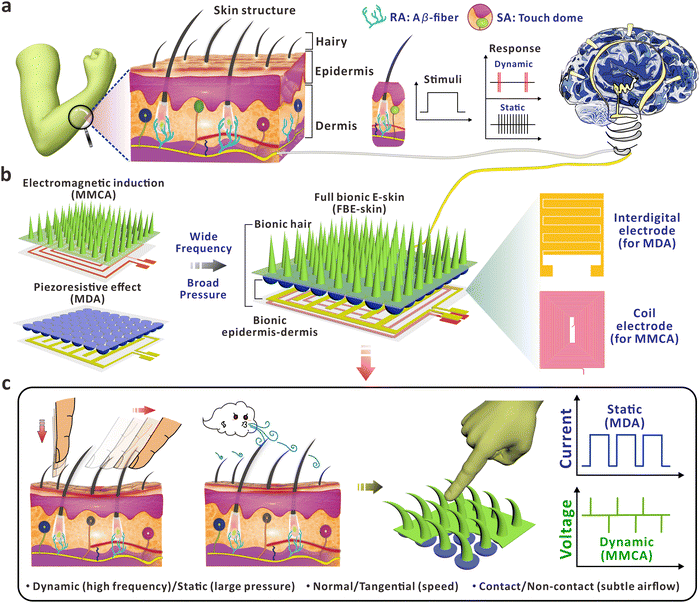
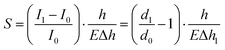Top-down architecture of magnetized micro-cilia and conductive micro-domes as fully bionic electronic skin for de-coupled multidimensional tactile perception†
Fengming
Hu
ab,
Qian
Zhou
c,
Ruolin
Liu
a,
Yanfei
Zhu
b,
Yuanzhe
Liang
a,
Dan
Fang
a,
Bing
Ji
d,
Zhiming
Chen
 *b,
Jianyi
Luo
*b,
Jianyi
Luo
 *b and
Bingpu
Zhou
*b and
Bingpu
Zhou
 *a
*a
aJoint Key Laboratory of the Ministry of Education, Institute of Applied Physics and Materials Engineering, University of Macau, Avenida da Universidade, Taipa, Macau 999078, China. E-mail: bpzhou@um.edu.mo
bResearch Center of Flexible Sensing Materials and Devices, School of Applied Physics and Materials, Wuyi University, Jiangmen 529020, China. E-mail: chenzhiming@wyu.edu.cn; luojiany@mail3.sysu.edu.cn
cSchool of Physics and Electronics, Central South University, Changsha 410083, China
dSchool of Physics and Electronics, Hunan Normal University, Changsha 410081, China
First published on 12th November 2024
Abstract
Electronic skin (E-skin) has attracted considerable attention for simulating the human sensory system for use in prosthetics, human–machine interactions, and healthcare monitoring. However, it is still challenging to fully mimic the skin function that can de-couple stimuli such as normal/tangential forces, contact/non-contact behaviors, and react to high-frequency inputs. Herein, we propose fully bionic E-skin (FBE-skin), which consists of a magnetized micro-cilia array (MMCA), a micro-dome array (MDA), and flexible electrodes to completely duplicate the hairy layer, epidermis/dermis interface, and subcutaneous mechanoreceptors of human skin. The optimized MDA and interdigital electrode enable the FBE-skin to perceive static forces with a linear sensitivity of 96.6 kPa−1 up to 100 kPa, while the branch of electromagnetic induction allows the FBE-skin to sensitively capture dynamic stimuli with vibrating signals up to 100 Hz. The top-down integration of MDA and MMCA not only replicates the three-dimensional structure of human skin, but also synergistically provides the FBE-skin with bionic rapidly adapting (RA) and slowly adapting (SA) receptors. Consequently, the FBE-skin is capable of perceiving dynamic/static, normal/tangential, and contact/non-contact stimuli with a broad range of working pressures and frequencies. We expect that the design of FBE-skin will be promising for widespread applications from intelligent sensing to human–machine interactions.
New conceptsMultifunctional electronic skin (E-skin) that de-couples normal/tangential, contact/non-contact, or high-frequency mechanical stimuli is still challenging in the field of flexible sensors. Herein, fully bionic E-skin (FBE-skin) with top-down architecture of magnetized micro-cilia, conductive micro-domes and flexible electrodes is developed by mimicking the “hair–epidermis–dermis–subcutaneous” layers of human skin. The optimized micro-domes enable the FBE-skin to perceive static forces with linear sensitivity of 96.6 kPa−1 up to 100 kPa, while electromagnetic induction from micro-cilia allows the FBE-skin to capture dynamic signals with frequency up to 100 Hz. The top-down assembly not only replicates the 3D skin structure, but also synergistically provides the FBE-skin with bionic RA and SA receptors. The FBE-skin is able to distinguish the tangential input via lateral deformation of the magnetized micro-cilia, while the micro-domes are mainly responsible for the static pressure branch to cover a full mechanical spectrum. A sensing array and visualization system was built up to digitize human sensations, showcasing the potentials for sliding monitoring, providing warnings for close-range stimuli and subtle airflow perception. We expect that the design could upgrade the applications of E-skin for potential uses from tactile sensing, intelligent interaction, to future prosthetics. |
1. Introduction
Skin is the largest sensory organ of human body, responsible for detecting various active/passive stimuli due to its unique and comprehensive internal and external biological structures. Intelligent E-skin aims to detect and distinguish different external stimuli by manufacturing flexible electronic devices that mimic or even surpass the functionality of human skin, converting them into processable and recognizable electrical signals. The powerful functionality and flexible physical and mechanical properties of E-skin offer extensive application potential in areas such as health monitoring,1,2 human–machine interactions,3 and robot haptic perception.4,5 Current E-skin primarily relies on sensors based on nanomaterials and micro/nanostructures to simulate the local structure of the skin and convert unimodal sensory information. These sensors are designed to detect stress, strain, temperature, optical and electrophysiological signals,6–9 with an emphasis on optimizing the physical performance of specific sensors, developing new manufacturing methods, and signal processing techniques. However, E-skin with single functionality alone is insufficient to convey comprehensive information and accomplish complex tasks in practical applications. Unlike unimodal sensing, multimodal sensing provides multi-functionality and can simulate different types of receptors in the skin to achieve higher bionic performance, endowing the E-skin with a more natural perception capability.In contrast to unimodal tactile perception that focuses on achieving a single stimulus, researchers have recently combined different types of flexible sensors based on various sensing mechanisms such as piezoresistive, capacitive, piezoelectric, and triboelectric mechanisms.10–13 These combinations are used to detect multiple types of information, such as object shape, temperature, humidity and pressure,14–18 providing flexibility and versatility in various applications. For example, Shin et al.19 developed a multi-modal sensor based on interlocked ferroelectric copolymer micro-structures, capable of simultaneously detecting mechanical and thermal stimuli. Qiao et al.20 developed a highly stretchable artificial ionic skin coupled with a resistive ion skin, simulating the dynamic and static pressure signals during the grasping process of soft robots or artificial fingers. Nowadays, with the continuous development of flexible electronics and emergence of intelligence, there are still significant challenges in mimicking different types of rapidly adapting (RA)/slowly adapting (SA) receptors on the human skin.21 The RA receptors are sensitive to mechanical stimuli with high vibration frequencies, while the SA receptors are mainly responsible for static pressure or mechanical inputs in low frequencies. The co-duplication of both receptors in biological skin can thus comprehensively perceive and interact with the external world to explore the practical applications of E-skin. In recent years, researchers have demonstrated a series of interlocked micro/nano-structured pressure sensors, including micro-spines,22 micro-pyramids,23,24 micro-pillars,25,26 micro-domes27–29 and nanohelixes,30 to enhance the multidimensional tactile perception function of the E-skin. Apart from the microstructures mentioned above, duplication of bio-inspired hairy structures on the superficial layer of human skin has also attracted abundant interest.31 The slight movements of flexible hair roots induced by external stimuli can be sensitively captured by nearby receptors, which plays an indispensable role in human tactile perception, e.g. subtle airflow.32 Such capability allows the artificial cilia to sensitively sense environmental changes, showcasing the potential for applications from environmental sensing, controllable manipulation, to biomedical diagnostics.33,34 However, it is still challenging to flexibly regulate the biomimetic hairy structures with controllable morphology, e.g. a high aspect ratio, in a straightforward approach. For example, hairy structures with a high aspect ratio are capable of responding to mechanical inputs in normal and tangential directions with obvious deformation. To fulfill the multiple functions of tactile perception, it is also essential that the cilia should be able to directly convert the mechanical stimuli to identifiable electrical signals.35,36
In view of the above, fully bionic E-skin (FBE-skin) based on the combination of electromagnetic induction and piezoresistive sensing principles is designed to re-establish the architecture of human skin for multidimensional analysis of mechanical signals. Using the concept of a top-down structure, artificial MMCA, MDA, and underlying flexible electrodes were manufactured and integrated to fully mimic the hairy layer, epidermis–dermis interface, subcutaneous RA (Aβ-fiber) and SA (Touch dome) mechanoreceptors of human skin as shown in Fig. 1a. Benefiting from the excellent elasticity of the high-aspect-ratio MMCA, the FBE-skin is capable of detecting high-frequency dynamic stimuli, which is limited by the responsive characteristics of conventional structures such as resistive micro-domes.37–42 As depicted in Fig. 1b, the synergistic effect of functionalized MMCA and MDA allows the FBE-skin to simultaneously perceive the external stimuli in a wide frequency and broad pressure range, which is analogous to the RA and SA channels in human skin. For the dual-layer integrated electrodes (Fig. 1b), the interdigital electrode serves as the channel to collect the inputs from the MDA, while the conductive coil transmits the induced voltage that is caused by the deformation of MMCA and related magnetic flux variation. Through the morphological regulation and magnetized optimization, the bionic MMCA part was able to provide reliable electrical feedback for dynamic inputs with frequencies of up to 100 Hz. Furthermore, the resistive MDA component was optimized to enable a linear detection of static pressure of up to 100 kPa at a high sensitivity of 96.6 kPa−1. The integration of MMCA and MDA thus functions in a complementary and cooperative behavior to the external mechanical stimuli with more detailed information, including the moment of touch/release, mechanical duration, and the sliding speed/direction. Integrated with the three-dimensional (3D) MMCA, the FBE-skin is capable of discriminating between multidimensional mechanical signals in dynamic/static, normal/tangential, and contact/non-contact formats (Fig. 1c). Via customization of a visualization system based on 16 FBE-skin units, this work simulates the functionality of human skin in proximal sensing before skin contact, recognition of mechanical intensity with harmful warning, monitoring the sliding information of tangential input, and the spatial distribution of subtle airflow.
