Helpful correlations to estimate the pKa of coordinated HNO: a potential–pH exploration in a pendant-arm cyclam-based ruthenium nitroxyl†‡
Abstract
The acid–base speciation of coordinated azanone (HNO) remains a highly relevant topic in bioinorganic chemistry. Ruthenium nitroxyl complexes with sufficient robustness towards ligand loss have gained significance as operating platforms to delve into such studies. In this work, we revisit an octahedral {RuNO}6 complex containing the cyclam-based pentadentate ligand Lpy = 1-(pyridine-2-ylmethyl)-1,4,8,11-tetraazacyclotetradecane and explore the thermodynamic and spectroscopic aspects of its reduced states in aqueous media. Upon in situ electro-generation of the bound HNO moiety, we have undertaken different strategies to determine both its acidity and electrochemical properties. This robust HNO complex does not undergo deprotonation in a wide pH range. We have found pKa ([Ru(Lpy)(HNO)]2+) = 13.0 ± 0.1 and 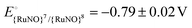 . There are indications that pKa (HNO) values in several ruthenium-based species correlate with the redox potential associated with the {RuNO}6,7 and {RuNO}7,8 couples. The present pKa extends the range of acidity of bound HNO to more than five pH units, confirming a remarkable sensitivity to the nature of the coordination sphere. This result lays new foundations to continue rational ligand design that may contribute to a better understanding of the different biological roles of both HNO and NO− by investigating key chemical aspects of model complexes.
. There are indications that pKa (HNO) values in several ruthenium-based species correlate with the redox potential associated with the {RuNO}6,7 and {RuNO}7,8 couples. The present pKa extends the range of acidity of bound HNO to more than five pH units, confirming a remarkable sensitivity to the nature of the coordination sphere. This result lays new foundations to continue rational ligand design that may contribute to a better understanding of the different biological roles of both HNO and NO− by investigating key chemical aspects of model complexes.



 Please wait while we load your content...
Please wait while we load your content...