132 CrossRef.
- D. A. Edwards, H. Brenner and D. T. Wasan, Interfacial transport processes and rheology, Butterworth-Heinemann, 1991 Search PubMed.
- H. Manikantan and T. M. Squires, J. Fluid Mech., 2020, 892, P1 CrossRef CAS PubMed.
- A. D. Nikolov, D. T. Wasan and P. Wu, Curr. Opin. Colloid Interface Sci., 2021, 51, 101387 CrossRef CAS.
- T. Svedberg, Nobel Lectures, 1966 Search PubMed.
- S. Claesson and K. O. Pedersen, Biogr. Mem. Fellows R. Soc., 1972, 18, 594–627 CrossRef.
- R. Bird, W. Stewart and E. Lightfoot, Transport Phenomena, Wiley, 2006 Search PubMed.
- A. Einstein, Ann. Phys., 1905, 17, 549 CrossRef CAS.
- R. M. German, P. Suri and S. J. Park, J. Mater. Sci., 2009, 44, 1–39 CrossRef CAS.
- J. Burton, J. Rutledge and P. Taborek, Phys. Rev. Lett., 2004, 92, 244505 CrossRef CAS PubMed.
- A.-L. Biance, C. Clanet and D. Quéré, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2004, 69, 016301 CrossRef PubMed.
- R. M. Iverson, M. E. Reid, M. Logan, R. G. LaHusen, J. W. Godt and J. P. Griswold, Nat. Geosci., 2011, 4, 116–121 CrossRef CAS.
- J. D. Paulsen, R. Carmigniani, A. Kannan, J. C. Burton and S. R. Nagel, Nat. Commun., 2014, 5, 1–7 Search PubMed.
- M. J. Murphy and R. J. Adrian, Exp. Fluids, 2010, 49, 193–202 CrossRef CAS.
- L. Bocquet and J.-L. Barrat, Soft Matter, 2007, 3, 685–693 RSC.
- M. Hénot, E. Drockenmuller, L. Léger and F. Restagno, ACS Macro Lett., 2018, 7, 112–115 CrossRef PubMed.
- C.-L. Navier, Mem. Acad. Sci. Inst. Fr., 1823, 6, 389–416 Search PubMed.
- B. Campanella, S. Legnaioli, S. Pagnotta, F. Poggialini and V. Palleschi, Atoms, 2019, 7, 57 CrossRef CAS.
- W. Lauterborn and C.-D. Ohl, Ultrason. Sonochem., 1997, 4, 65–75 CrossRef CAS PubMed.
- T. Gibaud, N. Dagès, P. Lidon, G. Jung, L. C. Ahouré, M. Sztucki, A. Poulesquen, N. Hengl, F. Pignon and S. Manneville, Phys. Rev. X, 2020, 10, 011028 CAS.
- J. Seinfeld, J. Wei, J. Anderson and K. Bischoff, Advances in Chemical Engineering, Elsevier Science, 1992 Search PubMed.
- I. M. Jánosi, D. Jan, K. G. Szabó and T. Tél, Exp. Fluids, 2004, 37, 219–229 CrossRef.
- M.-M. Mac Low and R. S. Klessen, Rev. Mod. Phys., 2004, 76, 125 CrossRef CAS.
- O. A. Basaran, AIChE J., 2002, 48, 1842 CrossRef CAS.
- E. Villermaux, Annu. Rev. Fluid Mech., 2007, 39, 419–446 CrossRef.
- J. Eggers and E. Villermaux, Rep. Prog. Phys., 2008, 71, 036601 CrossRef.
- H. P. Kavehpour, Annu. Rev. Fluid Mech., 2015, 47, 245–268 CrossRef.
- J. B. Keller and M. J. Miksis, SIAM J. Appl. Math., 1983, 43, 268–277 CrossRef.
- L. Courbin, J. C. Bird, M. Reyssat and H. Stone, J. Phys.: Condens. Matter, 2009, 21, 464127 CrossRef CAS PubMed.
- A. Eddi, K. G. Winkels and J. H. Snoeijer, Phys. Fluids, 2013, 25, 013102 CrossRef.
- A. Eddi, K. Winkels and J. Snoeijer, Phys. Rev. Lett., 2013, 111, 144502 CrossRef CAS PubMed.