2. Results and discussion
2.1. Optimization of MMCA components for dynamic perception
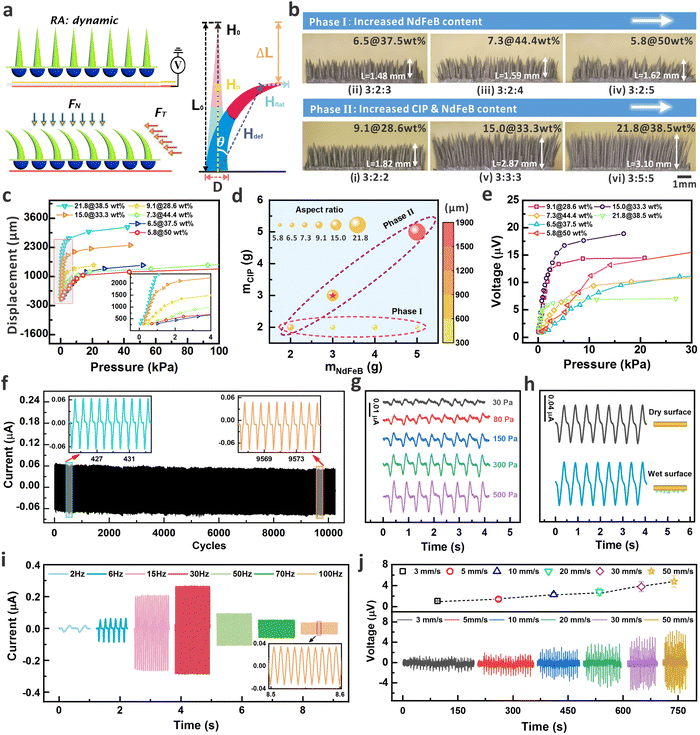
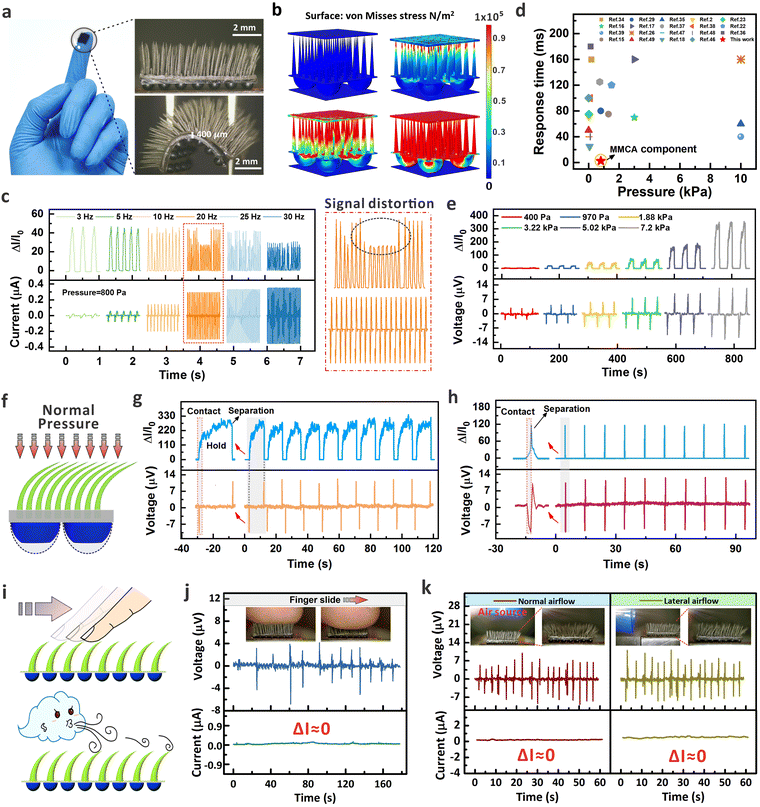
The fabrication process of the device is provided in Fig. S1 (ESI†), which includes the molding of MDA, self-assembled MMCA, and integration of MMCA and MDA as a hybrid structure (see details in the Experimental section). The SEM image also exhibits the well-aligned micro-cilia, and the uniform element distribution within the micro-cilia matrix as shown by the EDX results in Fig. S2 (ESI†). As shown in Fig. 2a, inspired by the perception of external stimuli by the hairy layer of human skin, a combination of MMCA structure and the single-layer copper coil electrode is used to simulate the detection of normal (FN) and tangential (FT) dynamic mechanical information through the RA mechanoreceptor. Because of the excellent ferromagnetic properties, the NdFeB-incorporated MMCA could serve as a flexible and permanent magnet after the magnetization in a vertical orientation (Fig. S3a, ESI†). Therefore, the overall orientation of the magnetic moments within the MMCA matrix can be altered if the MMCA is deformed. This will finally cause the changes in the spatial distribution of the localized magnetic field, as visualized from the simulation results in Fig. S3b (ESI†). When the magnetized micro-cilia are deformed by external forces, the total magnetic flux and normal magnetic flux density passing through the coil will subsequently vary. According to the law of electromagnetic induction, the induced electromotive force (voltage) in the coil can be expressed as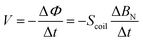 (Fig. S3c, ESI†), where Φ is the total magnetic flux, BN is the normal magnetic flux density through the coil, and Scoil is the constant area of the copper coil. As a result, the elastic MMCA could sensitively respond to the normal and tangential dynamic pressures with electrical outputs in a self-powered manner. As depicted in Fig. 2a, we approximated the global magnetic field intensity vector (H), as a superposition of the magnetic field intensity vectors of the flat body (Hflat) and the deflected body (Hdef) within the matrix of the bent micro-cilia. The magnetic field intensity vectors, Hflat, do not contribute to the intensity of the normal component (Hn). In this case, the value of H is equal to
(Fig. S3c, ESI†), where Φ is the total magnetic flux, BN is the normal magnetic flux density through the coil, and Scoil is the constant area of the copper coil. As a result, the elastic MMCA could sensitively respond to the normal and tangential dynamic pressures with electrical outputs in a self-powered manner. As depicted in Fig. 2a, we approximated the global magnetic field intensity vector (H), as a superposition of the magnetic field intensity vectors of the flat body (Hflat) and the deflected body (Hdef) within the matrix of the bent micro-cilia. The magnetic field intensity vectors, Hflat, do not contribute to the intensity of the normal component (Hn). In this case, the value of H is equal to 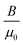 , where μ0 is the vacuum permeability and B is the spatial magnetic flux density. As discussed in our previous report,43 the resultant Hn is a function dependent on the parameters including H0, L0, ΔL, and θ, where H0 and L0 are the total magnetic field intensity and length of the unbent MMCA, ΔL is the deformation magnitude of the MMCA when exposed to external stimuli, and θ is the bending angle. Consequently, it is essential to regulate the intrinsic properties of the MMCA so that the sensing performance can be optimized.
, where μ0 is the vacuum permeability and B is the spatial magnetic flux density. As discussed in our previous report,43 the resultant Hn is a function dependent on the parameters including H0, L0, ΔL, and θ, where H0 and L0 are the total magnetic field intensity and length of the unbent MMCA, ΔL is the deformation magnitude of the MMCA when exposed to external stimuli, and θ is the bending angle. Consequently, it is essential to regulate the intrinsic properties of the MMCA so that the sensing performance can be optimized.
In principle, the sensing performance of the MMCA is mainly determined by the morphological and inherent remanence of the micro-cilia. We thus tuned the preparation parameters to optimize the overall performance of the micro-cilia layer, e.g. stability, sensitivity, and deformability. Fig. S4 (ESI†) shows the detailed formation process and mechanism of the MMCA architecture, which is attributed by the synergistic effect from the magnetic force, surface tension, and the composite gravity.43Via continuously spraying the composite droplets on the sample surface, the competitive balance of different forces would allow the micro-cilia to grow in a self-assembled behavior. As a result, by adjusting the mass ratio NdFeB and CIP in the mixture, the dimensional properties (e.g. aspect ratio) of the MMCA can be customized to optimize the sensing performance such as sensitivity and detection range. The optical images of the as-prepared MMCA are shown in Fig. 2b, where the mass ratios of mPDMS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mCIP
mCIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mNdFeB were 3
mNdFeB were 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (i), 3
2 (i), 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (ii), 3
3 (ii), 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (iii), 3
4 (iii), 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (iv), 3
5 (iv), 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (v) and 3
3 (v) and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (vi). Specifically, the comparison of samples (ii–iv) in Phase I and (i) in Phase II provides information about the ratio of NdFeB component. For this stage, we mainly focused on the influence of NdFeB contents on the morphology of the MMCA and the corresponding device performance. As shown in the optical images, the increased content of NdFeB would result in an increase in the base diameter (D) of the micro-cilia due to the residual deposited at the bottom surface. This will finally lead to a decrease in the aspect ratio of the micro-cilia as the heights remain almost unchanged when the CIP percentage is maintained. Consequently, micro-cilia with a smaller aspect ratio are less prone to undergo deformation if the same pressure was applied (Fig. 2c). For example, it can be observed that under the same pressure, the MMCA displacement of samples 5.8@50 wt%, 6.5@37.5 wt%, and 7.3@44.4 wt% are smaller when compared with others in a larger aspect ratio. Herein, we introduced the notation “aspect ratio@NdFeB content” to distinguish different parameters of the MMCA sample (e.g., 15.0@33.3 wt% represents an aspect ratio of 15.0 with NdFeB content of 33.3 wt% in the MMCA). As depicted in Fig. 2a, the aspect ratio is defined as the ratio of the micro-cilia length (L) to the diameter of the base circle (D). With the statistics obtained from the side-view optical images, the aspect ratio of the conical micro-cilia can be obtained (see Table S1 for the detailed dimensional information, ESI†). To demonstrate the positive dependence of larger aspect ratios for deformation, the COMSOL simulation was performed to evaluate the displacement behavior of four micro-cilia in different aspect ratios (see Fig. S5, ESI†). Under the same normal and lateral stimuli, the micro-cilia with a larger aspect ratio can provide a more obvious displacement, indicating the superior flexibility that is accompanied by the morphology of the micro-cilia.
5 (vi). Specifically, the comparison of samples (ii–iv) in Phase I and (i) in Phase II provides information about the ratio of NdFeB component. For this stage, we mainly focused on the influence of NdFeB contents on the morphology of the MMCA and the corresponding device performance. As shown in the optical images, the increased content of NdFeB would result in an increase in the base diameter (D) of the micro-cilia due to the residual deposited at the bottom surface. This will finally lead to a decrease in the aspect ratio of the micro-cilia as the heights remain almost unchanged when the CIP percentage is maintained. Consequently, micro-cilia with a smaller aspect ratio are less prone to undergo deformation if the same pressure was applied (Fig. 2c). For example, it can be observed that under the same pressure, the MMCA displacement of samples 5.8@50 wt%, 6.5@37.5 wt%, and 7.3@44.4 wt% are smaller when compared with others in a larger aspect ratio. Herein, we introduced the notation “aspect ratio@NdFeB content” to distinguish different parameters of the MMCA sample (e.g., 15.0@33.3 wt% represents an aspect ratio of 15.0 with NdFeB content of 33.3 wt% in the MMCA). As depicted in Fig. 2a, the aspect ratio is defined as the ratio of the micro-cilia length (L) to the diameter of the base circle (D). With the statistics obtained from the side-view optical images, the aspect ratio of the conical micro-cilia can be obtained (see Table S1 for the detailed dimensional information, ESI†). To demonstrate the positive dependence of larger aspect ratios for deformation, the COMSOL simulation was performed to evaluate the displacement behavior of four micro-cilia in different aspect ratios (see Fig. S5, ESI†). Under the same normal and lateral stimuli, the micro-cilia with a larger aspect ratio can provide a more obvious displacement, indicating the superior flexibility that is accompanied by the morphology of the micro-cilia.
