Unconventional gas-phase preparation of the prototype polycyclic aromatic hydrocarbon naphthalene (C10H8) via the reaction of benzyl (C7H7) and propargyl (C3H3) radicals coupled with hydrogen-atom assisted isomerization†
Abstract
Polycyclic aromatic hydrocarbons (PAHs) are ubiquitous in the interstellar medium and in meteorites such as Murchison and Allende and signify the missing link between resonantly stabilized free radicals and carbonaceous nanoparticles (soot particles, interstellar grains). However, the predicted lifetime of interstellar PAHs of some 108 years imply that PAHs should not exist in extraterrestrial environments suggesting that key mechanisms of their formation are elusive. Exploiting a microchemical reactor and coupling these data with computational fluid dynamics (CFD) simulations and kinetic modeling, we reveal through an isomer selective product detection that the reaction of the resonantly stabilized benzyl 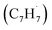 and the propargyl
and the propargyl 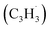 radicals synthesizes the simplest representative of PAHs – the 10π Hückel aromatic naphthalene (C10H8) molecule – via the novel Propargyl Addition–BenzAnnulation (PABA) mechanism. The gas-phase preparation of naphthalene affords a versatile concept of the reaction of combustion and astronomically abundant propargyl radicals with aromatic radicals carrying the radical center at the methylene moiety
radicals synthesizes the simplest representative of PAHs – the 10π Hückel aromatic naphthalene (C10H8) molecule – via the novel Propargyl Addition–BenzAnnulation (PABA) mechanism. The gas-phase preparation of naphthalene affords a versatile concept of the reaction of combustion and astronomically abundant propargyl radicals with aromatic radicals carrying the radical center at the methylene moiety 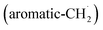 as a previously passed over source of aromatics in high temperature environments thus bringing us closer to an understanding of the aromatic universe we live in.
as a previously passed over source of aromatics in high temperature environments thus bringing us closer to an understanding of the aromatic universe we live in.



 Please wait while we load your content...
Please wait while we load your content...