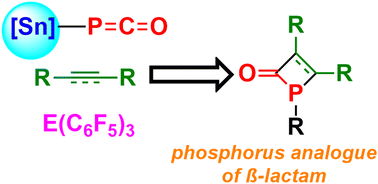
The reaction of the stannyl phosphaketene (Nacnac)SnPCO 1 (Nacnac = CH{(CMe)(2,6-iPr2C6H3N)}2) with B(C6F5)3 produced the 1,4-addition product of (Nacnac)SnPCO(B(C6F5)3). However, the corresponding reactions in the presence of dimethyl maleate, diisopropyl fumarate or diethyl-but-2-ynedioate gave [2+2] addition yielding four-membered phosphacycles, ((Nacnac)Sn(MeO2C))CHPC(OB(C6F5)3)CH(CO2Me), [(C6F5)3B)PC(OSn)C(CO2Me)CH(CO2Me)]2, (Nacnac)Sn(iPrO2C)CC(OAl(C6F5)3)P[CH(CO2iPr)CH2(CO2iPr)]CH(CO2iPr), and (Nacnac)SnP (EtO2CCC(CO2Et))CO(B(C6F5)3), respectively. In contrast, the corresponding reaction of phenylacetylene gave the FLP-addition product (Nacnac)SnOC(P)C(Ph)CH(B(C6F5)3). Collectively, this reactivity demonstrates that the stannyl phosphaketene 1 can act as a synthon for P-analogues of β-lactam derivatives.