As presented by the optical images in Phase II (Fig. 2b), the co-increased amount of CIP and NdFeB allows the further regulation of the aspect ratio. When the mass ratio reached 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, the aspect ratio of the as-prepared micro-cilia array was 21.8. Fig. 2d summarizes the aspect ratio and the displacement of the MMCA under a constant normal pressure of 1 kPa, which shows a positive relationship between the aspect ratio and the displacement. The results show that the shape of MMCA can be flexibly regulated via tuning the mass ratio of different components in the matrix. The controllable aspect ratio also allows the MMCA to maintain the deformation capability even with a relatively high content of the filler, e.g. NdFeB. Apart from the deformation capability, the voltage output is another critical parameter to evaluate the potential of the MMCA for tactile perception. The voltage profiles from different MMCA are provided in Fig. 2e and Fig. S6 (ESI†), with the dynamic pressure range of up to 30 kPa under a constant compression speed of 30 mm s−1. Normally, a larger aspect ratio renders a higher sensitivity because of the more obvious deformation under the identical pressure. For example, the samples of 15.0@33.3 wt%, and 21.8@38.5 wt% exhibit a sensitivity of 6.12 μV kPa−1 and 5.59 μV kPa−1, respectively (see Table S1 for the detailed sensing performance, ESI†). We also notice that for the sample of 21.8@38.5 wt%, the voltage generation reached the saturation point when the pressure is ∼1.0 kPa, which is narrower than that of the sample 15.0@33.3 wt%. In one way, the high aspect ratio of this sample results in a full deformation within a narrower pressure range. Furthermore, the smaller diameter of such MMCA leads to a smaller amount of the effective remanence, and thus the amount of magnetic flux variation is limited to produce a larger voltage for sensitivity guarantee. The plots in Fig. S7 (ESI†) directly relate the induced voltage and the displacement. From the curves, we can observe that even the high aspect ratio of 21.8@38.5 wt% renders an easier deformation, the voltage signals are relatively weaker because of the remanence per unit volume. Also, the smallest aspect ratio of 5.8@50 wt% can provide the significant voltage signals when exposed to the same displacement. However, it requires a larger pressure to obtain the same displacement due to the larger diameter of the MMCA. Considering the balanced effects from remanent magnetism, deformation behavior, and specifically the magnetic flux change per unit volume during MMCA bending, the micro-cilia array with parameters of 15.0@33.3 wt% (starred in Fig. 2d) and area of 10 × 10 mm2 were finally selected as the optimized device for the following studies. It should be noted that when reducing the area of the MMCA component, the detection limit for mechanical stimulation would also be decreased due to the decreased amount of MMCA and the corresponding magnetic flux magnitude. This behavior will finally affect the magnitude of induced electric signals and sensing performance as confirmed by the experimental results in Fig. S8 (ESI†).
5, the aspect ratio of the as-prepared micro-cilia array was 21.8. Fig. 2d summarizes the aspect ratio and the displacement of the MMCA under a constant normal pressure of 1 kPa, which shows a positive relationship between the aspect ratio and the displacement. The results show that the shape of MMCA can be flexibly regulated via tuning the mass ratio of different components in the matrix. The controllable aspect ratio also allows the MMCA to maintain the deformation capability even with a relatively high content of the filler, e.g. NdFeB. Apart from the deformation capability, the voltage output is another critical parameter to evaluate the potential of the MMCA for tactile perception. The voltage profiles from different MMCA are provided in Fig. 2e and Fig. S6 (ESI†), with the dynamic pressure range of up to 30 kPa under a constant compression speed of 30 mm s−1. Normally, a larger aspect ratio renders a higher sensitivity because of the more obvious deformation under the identical pressure. For example, the samples of 15.0@33.3 wt%, and 21.8@38.5 wt% exhibit a sensitivity of 6.12 μV kPa−1 and 5.59 μV kPa−1, respectively (see Table S1 for the detailed sensing performance, ESI†). We also notice that for the sample of 21.8@38.5 wt%, the voltage generation reached the saturation point when the pressure is ∼1.0 kPa, which is narrower than that of the sample 15.0@33.3 wt%. In one way, the high aspect ratio of this sample results in a full deformation within a narrower pressure range. Furthermore, the smaller diameter of such MMCA leads to a smaller amount of the effective remanence, and thus the amount of magnetic flux variation is limited to produce a larger voltage for sensitivity guarantee. The plots in Fig. S7 (ESI†) directly relate the induced voltage and the displacement. From the curves, we can observe that even the high aspect ratio of 21.8@38.5 wt% renders an easier deformation, the voltage signals are relatively weaker because of the remanence per unit volume. Also, the smallest aspect ratio of 5.8@50 wt% can provide the significant voltage signals when exposed to the same displacement. However, it requires a larger pressure to obtain the same displacement due to the larger diameter of the MMCA. Considering the balanced effects from remanent magnetism, deformation behavior, and specifically the magnetic flux change per unit volume during MMCA bending, the micro-cilia array with parameters of 15.0@33.3 wt% (starred in Fig. 2d) and area of 10 × 10 mm2 were finally selected as the optimized device for the following studies. It should be noted that when reducing the area of the MMCA component, the detection limit for mechanical stimulation would also be decreased due to the decreased amount of MMCA and the corresponding magnetic flux magnitude. This behavior will finally affect the magnitude of induced electric signals and sensing performance as confirmed by the experimental results in Fig. S8 (ESI†).
Fig. 2f shows the results of cyclic loading and unloading tests on the device with a normal pressure load of 5 kPa and a frequency of 2 Hz for up to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The repeatability of the current signal indicates that the MMCA possesses a good mechanical durability for potential long-term uses. Thanks to the excellent deformation capability, the MMCA-based E-skin can respond to tiny pressures in Fig. 2g, where the pressure load size is 30 Pa. Under a higher frequency of 30 Hz, the detection limit of external mechanical stimuli can be as low as 3.5 Pa (Fig. S9, ESI†). The principle of electromagnetic induction also ensures that the MMCA can produce reliable signal that is immune to the environmental conditions such as humidity. Compared to hair-like silicon-based sensors,44,45 our ciliated components have excellent environmental immunity. Fig. S10 (ESI†) further presents the summarized voltage response of the MMCA with different temperatures (25 °C, 50 °C, and 100 °C) and weak magnetic field environments (the magnetic field intensity in the daily environment is generally lower than 10 mT). In short, the constant output signals can be maintained even under a high temperature and weak magnetic field surroundings, indicating the immunity of our proposed device for stable operation in daily life. Fig. 2h demonstrates the output current signals of the MMCA device when it touches the surface of dry and wet objects, and the consistency of the signal amplitudes further reflects the anti-interference ability of the E-skin to the humid environments. According to the induction mechanism, the magnitude of the induced current in the coil (resistance of ∼20 Ω) is also negatively correlated with the frequency of external mechanical stimulation. As shown in Fig. 2i, the relationship between the induced current and the frequency of mechanical load applied to MMCA is verified, highlighting the advantage of the cilia device in perceiving high-frequency signals. Within the range of 2–30 Hz, the induced current in the coil increases monotonously with the increase of the applied frequency, which is consistent with the law of induction. Furthermore, the vibration signals with higher frequency of 50 Hz, 70 Hz, and 100 Hz can still be accurately detected. The results indicate that the MMCA could respond rapidly to vibrations at 100 Hz, fully reaching the response frequency of the RA receptors in human skin.46,47 Note that within range of 50–100 Hz, the current signals show an attenuated trend, which is because of the protective mechanism of the vibrator instrument via automatically reducing the vibration amplitude when the excitation frequency is too high. Similarly, the MMCA device also exhibits good perception performance for low-frequency mechanical stimuli down to 1 Hz, as shown in Fig. S11 (ESI†). At a higher pressure of 1.35 kPa, the cyclic pressure could quickly deform the micro-cilia to induce magnetic flux change that generates the periodical signals. Apart from the normal pressure that causes deformation of cilia, we also evaluate the capability of signal generation when a lateral bending of micro-cilia occurs. A motorized slider was applied to simulate the hand motion that passes over the device at different speeds ranging from 3 mm s−1 to 50 mm s−1. Fig. 2j illustrates the signal profiles when the micro-cilia are bent by a constant 900 μm (ΔL in Fig. 2a). As observed from the plots, a faster sliding speed results in a quicker bending and a larger voltage amplitude is produced accordingly. The dynamic response exhibits approximately symmetrical peak-to-peak values of the positive and negative voltages, which is attributed by the excellent inherent elasticity of the micro-cilia. Furthermore, the motorized platform was controlled at a sliding speed of 50 mm s−1 to periodically bend the micro-cilia array for robustness determination. As demonstrated in Fig. S12 (ESI†), the induced voltage signals could maintain a repeatable behavior after 5000 cycles, indicating the stability of the MMCA after the continuous lateral deformation. As demonstrated below, the capability of electrically reflecting tangential dynamic stimuli provides the MMCA-integrated E-skin with the potential to recognize the speed of in-plane stimuli under a contactless behavior. We further evaluated the repeatability of electrical signals from the MMCA devices, which were prepared by the same experimental procedure and conditions. As demonstrated in Fig. S13 (ESI†), four MMCA devices exhibited a similar morphology and dimensional properties, and the plots of pressure response further indicated the reproducible sensing capability from different MMCA samples.
000 cycles. The repeatability of the current signal indicates that the MMCA possesses a good mechanical durability for potential long-term uses. Thanks to the excellent deformation capability, the MMCA-based E-skin can respond to tiny pressures in Fig. 2g, where the pressure load size is 30 Pa. Under a higher frequency of 30 Hz, the detection limit of external mechanical stimuli can be as low as 3.5 Pa (Fig. S9, ESI†). The principle of electromagnetic induction also ensures that the MMCA can produce reliable signal that is immune to the environmental conditions such as humidity. Compared to hair-like silicon-based sensors,44,45 our ciliated components have excellent environmental immunity. Fig. S10 (ESI†) further presents the summarized voltage response of the MMCA with different temperatures (25 °C, 50 °C, and 100 °C) and weak magnetic field environments (the magnetic field intensity in the daily environment is generally lower than 10 mT). In short, the constant output signals can be maintained even under a high temperature and weak magnetic field surroundings, indicating the immunity of our proposed device for stable operation in daily life. Fig. 2h demonstrates the output current signals of the MMCA device when it touches the surface of dry and wet objects, and the consistency of the signal amplitudes further reflects the anti-interference ability of the E-skin to the humid environments. According to the induction mechanism, the magnitude of the induced current in the coil (resistance of ∼20 Ω) is also negatively correlated with the frequency of external mechanical stimulation. As shown in Fig. 2i, the relationship between the induced current and the frequency of mechanical load applied to MMCA is verified, highlighting the advantage of the cilia device in perceiving high-frequency signals. Within the range of 2–30 Hz, the induced current in the coil increases monotonously with the increase of the applied frequency, which is consistent with the law of induction. Furthermore, the vibration signals with higher frequency of 50 Hz, 70 Hz, and 100 Hz can still be accurately detected. The results indicate that the MMCA could respond rapidly to vibrations at 100 Hz, fully reaching the response frequency of the RA receptors in human skin.46,47 Note that within range of 50–100 Hz, the current signals show an attenuated trend, which is because of the protective mechanism of the vibrator instrument via automatically reducing the vibration amplitude when the excitation frequency is too high. Similarly, the MMCA device also exhibits good perception performance for low-frequency mechanical stimuli down to 1 Hz, as shown in Fig. S11 (ESI†). At a higher pressure of 1.35 kPa, the cyclic pressure could quickly deform the micro-cilia to induce magnetic flux change that generates the periodical signals. Apart from the normal pressure that causes deformation of cilia, we also evaluate the capability of signal generation when a lateral bending of micro-cilia occurs. A motorized slider was applied to simulate the hand motion that passes over the device at different speeds ranging from 3 mm s−1 to 50 mm s−1. Fig. 2j illustrates the signal profiles when the micro-cilia are bent by a constant 900 μm (ΔL in Fig. 2a). As observed from the plots, a faster sliding speed results in a quicker bending and a larger voltage amplitude is produced accordingly. The dynamic response exhibits approximately symmetrical peak-to-peak values of the positive and negative voltages, which is attributed by the excellent inherent elasticity of the micro-cilia. Furthermore, the motorized platform was controlled at a sliding speed of 50 mm s−1 to periodically bend the micro-cilia array for robustness determination. As demonstrated in Fig. S12 (ESI†), the induced voltage signals could maintain a repeatable behavior after 5000 cycles, indicating the stability of the MMCA after the continuous lateral deformation. As demonstrated below, the capability of electrically reflecting tangential dynamic stimuli provides the MMCA-integrated E-skin with the potential to recognize the speed of in-plane stimuli under a contactless behavior. We further evaluated the repeatability of electrical signals from the MMCA devices, which were prepared by the same experimental procedure and conditions. As demonstrated in Fig. S13 (ESI†), four MMCA devices exhibited a similar morphology and dimensional properties, and the plots of pressure response further indicated the reproducible sensing capability from different MMCA samples.