- F. Feslot, A. Cazabat and N. Fraysse, J. Phys.: Condens. Matter, 1989, 1, 5793 CrossRef.
- J. Kepler, Astronomia nova, American Philosophical Society, 1609, vol. 209 Search PubMed.
- G. Galilei, Dialogue concerning the two chief world systems, Univ. of California Press, 1632 Search PubMed.
- I. Newton, The Principia: mathematical principles of natural philosophy, Univ. of California Press, 1687 Search PubMed.
- C. V. Boys, Nature, 1894, 50, 571 CrossRef.
- L. Bocquet, A. Colin and A. Ajdari, Phys. Rev. Lett., 2009, 103, 036001 CrossRef PubMed.
- J. E. Mack, Semi-popular motion-picture record of the Trinity explosion, Technical Information Division, Oak Ridge Directed Operations, 1946, vol. 221 Search PubMed.
- L. Rayleigh, London, Edinburgh Dublin Philos. Mag. J. Sci., 1890, 30, 386–400 CrossRef.
- W. Thomson, London, Edinburgh Dublin Philos. Mag. J. Sci., 1871, 42, 362–377 Search PubMed.
- D. Nikolić and L. Nešić, Eur. J. Phys., 2012, 33, 1677 CrossRef.
- D. W. Wang and P. A. Hwang, J. At. Oceanic Technol., 2004, 21, 1936–1945 CrossRef.
- U. Frisch, Turbulence: the legacy of AN Kolmogorov, Cambridge University Press, 1995 Search PubMed.
- H. Grant, R. Stewart and A. Moilliet, J. Fluid Mech., 1962, 12, 241–268 CrossRef.
- O. Métais and M. Lesieur, Turbulence and coherent structures, Springer Science & Business Media, 2013, vol. 2 Search PubMed.
- P. A. Davidson, Y. Kaneda, K. Moffatt and K. R. Sreenivasan, A voyage through turbulence, Cambridge University Press, 2011 Search PubMed.
- B. Dubrulle, J. Fluid Mech., 2019, 867, P1 CrossRef.
- V. Steinberg, Annu. Rev. Fluid Mech., 2021, 53, 27–58 CrossRef.
- R. Alert, J. Casademunt and J.-F. Joanny, Ann. Rev. Condens. Matter Phys., 2022, 13, 143–170 CrossRef.
- H. A. Bethe, K. Fuchs, J. O. Hirschfelder, J. L. Magee, R. Peieris and J. von Neumann, Blast wave, Los Alamos National lab. (lanl), Los Alamos, NM (United States) technical report, 1947 Search PubMed.
- N. Rott, Annu. Rev. Fluid Mech., 1990, 22, 1–12 CrossRef.
- M. A. Deakin, Int. J. Math. Educ. Sci. Technol., 2011, 42, 1069–1079 CrossRef.
- J. M. Dealy, Rheol. Bull., 2010, 79, 14–18 Search PubMed.
- R. Poole, Rheol. Bull., 2012, 53, 32–39 Search PubMed.
- J. Burton, R. Waldrep and P. Taborek, Phys. Rev. Lett., 2005, 94, 184502 CrossRef CAS PubMed.
- R. Bolanos-Jiménez, A. Sevilla, C. Martnez-Bazán, D. Van Der Meer and J. Gordillo, Phys. Fluids, 2009, 21, 072103 CrossRef.
- W. Yao, H. Maris, P. Pennington and G. Seidel, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2005, 71, 016309 CrossRef CAS PubMed.
- D. G. Aarts, H. N. Lekkerkerker, H. Guo, G. H. Wegdam and D. Bonn, Phys. Rev. Lett., 2005, 95, 164503 CrossRef PubMed.
- D. G. Aarts and H. N. Lekkerkerker, J. Fluid Mech., 2008, 606, 275–294 CrossRef CAS.
- J. D. Paulsen, J. C. Burton and S. R. Nagel, Phys. Rev. Lett., 2011, 106, 114501 CrossRef PubMed.
- M. M. Rahman, W. Lee, A. Iyer and S. J. Williams, Phys. Fluids, 2019, 31, 012104 CrossRef.
- J. Eggers, Phys. Rev. Lett., 1993, 71, 3458 CrossRef PubMed.
- M. P. Brenner, J. R. Lister and H. A. Stone, Phys. Fluids, 1996, 8, 2827–2836 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2024 |