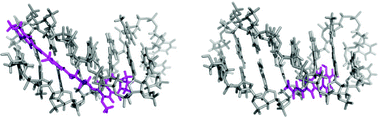
Pyrrolobenzodiazepines (PBDs) are sequence-selective DNA minor-groove binding agents that covalently bond to guanine with a reported preference for Pu-G-Pu sequences (Pu = Purine). Using HPLC/MS and Circular Dichroism (CD) methodologies, we have established for the first time that the aminal bond formed between PBD molecules and DNA is reversible. Furthermore, we have shown that while the rate of aminal bond cleavage does not depend on the sequence preference of a PBD molecule for a particular binding site, the rate of re-formation of the PBD-DNA adduct does. We have also shown that the PBD anthramycin (2) appears to be an exception to this rule in that, during cleavage from the DNA, its C-ring aromatizes and it cannot then re-attach due to a loss of electrophilicity at the C11-position. Although the C-ring aromatization of anthramycin has been previously reported to occur in the absence of DNA and after treatment with trifluoroacetic acid (TFA), in this case no pH lowering was required, with the DNA itself appearing to catalyse the process.