2.2. Optimization of piezoresistive performance of the E-skin
For the perception of normal static pressure with larger magnitude, conductive micro-structured components were introduced and integrated under the MMCA to fully mimic the human skin. Fig. 3a shows the layout of a piezoresistive sensing component, which is constructed using a conductive micro-dome array and interdigitated electrodes. The double-layered flexible electrode is composed of the conductive coil and the interdigital component (see Fig. S14 (ESI†) with the detailed design of the layout). Furthermore, Fig. S15 (ESI†) shows the individual electrode patterning of the top and bottom layers, which also exhibits the electrical resistance stability after different bending angles (maximum value of 120°). With an external electrical meter, the mechanical input can be recognized via collecting the signal variation, which primarily simulates the detection of static mechanical stimuli through SA. When a pressure load is applied, the MDA structure touches the bottom electrodes and forms a closed loop circuit with each row and column in parallel. As depicted in Fig. S16 (ESI†), the optimized MDA component consists of the conductive micro-dome and a non-conductive transparent PDMS substrate. Such design ensures a flat substrate morphology and results in an equivalent circuit of the micro-domes in parallel. The optical images also demonstrate that the MDA branch consists of the PDMS/CNT micro-domes in black and the pure PDMS substrate with transparency. For a piezoresistive sensor, the sensitivity, S, is typically defined as the ratio of the relative current change to the applied pressure,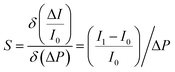 , where I and P represent the final (I1) and initial (I0) current and applied pressure, respectively. According to Holm's theory48 for contact resistance and Hooke's law, the sensitivity can finally be expressed as
, where I and P represent the final (I1) and initial (I0) current and applied pressure, respectively. According to Holm's theory48 for contact resistance and Hooke's law, the sensitivity can finally be expressed aswhere E is the elastic modulus of the Ecoflex/PDMS composite that forms the micro-dome structure, d1 is the resultant contact diameter between the micro-dome and the electrode when pressure is applied, d0 is the initial contact diameter, and h is the height of the micro-domes (see Supplementary Note S1 for details, ESI†). Note that the diameter (d) of the micro-domes in this work was fixed at 1 mm, which is determined by the ball-end cutter in the mold carving process (Fig. S1, ESI†). Consequently, the sensitivity can be possibly improved either by adjusting the mass ratio of Ecoflex/PDMS to control the elastic modulus, or by regulating the height of the MDA to control the relative dimensional variation under a specific pressure.
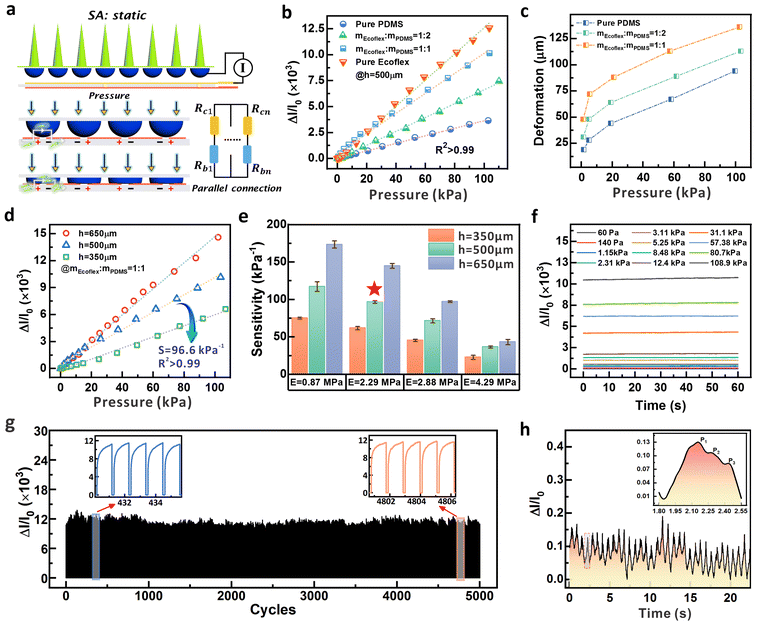
The sample resistances based on different carbon nanotube (CNT) contents, ranging from 5 wt% to 10 wt%, were firstly investigated to optimize the conductivity of the circular structure (Fig. S17, ESI†). The resistance of the samples decreased continuously with increasing CNT content, indicating that a higher CNT content translates to a better conductivity. Among them, the sample with 8 wt% CNT content reached a saturation point in terms of resistivity change. It is evident that there is little difference in the resistance between CNT contents of 8 wt%, 9 wt%, and 10 wt%. Additionally, higher CNT content in the prepared conductive gel results in a higher viscosity, which inevitably increases the difficulty in device fabrication and repeatability. Therefore, we selected 8 wt% CNT content to prepare the conductive dome-shaped array structure in this study. To optimize the sensitivity performance of the piezoresistive part, we fixed the height of the micro-dome at 500 μm, and investigated the effect from the elastic modulus of the sample via regulating the component ratios between PDMS and EcoflexTM 0010 (mEcoflex![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mPDMS). Fig. S18a (ESI†) provides the elastic modulus of the composite under different mass ratios of pure PDMS, mEcoflex
mPDMS). Fig. S18a (ESI†) provides the elastic modulus of the composite under different mass ratios of pure PDMS, mEcoflex![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mPDMS at 1
mPDMS at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 1
2 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and pure Ecoflex. Owing to the obvious modulus difference between PDMS and Ecoflex, a higher content of Ecoflex results in a decreased elastic modulus and better compressibility of the composite.49 Consequently, the micro-dome arrays prepared by pure Ecoflex exhibit the highest sensitivity (Fig. 3b). This is attributed by the more obvious deformation and intensified contact area change that cause a more significant current variation under the same static pressure. We also observed the linear sensing behavior for pressure range up to ∼100 kPa (R2 > 0.99) when the sensing configuration was composed of the circular micro-dome and the underlying interdigital electrodes. Note that for the pure Ecoflex-based (EcoflexTM 00-10) micro-domes, the capability of elastic recovery is weak owing to the low modulus of ∼0.87 MPa (Fig. S18b, ESI†). When a pressure of 100 kPa was applied to the device, the sticky pure Ecoflex-based micro-structures could not quickly recover to the initial status, raising the concerns about the sensing repeatability and hysteresis. With incorporation of PDMS, the micro-domes exhibit restorable morphological change under external stimuli, and the pressure-dependent deformation processes are recorded in Fig. S19 (ESI†). Fig. 3c further lists the compressive deformation of the micro-domes based on the applied normal pressure, which was obtained from the real-time optical images. From the curves, we can find that if the same pressure was applied, in the case of mEcoflex
1, and pure Ecoflex. Owing to the obvious modulus difference between PDMS and Ecoflex, a higher content of Ecoflex results in a decreased elastic modulus and better compressibility of the composite.49 Consequently, the micro-dome arrays prepared by pure Ecoflex exhibit the highest sensitivity (Fig. 3b). This is attributed by the more obvious deformation and intensified contact area change that cause a more significant current variation under the same static pressure. We also observed the linear sensing behavior for pressure range up to ∼100 kPa (R2 > 0.99) when the sensing configuration was composed of the circular micro-dome and the underlying interdigital electrodes. Note that for the pure Ecoflex-based (EcoflexTM 00-10) micro-domes, the capability of elastic recovery is weak owing to the low modulus of ∼0.87 MPa (Fig. S18b, ESI†). When a pressure of 100 kPa was applied to the device, the sticky pure Ecoflex-based micro-structures could not quickly recover to the initial status, raising the concerns about the sensing repeatability and hysteresis. With incorporation of PDMS, the micro-domes exhibit restorable morphological change under external stimuli, and the pressure-dependent deformation processes are recorded in Fig. S19 (ESI†). Fig. 3c further lists the compressive deformation of the micro-domes based on the applied normal pressure, which was obtained from the real-time optical images. From the curves, we can find that if the same pressure was applied, in the case of mEcoflex![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mPDMS at 1
mPDMS at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, deformation occurs more significantly when compared with the other two cases. We thus confirm the mass ratio at 1
1, deformation occurs more significantly when compared with the other two cases. We thus confirm the mass ratio at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and regulate the height of the micro-domes for sensitivity optimization (Fig. 3d). Via changing the heights from 300 μm, 500 μm, to 650 μm, the plots indicate that an increased height is positive for sensitivity enhancement, which is consistent with the analysis in Section S1 (ESI†) and the governing formula discussed above. Via changing the heights, the sensing performance of micro-domes in other mass ratios were evaluated (Fig. S20, ESI†). The plots indicate a similar sensing behavior for the micro-domes in different moduli, where a higher micro-dome normally renders a higher sensitivity with linear sensing capability (see Table S2 for detailed parameters, ESI†). Combining the results of the above two strategies, Fig. 3e statistically summarizes the dependence of sensitivity on the elastic modulus of the materials and the heights of the circular micro-domes. The micro-dome based sensor with mEcoflex
1 and regulate the height of the micro-domes for sensitivity optimization (Fig. 3d). Via changing the heights from 300 μm, 500 μm, to 650 μm, the plots indicate that an increased height is positive for sensitivity enhancement, which is consistent with the analysis in Section S1 (ESI†) and the governing formula discussed above. Via changing the heights, the sensing performance of micro-domes in other mass ratios were evaluated (Fig. S20, ESI†). The plots indicate a similar sensing behavior for the micro-domes in different moduli, where a higher micro-dome normally renders a higher sensitivity with linear sensing capability (see Table S2 for detailed parameters, ESI†). Combining the results of the above two strategies, Fig. 3e statistically summarizes the dependence of sensitivity on the elastic modulus of the materials and the heights of the circular micro-domes. The micro-dome based sensor with mEcoflex![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mPDMS = 1
mPDMS = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1@h = 500 μm exhibits superior comprehensive performance for static pressure perception. On the one hand, the material of mEcoflex
1@h = 500 μm exhibits superior comprehensive performance for static pressure perception. On the one hand, the material of mEcoflex![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mPDMS at 1
mPDMS at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 has a lower elastic modulus (E = 2.29 MPa) to obtain a high sensitivity of 96.6 kPa−1 (S > 50 kPa−1) that is sufficient to detect the subtle biosignals.50 More importantly, multiple tests were performed on the device to evaluate the sensing repeatability (Fig. S21, ESI†). The consistent sensing plots confirm the excellent elasticity and recovery of the micro-structures when exposed to periodical pressure application and revocation. Under different pressures, the output current signal of the device is recorded for one minute to evaluate the stability performance (Fig. 3f). The results indicate that the device can stably reflect the applied mechanical stimuli in the low or high-pressure range. Note that even though pure Ecoflex with micro-dome height of 650 μm exhibits the highest sensitivity, we did not select such specific composition for the subsequent investigation. Firstly, recovering the hysteresis of pure Ecoflex under high pressure limits the applications as indicated in Fig. S18 (ESI†). Furthermore, a relatively smaller height of 500 μm (instead of 650 μm) was selected for study to avoid the excessively increasing height of the micro-dome structure.
