Kinetics of the ClO + CH3O2 reaction over the temperature range T = 250–298 K
Abstract
The kinetics of the potentially atmospherically important ClO + CH3O2 reaction (1) have been studied over the range T = 250–298 K at p = 760 Torr using laser flash photolysis radical generation, coupled with time resolved ultraviolet absorption spectroscopy, employing broad spectral monitoring using a charge coupled device detector array. ClO radicals were monitored unequivocally using this technique, and introduction of CH3O2 precursors ensured known initial methylperoxy radical concentrations. ClO temporal profiles were thereafter analysed to extract kinetic parameters for reaction (1). A detailed sensitivity analysis was also performed to examine any potential systematic variability in k1 as a function of kinetic or physical uncertainties. The kinetic data recorded in this work show good agreement with the most recent previous study of this reaction, reported by Leather et al. The current work reports an Arrhenius parameterisation for k1, given by: 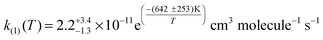 . This work therefore concurs with that of Leather et al. implying that the title reaction is potentially less significant in the atmosphere than inferred from preceding studies. However, reaction (1) is evidently a non-terminating radical reaction, whose effects upon atmospheric composition therefore need to be ascertained through atmospheric model studies.
. This work therefore concurs with that of Leather et al. implying that the title reaction is potentially less significant in the atmosphere than inferred from preceding studies. However, reaction (1) is evidently a non-terminating radical reaction, whose effects upon atmospheric composition therefore need to be ascertained through atmospheric model studies.


 Please wait while we load your content...
Please wait while we load your content...