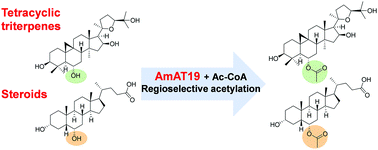
Tetracyclic triterpenes and steroids are pharmacologically important molecules, and acetylation could improve their bioactivities. In this study, a highly regio- and stereo-specific acetyltransferase, AmAT19, was discovered from Astragalus membranaceus. AmAT19 could selectively catalyze the 6α-OH acetylation of four tetracyclic triterpenes and steroids. The strict selectivity is associated with different orientations of the 6α/β-OH as indicated by molecular docking. Acetylated products 1a, 3a and 4a remarkably increased the inhibitory activity against the 3-chymotrypsin-like protease (3CLpro) of SARS-CoV-2, compared to 1, 3, and 4. AmAT19 could be a promising catalyst for specific 6α-OH acetylation to expand the molecular diversity of triterpenes and steroids.