1 has a lower elastic modulus (E = 2.29 MPa) to obtain a high sensitivity of 96.6 kPa−1 (S > 50 kPa−1) that is sufficient to detect the subtle biosignals.50 More importantly, multiple tests were performed on the device to evaluate the sensing repeatability (Fig. S21, ESI†). The consistent sensing plots confirm the excellent elasticity and recovery of the micro-structures when exposed to periodical pressure application and revocation. Under different pressures, the output current signal of the device is recorded for one minute to evaluate the stability performance (Fig. 3f). The results indicate that the device can stably reflect the applied mechanical stimuli in the low or high-pressure range. Note that even though pure Ecoflex with micro-dome height of 650 μm exhibits the highest sensitivity, we did not select such specific composition for the subsequent investigation. Firstly, recovering the hysteresis of pure Ecoflex under high pressure limits the applications as indicated in Fig. S18 (ESI†). Furthermore, a relatively smaller height of 500 μm (instead of 650 μm) was selected for study to avoid the excessively increasing height of the micro-dome structure.
To investigate the mechanical durability of the MDA device, cyclic tests were performed over 5000 times with a pressure of ∼108 kPa (Fig. 3g). The inset illustrates the response of the device to periodical pressure at different time intervals. It can be observed that after undergoing 5000 loading–unloading tests, the relative current variation remains essentially unchanged, indicating the potential for long-term use of the piezoresistive sensing component. To quantitatively evaluate the response behavior, we applied mechanical input at a loading speed of 30 mm s−1 to the MDA. The response time and recovery time of MDA are about 180 ms and 47 ms respectively (Fig. S22, ESI†). We further characterized the hysteresis via monitoring the real-time current variation when the MDA was exposed to a pressure loading up to 110 kPa and then continuously released (Fig. S23, ESI†). As presented in the curve, the rapid recovery is attributed by the outstanding durability and elastic properties of Ecoflex/PDMS at this mass ratio, allowing the dome-shaped structure to endure prolonged mechanical deformation. To demonstrate the application for sensitive monitoring of physiological signals, the device was fixed on the human wrist to detect the pulse wave of the radial artery as shown in Fig. 3h. The inset provides an enlarged view of an individual pulse wave, which clearly distinguishes the characteristic signals such as percussion (P1), tidal (P2), and diastolic (P3) peaks. For flexible sensors based on micro-structures for pressure detection, linearity is easily lost over a wide pressure range because of the saturated deformation when the applied pressure was continuously increased.51 One possible solution to relieve this effect is by introducing the multi-layer structures with electrical conductivity gradients on the micro-structures to maintain the sensing performance (sensitivity and linearity).50 In contrast to the surface-conductive design, the micro-domes in this work are conductive throughout the whole body, providing the synergistic effect of changes in bulk resistance and elastic properties to resolve the sensitivity decay during pressure loading.
2.3. Tactile perception of fully bionic E-skin
Thanks to the three-dimensional configuration, the fully biomimetic E-skin is capable of perceiving the normal static pressure, normal dynamic pressure, and the in-plane dynamic sliding inputs. Fig. 4a shows the optical images of the assembled E-skin, which is composed of the MMCA and MDA components to mimic the sensory function of human skin to mechanical inputs. Based on the dual-mode sensing mechanism of electromagnetic induction (MMCA) and piezoresistive effect (MDA), the device can imitate the response of SA and RA receptors, enabling simultaneous monitoring of both dynamic and static external mechanical stimuli. The MMCA component operates in a small pressure range and can respond to high frequency stimuli, whereas the MDA component has a broader pressure-responsive range at low frequency. To demonstrate the complementary effects of MMCA and MDA components on the sensing characteristics, we conducted simulation to analyze the stress distribution under typical normal pressures from 1 kPa to 50 kPa (Fig. 4b and Fig. S24, ESI†). The simulation results show that with gradually increased normal pressures, the mechanical stimuli were continuously transmitted from flexible micro-cilia to the micro-domes (see the discussion on the simulation model in Section S2, ESI†). Note that for the displacement simulation of the micro-cilia under normal and tangential stimuli, the results are provided in Fig. S5 (ESI†) and here we mainly focused on the stress distribution. To evaluate the tactile response of the assembly, we applied the electrical meters and connected to the interdigital electrode and conductive coil so that signals from the MDA and MMCA components could be recorded simultaneously in real-time. As presented in Fig. 4c and Fig. S25 (ESI†), the MDA and MMCA components exhibit completely synchronized real-time responses when the excitation frequencies of the periodical pressures were from 1 Hz to 5 Hz at 800 Pa. However, with increased excitation frequency to 10 Hz, 20 Hz, and 30 Hz under the same pressure magnitude, the voltage output by the MMCA component could accurately reflect the stimuli, while the current variations from the MDA branch exhibited obvious signal distortion. The results indicate that the high-aspect-ratio micro-cilia play an important role in compensating for the shortcomings of the micro-domes, which serves to accurately respond to high-frequency vibration signals as the RA receptors in the human skin system. Fig. 4d summarizes the response time of the E-skin which is related to the applied pressure. Thanks to the excellent elastic performance of the micro-cilia, the response time of the E-skin was ∼10 ms (frequency of 100 Hz as shown in Fig. 2i), which is generally higher than that of the traditional piezoresistive pressure sensors.39,51–54 We further evaluated the sensing performance of the device via applying different magnitudes of the normal pressure to the dual-mode FBE-skin (Fig. 4e). Both the current variation from the micro-domes and the induced voltage peaks from the micro-cilia are consistent with the strength of the applied mechanical inputs. Although the top-down artificial configuration enables the dual-mode sensing, we noticed that the integrated skin somehow reduced the sensitivity of the MDA in low-pressure range. This phenomenon is attributed by the buffered pressure distributions from the elastic micro-cilia layer on-top (see Fig. S26, ESI†).When static mechanical stimuli are applied to the human skin, the response characteristics of RA receptors enables a transient voltage spike related to the moment of touch, while the SA receptors record the maintained signal to accurately reflect the existent static inputs.20,55 These signals from RA and SA receptors are delivered individually via the neural system, and combined as hierarchical information for the brain to comprehensively cognize the mechanical input. Regarding this, we applied the periodical normal pressure (4 kPa) to the FBE-skin with long and short durations to evaluate the recognition function of the bionic system (Fig. 4f). As shown in Fig. 4g and h, the real-time voltage and current profiles from the FBE-skin fully mimic the response characteristics of the RA and SA mechanoreceptors, providing the capability of differentiating pressure loads without signal crosstalk. For the pressure with longer duration (12 s, Fig. 4g), current signals were maintained during the application of static pressure, while the voltage spikes occurred only at the moment of touch and release. For the pressure with shorter duration (0.5 s, Fig. 4h), both the current and voltage branches exhibited the spike profile to accurately reflect the behavior of fast mechanical release. The consistency of the signals after multiple contact and release demonstrates the reliable sensing of the FBE-skin, which integrates the function of RA and SA mechanoreceptors via two distinct mechanical-to-electrical principles (Video S1, ESI†).
The above results show that the direct contact with the FBE-skin can generate dual-mode electrical signals from the MMCA and MDA components, respectively. Apart from that, the 3D architecture of the micro-cilia allows the sensitive perception of mechanical input without involvement of the underlying micro-dome component. Attributed by the integrated MMCA layer, the FBE-skin can be applied to situations, e.g. in-plane tangential force sensing or non-contact airflow detection, which involve a close-range perception of the superficial surface (Fig. 4i). Such subtle inputs cannot obviously induce sufficient compression of the micro-domes with current variation, but the elastic micro-cilia can be easily deformed to reflect the mechanical inputs via voltage spikes thanks to the electromagnetic induction. The dual-channel signal acquisition platform is shown in Fig. S27 (ESI†), which can apply directional forces to the FBE-skin with real-time signal collections. Fig. 4j demonstrates the real-time response of the FBE-skin when simulating horizontal sliding of a finger across the micro-cilia layer, with the entire process recorded in Video S2 (ESI†). It can be observed that the back-and-forth finger sliding caused the micro-cilia deformation and instantaneous voltage spikes, whereas the micro-dome branch remained inactive (ΔI ≈ 0) because of the negligible morphological deformations. Fig. 4k displays the current and voltage signals under non-contact airflow in both vertical and horizontal directions (see Video S3 for the real-time record, ESI†). The optical images clearly demonstrate that the flexible micro-cilia were bent by the mechanical constraint from the airflow. In these scenarios, only the MMCA sensing components exhibit a noticeable response in the format of induced voltage spikes. However, the underlying micro-domes remained inert, and no obvious current variations were measured during the application of continuous airflow. From this perspective, the integration of micro-cilia arrays on top provides the FBE-skin system the capability of delivering warning prior to the direct contact with the underlying skin layer. Even though the hairy layer has been explored as an effective tool for mechanical perception,56 the magnetized MMCA component alternatively relies on the variation of magnetic flux and induced electric signals to reflect the input stimuli. Compared with the resistive hairy skin, the magnetized functionality allows the MMCA to incorporate conductive micro-domes in the top-down architecture to fully mimic the human skin. Note that when exposed to normal or tangential forces, the MMCA would produce similar electrical signals (voltage) based on the principle of electromagnetic induction. It is thus difficult to distinguish the type of the stimuli only through the response of MMCA. However, as demonstrated in Fig. 4g and j, the combination with MDA allows the E-skin to distinguish the inputs through the dual-channel signals. For example, normal force can generally result in voltage production in MMCA and current variation in the MDA branch, while the tangential input would only induce voltage pulses from MMCA in the dynamic process. We also have to mention that the current design of the FBE-skin is unable to identify arbitrary force angles, which might be achieved by elaborating the morphologies of the MMCA or introducing the effect of magnetized orientations in the future. Overall, apart from the template-free methodology that could flexibly control the MMCA morphology, the distinct sensing mechanism endows the FBE-skin to perceive different mechanical stimuli with identifiable signals as demonstrated below.
