Shuo Yang, Wei-Tao Jiang and Bin Xiao
Chem. Commun., 2021,57, 8143-8146
DOI:
10.1039/D1CC02930D,
Communication
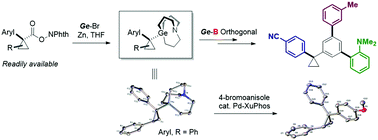
The construction of the cyclopropyl quaternary carbon center can afford a series of 1,1-olefin bioisosteres. Here, we report tertiary cyclopropyl carbagermatranes, which can be easily obtained by the zinc-mediated decarboxylation of NHP esters. In addition, they exhibit efficient reactivity in the palladium-catalyzed cross-coupling reaction and orthogonal reactivity with boron reagents, therefore acting as robust nucleophiles for the synthesis of tertiary cyclopropane and efficient intermediates for the formation of quaternary centers.