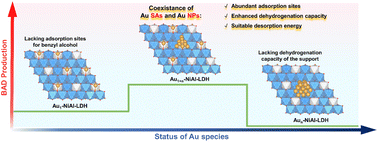
Introducing different active sites into heterogeneous catalysts provides new prospects to address the challenges in single-atom catalysis. Herein, the Au single atoms together and the Au nanoparticles were loaded onto NiAl-LDH by a facile impregnation–reduction method for the first time, resulting in the formation of Au1+n-NiAl-LDH, in which abundant Au single atoms are located around the Au nanoparticles with ∼5 nm size. When applied in the electrocatalytic benzyl alcohol oxidation reaction (BAOR), the as-prepared Au1+n-NiAl-LDH exhibits a remarkable selectivity of 91% and 177.63 μmol for benzaldehyde in 5 hours, while in contrast examples using solely Au single atom loaded NiAl-LDH (Au1-NiAl-LDH) and solely Au nanoparticle loaded NiAl-LDH (Aun-NiAl-LDH) can only realize 87.36 μmol production (75% selectivity) and 48.90 μmol production (28% selectivity) of benzaldehyde, respectively. Such a dramatic difference can be attributed to the synergistic effects of Au single atoms and Au nanoparticles. DFT calculation results reveal that for Au1+n-NiAl-LDH, Au single atoms promote the dehydrogenation capacity of LDH laminates, while Au nanoparticles offer adsorption sites for the electrophilic attachment of benzyl alcohol.