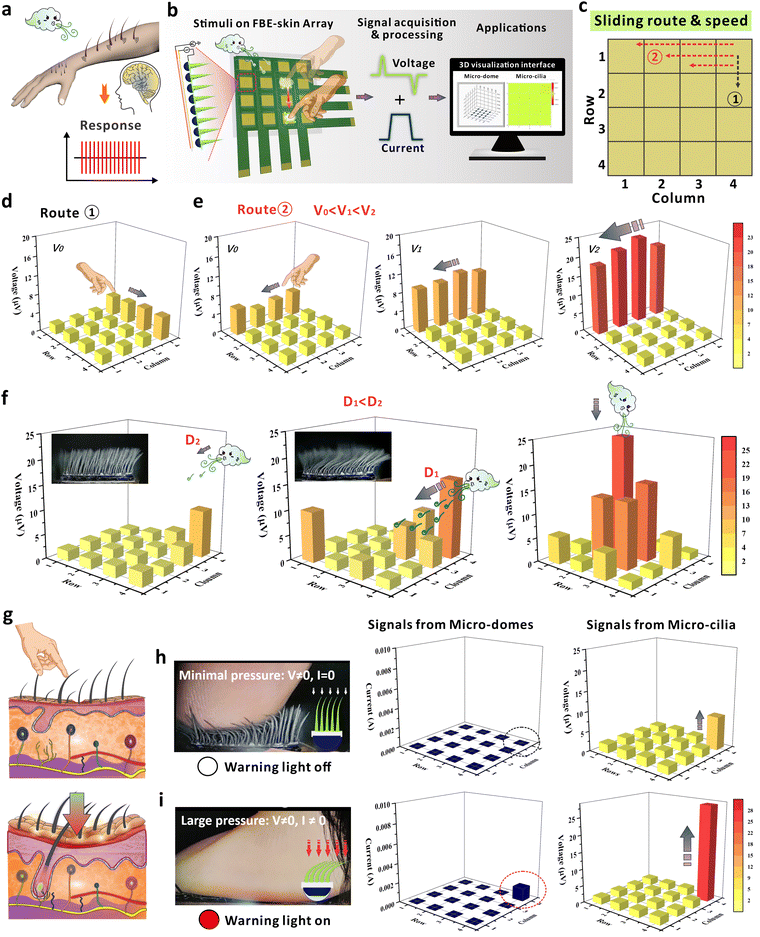
2.4. FBE-skin array for 3D visualization of biomechanical signal decoding
The neural system of the human body is capable of perceiving the tangential mechanical stimuli that slide across the skin surface. Especially, the distributed hairy layer on human skin allows the identification of input information that is not directly acting on the dermis layer. This sensing capability not only reflects the input strength, but also enables the receptors to recognize associated information such as the sliding direction and moving velocity of the dynamic input (Fig. 5a).31,57 Inspired by this, we developed a corresponding 3D visualization system to decode the biomechanical loading and achieve the digitization of human sensation through in-depth signal analysis (Fig. 5b). In this customized system, the 4 × 4 array is composed of 16 FBE-skin units, and the 3D visualization interface can simultaneously collect and display the dual-mode signals from voltage and current branches (Fig. S28 and S29, ESI†). Owing to the excellent deformation capability of the MMCA, the branch of MDA at the bottom could still maintain a high sensitivity for pressure perception as discussed in Fig. S26 (ESI†). As demonstrated in Fig. 5c–e, one primary functionality of this system is to mimic the human skin that can discriminate the route and speed of sliding mechanical stimuli on the epidermis. Without contact on the dermis (micro-domes), the deformation of micro-cilia resulted in obvious voltage signals so that the proximal inputs could be perceived. When the human finger slid across Column 4 (Route 1), the voltage in four related rows was generated in sequence to reflect the sliding direction (Fig. 5d and Video S4, ESI†). The finger sliding was further defined at three different speeds, V0, V1, and V2, following Route 2 from Column 4 to Column 1 at the first Row to compare the intensity of induced voltage signals. Based on the mapping relationship between the voltages and color maps, the sliding speed relationship of V0 < V1 < V2 was accurately determined (Fig. 5e). The visualization system indicates that the FBE-skin array can electrically monitor the motion trajectories of mechanical input that is tangential to the surface, even though the input does not cause direct changes in the underlying piezoresistive layer. Furthermore, the sliding speed could also be directly observed from the voltage map thanks to the principle of electromagnetic induction.Owing to the excellent flexibility of the micro-cilia, the FBE-skin array can also sense and decode the spatial distribution of airflow, e.g. the source location or flow magnitude. The perception process of dynamic airflow was recorded in Video S5 (ESI†), and the reconstructed spatial distribution of airflow based on the voltage profiles is presented in Fig. 5f. From left to right panels, the figures illustrate the airflow from different directions and intensities, including the lateral airflow source at distances of D1 and D2 (D1 < D2), and a downward airflow source situated directly above. Herein, the distance difference was applied to represent the airflow intensity, e.g. a larger distance brings a smaller mechanical intensity to the micro-cilia. Compared with the sensor unit under a larger distance D2, the airflow intensity at distance D1 is stronger. Consequently, the areas with darker colors indicate a greater airflow intensity, which is attributed by the more obvious deformation of the micro-cilia as shown in the optical images. Additionally, the orientation of the airflow can be determined by the positions where the colors exhibit concentrated changes. For the airflow from the normal direction, the dynamic pressure was mainly distributed in the central FBE-skin and the distribution of the pressure inputs can be accurately re-established from the 3D visualization system. As recorded in the video, the subtle airflow could only deform the micro-cilia, and the current signals from the micro-domes remained inactive during the sensing process. Benefiting from the 3D architecture and the sensing characteristics of MMCA components, the FBE-skin array can perceive the minimal stress before the finger contacts the skin, demonstrating the potential for predict possible injury on the skin layer (Fig. 5g). Combined with the MDA structures, the FBE-skin can detect minimal, moderate, and large pressure loads with distinct outputs. To simulate the ability of the 3D visual decoding system based on the FBE-skin array, a corresponding set of 16 signal lights was integrated into the array device to issue warning signals for harmful touches (Video S6, ESI†). As shown in Fig. 5h, the small pressure deformed the micro-cilia while the micro-domes were unaffected. This caused an induced voltage (non-zero) while the warning light was off to represent that the fingertip only touched the outer hairy layer with a small pressure. The inset also indicates the compressive behavior of each component under such a pressure range. At moderate pressure, the micro-cilia and the micro-domes deformed to produce both the voltage and current signals (Fig. S30, ESI†). A green warning light is on to indicate the possibility of harmful touch to the skin surface, which is different from the proximal touch that only causes the micro-cilia deformation. When the dynamic force was further increased, a more obvious deformation was generated both in the micro-cilia and the micro-domes (Fig. 5i). For this case, the signals in current branch surpassed the pre-defined threshold and a red warning light is on to urge necessary protection so that injury can be avoided. From above, the demonstrations of FBE-skin array successfully simulate the capabilities of the human neural system for proximal tactile perception, issue of timely warnings for potentially harmful touches, and sensitive detection of the spatial airflow in contact-less mode. Even though the design of conductive MDA has been raised as a flexible sensing element in previous studies, the mono-resistive signals cannot well mimic the function of human skin that covers a broad mechanical spectrum. However, the cooperation of MMCA and MDA in this study can respond from normal to tangential force inputs with broad mechanical frequencies and magnitudes, which is shown by the complementary behavior from the two distinct branches. The integration of micro-cilia and micro-domes not only mimic the functionality of hairy structures on human skin, but also provide a solution to couple the RA and SA receptors in the intelligent sensing system.
3. Conclusions
In this work, we developed FBE-skin based on the top-down assembled structure of a bionic hair–epidermis–dermis–subcutaneous tissue for multidimensional tactile perception. By fully utilizing the design concept of human skin, the MMCA, MDA structures, and flexible electrodes are manufactured to replicate the biological hair layer, epidermis–dermis, and RA/SA receptors located subcutaneously. The sensing performance of each component in the top-down configuration was further improved by optimizing the morphological and magnetic/electrical properties of the micro-cilia and the conductive micro-domes. Based on this, the E-skin exhibits outstanding features including high sensitivity to small pressures, real-time response to high-frequency mechanical vibrations, low detection limits, in-plane shear force/out-of-plane normal force perception, etc. The dual-channel signals were completely decoupled in a cross-talk-free behavior, which provides detailed information about the mechanical stimuli in a distinguishable format. Upon this, the single FBE-skin device was arrayed and constructed as a visualization system to digitize human sensations, showcasing the potentials for sliding speed detection, providing warnings for close-range stimuli before direct skin contact, subtle airflow perception, etc. Along with the long-term durability and low hysteresis, we expect that the multifunctional and wearable FBE-skin can be promising for future application in the fields of prosthetics, human–machine interactions, and intelligent tactile sensing.4. Methods
4.1. Fabrication process of the flexible MDA
Firstly, to make the mold for the dome-shaped pattern array, the mechanical engraving machine was used with a ball end mill to engrave a 7 × 7 dome-shaped pattern array on a plastic substrate. The total area of the pattern is 1 cm × 1 cm. The diameter of a single dome-shaped structure is 1000 μm, and the height is 500 μm. Then, 2.3 g PDMS (Sylgard 184, Dow Corning, USA) and 2.3 g Ecoflex™ 00-10 (Smooth-On, Inc., USA) gels were uniformly dispersed in cyclohexane solvent (20 mL, Aladdin, China). The PDMS was composed of the base elastomer and curing agent in a typical ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, while Ecoflex was composed of a base elastomer and the crosslinker in a mass ratio of 1
1, while Ecoflex was composed of a base elastomer and the crosslinker in a mass ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. After adding 0.4 g carbon nanotubes (CNT, National Technology, China) and ultra-sonicated for 10 min, the mixed solution was placed on a magnetic stirrer and stirred at room temperature to evaporate the cyclohexane solvent, resulting in a viscous CNT/PDMS/Ecoflex mixture. The mixture was then poured into the mold and cured (80 °C, 30 min) to obtain a conductive dome-shaped array structure. Subsequently, pure PDMS (base and curing agent at mass ratio of 10
1. After adding 0.4 g carbon nanotubes (CNT, National Technology, China) and ultra-sonicated for 10 min, the mixed solution was placed on a magnetic stirrer and stirred at room temperature to evaporate the cyclohexane solvent, resulting in a viscous CNT/PDMS/Ecoflex mixture. The mixture was then poured into the mold and cured (80 °C, 30 min) to obtain a conductive dome-shaped array structure. Subsequently, pure PDMS (base and curing agent at mass ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was spin-coated (900 rpm, 10 s) on the dome-shaped array structure to achieve a transparent thin film. Further curing (80 °C, 20 min) resulted in the final product of the flexible MDA layer, which was composed of a conductive micro-dome array and a transparent pure PDMS substrate.
1) was spin-coated (900 rpm, 10 s) on the dome-shaped array structure to achieve a transparent thin film. Further curing (80 °C, 20 min) resulted in the final product of the flexible MDA layer, which was composed of a conductive micro-dome array and a transparent pure PDMS substrate.
