High-pressure transformations of CaC2O5 – a full structural trend from double [CO3] triangles through the isolated group of [CO4] tetrahedra to framework and layered structures†
Abstract
Over the past few years, the concept of carbonates, as the salts of MCO3 or 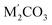 composition with [CO3] triangles in the crystal structures, was sufficiently extended. In addition to carbonates, crystal structures with stoichiometry M3CO5, M2CO4 and MC2O5 were predicted and successfully synthesized. In the present study, based on density functional theory and crystal structure prediction algorithms, we found a novel structure of CaC2O5, namely Ca-pyrocarbonate with monoclinic symmetry Cc, which is one of the possible agents of the global carbon cycle. This structure is characterized by the isolated [C2O5] groups consisting of two [CO3] triangles connected through a common oxygen atom. The thermodynamic stability field of Ca-pyrocarbonate with respect to the decomposition reaction into calcium carbonate and carbon dioxide begins at a pressure of 10 GPa. As the pressure increases to 21 GPa, the structure of Ca-pyrocarbonate transforms into the recently synthesized tetragonal modification I
composition with [CO3] triangles in the crystal structures, was sufficiently extended. In addition to carbonates, crystal structures with stoichiometry M3CO5, M2CO4 and MC2O5 were predicted and successfully synthesized. In the present study, based on density functional theory and crystal structure prediction algorithms, we found a novel structure of CaC2O5, namely Ca-pyrocarbonate with monoclinic symmetry Cc, which is one of the possible agents of the global carbon cycle. This structure is characterized by the isolated [C2O5] groups consisting of two [CO3] triangles connected through a common oxygen atom. The thermodynamic stability field of Ca-pyrocarbonate with respect to the decomposition reaction into calcium carbonate and carbon dioxide begins at a pressure of 10 GPa. As the pressure increases to 21 GPa, the structure of Ca-pyrocarbonate transforms into the recently synthesized tetragonal modification I![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 2d, in the structure of which carbon is in the sp3-hybridized state and [CO4] tetrahedra form isolated pyramidal [C4O10] anionic groups. At 59 GPa in the temperature range of 0–2500 K, CaC2O5-I
2d, in the structure of which carbon is in the sp3-hybridized state and [CO4] tetrahedra form isolated pyramidal [C4O10] anionic groups. At 59 GPa in the temperature range of 0–2500 K, CaC2O5-I![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 2d undergoes a phase transition to CaC2O5-Fdd2, with the framework structure of [CO4] tetrahedra. On further compression to about 80 GPa, the framework structure transforms into layered ones, C2 and Pc. In addition, we estimated the thermodynamic stability of CaC2O5 with respect to the minerals of the Earth's mantle. We found that CaC2O5 can coexist with bridgmanite up to pressures of 54 GPa at 300 K, where it reacts with the formation of a Ca-perovskite, magnesite, and solid CO2-V.
2d undergoes a phase transition to CaC2O5-Fdd2, with the framework structure of [CO4] tetrahedra. On further compression to about 80 GPa, the framework structure transforms into layered ones, C2 and Pc. In addition, we estimated the thermodynamic stability of CaC2O5 with respect to the minerals of the Earth's mantle. We found that CaC2O5 can coexist with bridgmanite up to pressures of 54 GPa at 300 K, where it reacts with the formation of a Ca-perovskite, magnesite, and solid CO2-V.
![Graphical abstract: High-pressure transformations of CaC2O5 – a full structural trend from double [CO3] triangles through the isolated group of [CO4] tetrahedra to framework and layered structures](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D2CP01748B&imageInfo.ImageIdentifier.Year=2022)


 Please wait while we load your content...
Please wait while we load your content...