4.2. Preparation of the flexible MMCA
First, the NdFeB particles (Magnequench, China), PDMS gel, and curing agent were mixed in the mass ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, followed by a de-gassing process to remove the bubbles. The mixture was then spin-coated (900 rpm for 10 s) onto the MDA transparent substrate to prepare the base layer for a micro-cilia array. Next, carbonyl iron particles (CIP, 3 g, Sigma-Aldrich, USA) and NdFeB particles (3 g) were uniformly dispersed in 25 mL cyclohexane (Aladdin, China), followed by vortex mixing and ultrasonic treatment (10 min). PDMS gel (3 g) and curing agent (1 g) were then added to the CIP/NdFeB/cyclohexane mixture, followed by the same mixing process to ensure the uniform distribution. The mass ratio of PDMS gel to curing agent was fixed at 3
1, followed by a de-gassing process to remove the bubbles. The mixture was then spin-coated (900 rpm for 10 s) onto the MDA transparent substrate to prepare the base layer for a micro-cilia array. Next, carbonyl iron particles (CIP, 3 g, Sigma-Aldrich, USA) and NdFeB particles (3 g) were uniformly dispersed in 25 mL cyclohexane (Aladdin, China), followed by vortex mixing and ultrasonic treatment (10 min). PDMS gel (3 g) and curing agent (1 g) were then added to the CIP/NdFeB/cyclohexane mixture, followed by the same mixing process to ensure the uniform distribution. The mass ratio of PDMS gel to curing agent was fixed at 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 based on our previous studies,58 while the content of NdFeB/CIP was varied in this study to optimize the micro-cilia morphology and the device performance. Subsequently, ∼25 mL solution of the magnetic composites was loaded into a spraying tool (outlet diameter of 0.5 mm) and sprayed onto the uncured base layer (spray distance ∼15 cm). Before each spraying, the loadings were shaken for ∼30 s to maintain the uniform dispersion. The sprayed liquid droplets accumulated along the magnetic field lines, forming preliminary micro-cilia-like microstructures through a combinational effect of gravity, surface tension, and the forces provided by the underlying permanent NdFeB magnet. During this process, the sample was placed on the platform with a vertical distance of 3 cm to the permanent magnet surface. To obtain a robust micro-cilia array, the entire device was cured in an oven (60 °C) for 8 hours. Finally, the peeled off micro-cilia array was placed in a commercial magnetizer (MA-3030, Jiu Juok, China) with a constant magnetic field of 3 T to obtain the finalized MMCA.
1 based on our previous studies,58 while the content of NdFeB/CIP was varied in this study to optimize the micro-cilia morphology and the device performance. Subsequently, ∼25 mL solution of the magnetic composites was loaded into a spraying tool (outlet diameter of 0.5 mm) and sprayed onto the uncured base layer (spray distance ∼15 cm). Before each spraying, the loadings were shaken for ∼30 s to maintain the uniform dispersion. The sprayed liquid droplets accumulated along the magnetic field lines, forming preliminary micro-cilia-like microstructures through a combinational effect of gravity, surface tension, and the forces provided by the underlying permanent NdFeB magnet. During this process, the sample was placed on the platform with a vertical distance of 3 cm to the permanent magnet surface. To obtain a robust micro-cilia array, the entire device was cured in an oven (60 °C) for 8 hours. Finally, the peeled off micro-cilia array was placed in a commercial magnetizer (MA-3030, Jiu Juok, China) with a constant magnetic field of 3 T to obtain the finalized MMCA.
4.3. Preparation of the flexible electrodes
The double-layer flexible printed circuit board (FPCB) was fabricated by laser etching on a PI-copper-clad substrate using an LPKF ProtoLaser U4 machine. The top layer of the FPCB is the interdigitated electrode (finger width of 1.4 mm, spacing of 100 μm, and area of 1.2 cm × 1 cm). The bottom layer consists of a copper coil electrode with 30 turns (line width of 70 μm and spacing of 15 μm, area of 1.5 cm × 1 cm). It should be noted that the electrodes in the array utilize a 3-layer FPCB, where the middle layer and bottom layer form the double-layered copper coil electrode.4.4. Assembly of fully bionic E-skin
The MMCA layer was stacked on top of the MDA layer, with a thin PDMS film as the connection layer via a spin-coating process (900 rpm, 5 s). Then, it was placed on the flexible double-layer electrode and secured using double-sided conductive copper tape to complete the assembly of the FBE-skin.4.5. Fabrication of flexible perception array
The FPCB consisting of 16 three-layer electrodes was designed, with an area of 6.5 cm × 6.5 cm. The spacing between adjacent electrodes in each row is 6 mm, and the spacing between adjacent electrodes in each column is 3 mm. By placing each full bionic E-skin at the corresponding central position of the electrodes, a flexible perception array (4 × 4 matrix) was formed.4.6. Characterization and measurements
Scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDX) and optical images were obtained by Sigma FE-SEM (Zeiss, Germany), and optical microscope (Olympus, Japan). Normal pressure loading–releasing were controlled via a micro-displacement platform (Zolix, TSA50, China) and a motion controller (Zolix, MC600, China), while the dynamic tangential force was controlled using a horizontal slider (Zolix, MAR 100-90, China). Pressure was measured using a high-precision balance (LiChen, YP200001D, China) with a wide range. Output voltage was recorded using a Keysight 34465A digital multimeter, and output current was obtained using a Keysight B2902B source meter. High-frequency signals were generated using a generator (Tektronix, AFG 31000, USA) and amplified using a power amplifier (Koshin Instruments, KSI-758PA100). The loading–unloading of high-frequency normal pressure was dynamically controlled using a commercial vibrator (Sinocera Piezotronics Inc., JZK-2, China) with frequencies up to 100 Hz. A low-noise current preamplifier (SR-570, SRS, USA) with high sampling rate was applied to precisely collect the induced current signals in the coil electrode. The voltage and current signals for the arrayed device were recorded using a Keysight DAQ970A data acquisition unit equipped with a Keysight DAQM900A multiplexer module (40 parallel channels). Wearable demonstrations involving external stimulation through finger simulation were conducted by a research volunteer, and the consent was obtained before the experiments.Author contributions
Fengming Hu: conceptualization, methodology, software, formal analysis, investigation, data curation, writing – original draft, writing – review & editing, visualization. Qian Zhou: methodology & investigation. Ruolin Liu: methodology. Yanfei Zhu: software & visualization. Yuanzhe Liang: methodology. Dan Fang: investigation. Bing Ji: conceptualization & methodology. Zhiming Chen: resources and supervision. Jianyi Luo: resources, writing editing, supervision, funding acquisition. Bingpu Zhou: conceptualization, writing review & editing, supervision, project administration, funding acquisition.Data availability
The data supporting this article have been included as part of the ESI.†Video S1–S6: Voltage signals of the dual-mode sensor when exposed to mechanical presses with/without duration; response of the dual-mode sensor to the tangential force applied by finger sliding; records of MMCA deformation of FBE-skin under lateral and normal airflow; demonstration of the FBE-skin array for recognition of different sliding speeds and routes; demonstration of the FBE-skin array for perception of spatial airflow distributions; demonstration of the FBE-skin array's proximity perception and simulation of human-skin by issuing a warning signal to indicate harmful touch. Fig. S1–S30: Fabrication process of the FBE-skin; SEM image and EDX mapping results of the MMCA layer; illustration and simulation of the variation in the magnetic field during MMCA bending; schematic diagram of the preparation setup and dimensional adjustability of MMCA; simulated displacements for different MMCAs under the normal and tangential pressures; induced voltages for MMCAs with different aspect ratios under different pressures; the relationship between the micro-cilia deformation and induced voltages with different aspect ratios; effect of the micro-cilia number on the sensing performance; real-time response of MMCA devices to ultra-low pressures; influence of pressure loading frequency on MMCA voltage signals; long-term stability of MMCA under tangential stimulation; repeatability test of MMCA morphology and sensing performance; schematic diagrams of the flexible double-layered electrode; optical images and resistance stability of the double-layered electrode; equivalent circuit and optical image of the MDA device; statistics and measurement of resistivity of PDMS/CNT composites with different CNT contents; elastic moduli of PDMS/Ecoflex composites with different mass ratios; typical optical image of MDA deformation processes with different elastic modulus; effect of height on the sensitivity of dome devices composed of pure PDMS, mEcoflex![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mPDMS =1
mPDMS =1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and pure Ecoflex; repeatability of MDA device sensitivity; hysteretic behavior of the MDA component; simulated mechanical distribution of the FBE-skin under normal pressures of 10 kPa and 50 kPa; the FBE-skin with a pressure of 800 Pa under different frequencies; comparison of MDA and MMCA compressed deformation of FEB-skin under smaller and larger pressures; optical image of the FBE-skin test system; image of 3D visualization decoding system and the experimental platform; 3D visualization interface for FBE-skin array; schematic diagram of the FBE-skin deformation under a moderate pressure; equivalent diagram of the total contact resistance between the micro-dome and the interdigital electrode; top view and side view of microdome structure deformation under different pressure loads. Tables S1 and S2: Statistics of the aspect ratio, device sensitivity, and detection limit of micro-cilia arrays; summarized sensitivity and sensing linearity of the MDA device with varying heights of the micro-domes. Supplementary Notes S1 and S2: Theoretical analysis of the piezoresistive component; COMSOL simulation for mechanical properties of the FBE-skin.
2 and pure Ecoflex; repeatability of MDA device sensitivity; hysteretic behavior of the MDA component; simulated mechanical distribution of the FBE-skin under normal pressures of 10 kPa and 50 kPa; the FBE-skin with a pressure of 800 Pa under different frequencies; comparison of MDA and MMCA compressed deformation of FEB-skin under smaller and larger pressures; optical image of the FBE-skin test system; image of 3D visualization decoding system and the experimental platform; 3D visualization interface for FBE-skin array; schematic diagram of the FBE-skin deformation under a moderate pressure; equivalent diagram of the total contact resistance between the micro-dome and the interdigital electrode; top view and side view of microdome structure deformation under different pressure loads. Tables S1 and S2: Statistics of the aspect ratio, device sensitivity, and detection limit of micro-cilia arrays; summarized sensitivity and sensing linearity of the MDA device with varying heights of the micro-domes. Supplementary Notes S1 and S2: Theoretical analysis of the piezoresistive component; COMSOL simulation for mechanical properties of the FBE-skin.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors appreciate the support from the Science and Technology Development Fund, Macao S.A.R. (006/2022/ALC and 0057/2023/RIB2), the Guangdong Science and Technology Department (2022A0505030024), and the Innovation and Strong School Engineering Fund of Guangdong Province (2020ZDZX2022 and 2021ZDJS094).References
- Y. Gao, L. Yu, J. C. Yeo and C. T. Lim, Adv. Mater., 2019, 32, 1902133 CrossRef.
- X. Cui, Y. Jiang, L. Hu, M. Cao, H. Xie, X. Zhang, F. Huang, Z. Xu and Y. Zhu, Adv. Mater. Technol., 2022, 8, 2200609 CrossRef.
- S. Pyo, J. Lee, K. Bae, S. Sim and J. Kim, Adv. Mater., 2021, 33, 2205902 Search PubMed.
- W. W. Lee, Y. J. Tan, H. Yao, S. Li, H. H. See, M. Hon, K. M. Ng, B. Xiong, J. S. Ho and B. C.-K. Tee, Sci. Rob., 2019, 4, eaax2198 CrossRef.
- J. C. Yang, J. Mun, S. Y. Kwon, S. Park, Z. Bao and S. Park, Adv. Mater., 2019, 31, 1904765 CAS.
- Y. Wang, S. Lee, T. Yokota, H. Wang, Z. Jiang, J. Wang, M. Koizumi and T. A. Someya, Sci. Adv., 2020, 6, eabb7043 CAS.
- X. Yang, L. Li, S. Wang, Q. Lu, Y. Bai, F. Sun, T. Li, Y. Li, Z. Wang, Y. Zhao, Y. Shi and T. Zhang, Adv. Electron. Mater., 2020, 6, 200006 Search PubMed.
- S. Lee, F. Sae, F. A. Hassani, T. Yokota, O. M. G. Nayeem, Y. Wang, R. Leib, G. Cheng, D. W. Franklin and T. Someya, Science, 2020, 370, 966–970 CrossRef CAS.
- M. Wang, Y. Luo, T. Wang, C. Wan, L. Pan, S. Pan, K. He, A. Neo and X. Chen, Adv. Mater., 2020, 33, 2003014 CrossRef.
- X. Hu, M. Wu, L. Che, J. Huang, H. Li, Z. Liu, M. Li, D. Ye, Z. Yang, X. Wang, Z. Xie and J. Liu, Small, 2023, 19, 2208015 CrossRef CAS.
- N. Bai, L. Wang, Y. Xue, Y. Wang, X. Hou, G. Li, Y. Zhang, M. Cai, L. Zhao, F. Guan, X. Wei and C. F. Guo, ACS Nano, 2022, 16, 4338–4347 CrossRef CAS PubMed.
- Y. Qiu, C. Wang, X. Lu, H. Wu, X. Ma, J. Hu, H. Qi, Y. Tian, Z. Zhang, G. Bao, H. Chai, J. Song and A. Liu, Adv. Funct. Mater., 2021, 32, 2110296 CrossRef.
- X. Wei, B. Wang, Z. Wu and Z. L. Wang, Adv. Mater., 2022, 34, 2203073 CrossRef CAS.
- C. Shang, Q. Xu, N. Liang, J. Zhang, L. Li and Z. Peng, npj Flexible Electron., 2023, 7, 19 CrossRef CAS.
- L. Chen, Y. Xu, Y. Liu, J. Wang, J. Chen, X. Chang and Y. Zhu, ACS Appl. Mater. Interfaces, 2023, 15, 24923–24932 CrossRef CAS.
- L. Lu, N. Zhao, J. Liu and B. Yang, J. Mater. Chem. C, 2021, 9, 9309–9318 RSC.
- J. Luo, F. Liu, A. Yin, X. Qi, J. Liu, Z. Ren, S. Zhou, Y. Wang, Y. Ye, Q. Ma, J. Zhu, K. Li, C. Zhang, W. Zhao, S. Yu and J. Wei, Carbohydr. Polym., 2023, 315, 120983 CrossRef CAS PubMed.
- Y. Yin, Y. Wang, H. Li, J. Xu, C. Zhang, X. Li, J. Cao, H. Feng and G. Zhu, Chem. Eng. J., 2022, 430, 133158 CrossRef CAS.
- Y. E. Shin, Y. J. Park, S. K. Ghosh, Y. Lee, J. Park and H. Ko, Adv. Sci., 2022, 9, 2105423 CrossRef CAS PubMed.
- H. Qiao, S. Sun and P. Wu, Adv. Mater., 2023, 35, 2300593 CrossRef CAS PubMed.
- J. Yin, R. Hinchet, H. Shea and C. Majidi, Adv. Funct. Mater., 2020, 31, 2007428 CrossRef.
- Y. Pang, K. Zhang, Z. Yang, S. Jiang, Z. Ju, Y. Li, X. Wang, D. Wang, M. Jian, Y. Zhang, R. Liang, H. Tian, Y. Yang and T.-L. Ren, ACS Nano, 2018, 12, 2346–2354 CrossRef CAS PubMed.
- G. Li, D. Chen, C. Li, W. Liu and H. Liu, Adv. Sci., 2020, 7, 2000154 CrossRef CAS PubMed.
- C. M. Boutry, M. Negre, M. Jorda, O. Vardoulis, A. Chortos, O. Khatib and Z. Bao, Sci. Rob., 2018, 3, eaau6914 CrossRef PubMed.
- M.-J. Yin, Z. Yin, Y. Zhang, Q. Zheng and A. P. Zhang, Nano Energy, 2019, 58, 96–104 CrossRef CAS.
- J. Park, J. Kim, J. Hong, H. Lee, Y. Lee, S. Cho, S.-W. Kim, J. J. Kim, S. Y. Kim and H. Ko, NPG Asia Mater., 2018, 10, 163–176 CAS.
- J. Park, M. Kim, Y. Lee, H. S. Lee and H. Ko, Sci. Adv., 2015, 1, e1500661 CrossRef PubMed.
- T. Yang, W. Deng, X. Chu, X. Wang, Y. Hu, X. Fan, J. Song, Y. Gao, B. Zhang, G. Tian, D. Xiong, S. Zhong, L. Tang, Y. Hu and W. Yang, ACS Nano, 2021, 15, 11555–11563 CrossRef CAS PubMed.
- K. Chang, L. Li, C. Zhang, P. Ma, W. Dong, Y. Huang and T. Liu, Composites, Part A, 2021, 151, 106671 CrossRef CAS.
- S. Chun, I. Y. Choi, W. Son, J. Jung, S. Lee, H. S. Kim, C. Pang, W. Park and J. K. Kim, ACS Energy Lett., 2019, 4, 1748–1754 CrossRef CAS.
- S. G. Lechner and G. R. Lewin, Physiology, 2013, 28, 142–150 CrossRef CAS.
- H. Niu, H. Li, S. Gao, Y. Li, X. Wei, Y. Chen, W. Yue, W. Zhou and G. Shen, Adv. Mater., 2022, 34, 2202622 CrossRef CAS PubMed.
- P. Glass, A. Shar, C. Pemberton, E. Nguyen, S. H. Park and D. Joung, Adv. Sci., 2023, 10, 2303164 CrossRef CAS.
- J. Belardi, N. Schorr, O. Prucker and J. Rühe, Adv. Funct. Mater., 2011, 21, 3314–3320 CrossRef CAS.
- J. M. J. d Toonder and P. R. Onck, Trends Biotechnol., 2013, 31, 85–91 Search PubMed.
- T. ul Islam, Y. Wang, I. Aggarwal, Z. Cui, H. Eslami Amirabadi, H. Garg, R. Kooi, B. B. Venkataramanachar, T. Wang, S. Zhang, P. R. Onck and J. M. J. den Toonder, Lab Chip, 2022, 22, 1650–1679 CAS.
- W. Huang, H. Li, L. Zheng, X. Lai, H. Guan, Y. Wei, H. Feng and X. Zeng, Chem. Eng. J., 2021, 426, 130837 CrossRef CAS.
- L. Huang, R. Zeng, D. Tang and X. Cao, Nano Energy, 2022, 99, 107376 CAS.
- Y. Xia Zhang, Y. He, Y. Liang, J. Tang, Y. Yang, H. Ming Song, M. Zrínyi and Y. Mei Chen, Appl. Surf. Sci., 2023, 615, 156328 CrossRef.
- Y. Cheng, Y. Ma, L. Li, M. Zhu, Y. Yue, W. Liu, L. Wang, S. Jia, C. Li, T. Qi, J. Wang and Y. Gao, ACS Nano, 2020, 14, 2145–2155 CrossRef CAS PubMed.
- Y. Zhang, Y. Hu, P. Zhu, F. Han, Y. Zhu, R. Sun and C.-P. Wong, ACS Appl. Mater. Interfaces, 2017, 9, 35968–35976 CrossRef CAS.
- Y. Lee, J. Park, S. Cho, Y.-E. Shin, H. Lee, J. Kim, J. Myoung, S. Cho, S. Kang, C. Baig and H. Ko, ACS Nano, 2018, 12, 4045–4054 CrossRef CAS.
- Q. Zhou, B. Ji, F. Hu, Z. Dai, S. Ding, H. Yang, J. Zhong, Y. Qiao, J. Zhou, J. Luo and B. Zhou, Adv. Funct. Mater., 2022, 32, 2208120 CrossRef CAS.
- Y. Li, P. Cao, P. Zhang, H. Yang, X. Zhu and R. Guo, Sensors, 2023, 23, 7354 CrossRef CAS PubMed.
- D. Shen, Y. Jiang, Z. Ma, P. Zhao, Z. Gong, Z. Dong and D. Zhang, J. Bionic Eng., 2021, 19, 73–82 CrossRef.
- R. S. Johansson and J. R. Flanagan, Nat. Rev. Neurosci., 2009, 10, 345–359 CrossRef CAS.
- Y. Zi, H. Guo, Z. Wen, M.-H. Yeh, C. Hu and Z. L. Wang, ACS Nano, 2016, 10, 4797–4805 CrossRef CAS PubMed.
- B. N. J. Persson, Tribol. Lett., 2022, 70, 88 CrossRef.
- S. Peng, Y. Yu, S. Wu and C.-H. Wang, ACS Appl. Mater. Interfaces, 2021, 13, 43831–43854 CrossRef CAS.
- Y. Lee, J. Myoung, S. Cho, J. Park, J. Kim, H. Lee, Y. Lee, S. Lee, C. Baig and H. Ko, ACS Nano, 2020, 15, 1795–1804 CrossRef.
- B. Ji, Y. Mao, Q. Zhou, J. Zhou, G. Chen, Y. Gao, Y. Tian, W. Wen and B. Zhou, ACS Appl. Mater. Interfaces, 2019, 11, 28060–28071 CrossRef CAS.
- M. Zhong, L. Zhang, X. Liu, Y. Zhou, M. Zhang, Y. Wang, L. Yang and D. Wei, Chem. Eng. J., 2021, 412, 128649 CrossRef CAS.
- Y. Jung, J. Choi, W. Lee, J. S. Ko, I. Park and H. Cho, Adv. Funct. Mater., 2022, 32, 2201147 CrossRef CAS.
- X. Shi, X. Fan, Y. Zhu, Y. Liu, P. Wu, R. Jiang, B. Wu, H.-A. Wu, H. Zheng, J. Wang, X. Ji, Y. Chen and J. Liang, Nat. Commun., 2022, 13, 1119 CrossRef CAS PubMed.
- S. Chun, J.-S. Kim, Y. Yoo, Y. Choi, S. J. Jung, D. Jang, G. Lee, K.-I. Song, K. S. Nam, I. Youn, D. Son, C. Pang, Y. Jeong, H. Jung, Y.-J. Kim, B.-D. Choi, J. Kim, S.-P. Kim, W. Park and S. Park, Nat. Electron., 2021, 4, 429–438 CrossRef.
- S. Chun, W. Son, C. Choi, H. Min, J. Kim, H. J. Lee, D. Kim, C. Kim, J.-S. Koh and C. Pang, ACS Appl. Mater. Interfaces, 2019, 11, 13608–13615 CrossRef CAS.
- V. E. Abraira and D. D. Ginty, Neuron, 2013, 79, 618–639 CrossRef CAS.
- B. Ji, Q. Zhou, B. Hu, J. Zhong, J. Zhou and B. Zhou, Adv. Mater., 2021, 33, 2100859 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Supplementary Videos S1–S6, Supplementary Fig. S1–S30, Supplementary Notes S1 and S2, and Supplementary Tables S1 and S2. See DOI: https://doi.org/10.1039/d4mh01217h |
| This journal is © The Royal Society of Chemistry 2025 |