Emerging 2D materials for antimicrobial applications in the pre- and post-pandemic era
Cemile
Gokce
 a,
Cansu
Gurcan
ab,
Omur
Besbinar
ab,
Mehmet Altay
Unal
a,
Cansu
Gurcan
ab,
Omur
Besbinar
ab,
Mehmet Altay
Unal
 b and
Acelya
Yilmazer
b and
Acelya
Yilmazer
 *ab
*ab
aDepartment of Biomedical Engineering, Ankara University, Golbasi, Ankara, Turkey. E-mail: ayilmazer@ankara.edu.tr
bStem Cell Institute, Ankara University, Balgat, Ankara, Turkey
First published on 30th November 2021
Abstract
Infectious diseases caused by viral or bacterial pathogens are one of the most serious threats to humanity. Moreover, they may lead to pandemics, as we have witnessed severely with the coronavirus disease 2019 (COVID-19). Nanotechnology, including technological developments of nano-sized materials, has brought great opportunities to control the spreading of such diseases. In the family of nano-sized materials, two-dimensional (2D) materials with intrinsic physicochemical properties can efficiently favor antimicrobial activity and maintain a safer environment to protect people against pathogens. For this purpose, they can be used alone or combined for the disinfection process of microbes, antiviral or antibacterial surface coatings, air filtering of medical equipment like face masks, or antimicrobial drug delivery systems. At the same time, they are promising candidates to deal with the issues of conventional antimicrobial approaches such as low efficacy and high cost. This review covers the antiviral or antibacterial activities of 2D materials and highlights their current and possible future applications. Considering their intrinsic properties, 2D materials will become part of the leading antimicrobial technologies for combating future pandemics anytime soon.
A. Introduction
The word “microbes” or “microorganisms” refers to small organisms that can only be seen under a microscope and cover bacteria and viruses. Viruses do not contain ribosomes, mitochondria or other cell-like organelles. For this reason, they cannot replicate without the metabolic processes of the host cell, and therefore they are completely parasitic. This property of viruses differentiates them from other microorganisms including bacteria, most of which can replicate on their own, although they can also be parasitic.1 Infectious diseases caused by pathogenic bacteria and viruses have been threatening human health for many years and resulted in losses of millions of lives.2–5 Mankind has seen several worldwide pandemics that were caused by influenza viruses in the 1900s and 2000s, human immunodeficiency virus infection/acquired immune deficiency syndrome (HIV/AIDS) in 1981, severe acute respiratory syndrome (SARS) in 2003, Middle East respiratory syndrome (MERS) in 2012 and most recently a novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2 or namely COVID-19) in 2019.2,3,6,7 These diseases can be easily transmitted from infected to healthy individuals through respiratory droplets, aerosols, contacts or body fluids.8 Therefore, the need for antimicrobial drugs, vaccines, and new scientific and technological developments has come into prominence in order to deal with the growing problem of such infections.3,9,10 In particular, since the beginning of the COVID-19 pandemic, nanotechnology has shown to be successful in combating viruses in different applications, including the development of vaccines or personal protective equipment (PPE).11–152D materials include graphene, graphene-based materials (GBMs) (e.g., graphene oxide (GO), reduced graphene oxide (rGO)), graphitic carbon nitride (g-C3N4), transition metal dichalcogenides (TMDs) such as molybdenum disulfide (MoS2), transition metal oxides such as manganese dioxide (MnO2), black phosphorus (BP), transition metal carbides and nitrides (MXenes) such as titanium carbide (Ti3C2Tx), mono elemental materials like phosphorene (P) and layered double hydroxides (LDHs). They have unique optical, electrical, and mechanical properties enabling their use in various biomedical applications.11,16–24 Thanks to these intrinsic properties, their low cost and large-scale production, they have also been studied for various antimicrobial applications.11–14,16–22,25 Firstly, in order to enhance the barrier properties of face masks/personal protective equipment (PPEs), 2D materials have been used to coat their surfaces. Unlike existing masks such as N95, these materials may allow a mask to be used several times by cleaning itself, eliminating waste from using too many masks. Secondly, they can also be used during disinfection processes in order to eliminate pathogens. They exhibit higher and safer antimicrobial activity for these applications over conventional methodologies including chlorination, ozonation, and UV light. That is because 2D materials circumvent the inherent problems of such methodologies such as the carcinogenic byproducts of chlorination and ozonation causing environmental harmfulness, and naturally possible UV-light–resistant microorganisms.26 As a result, developing new materials against microbes is highly preferred rather than using conventional methodologies to decrease the resource consumption, cost, environmental pollution issues and obtain better potential safety. Finally, antiviral or antibacterial drug delivery can also make use of 2D materials to achieve a safer and efficient therapeutic efficacy. In this case, 2D materials may provide effective drug release, ease of complexation and increased solubility for antimicrobial drugs.
There are excellent review articles published on “2D materials and antibacterial applications” or “2D materials and antiviral applications” separately. Most of these articles focus on antibacterial applications and a majority of them refers to graphene or its derivatives.17,27,28 On the other hand, this review article provides an overall view on the antiviral and antibacterial studies performed using 2D materials so far and sheds light on how these materials can reduce the chance of infection effectively (Fig. 1). We specifically aim to emphasize the potential of 2D materials in antimicrobial applications in general by taking attention to already-reported antibacterial or antiviral studies in addition to the recent research efforts in response to the COVID-19 pandemic.11,29 This review will provide new inspirations to develop and design novel antimicrobial agents in combination with 2D materials for combating global infectious diseases in the future.
B. 2D materials among other nanomaterials
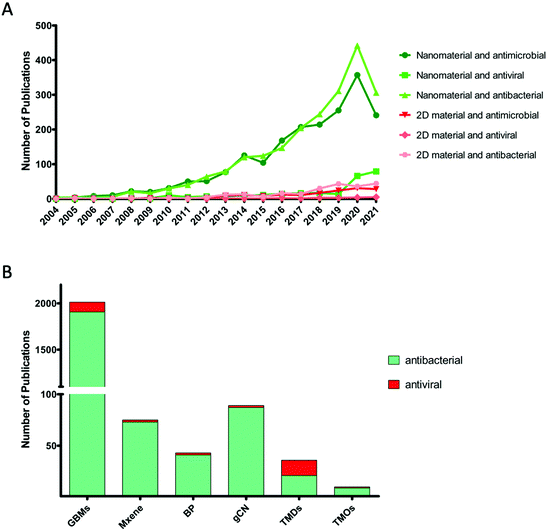
The discovery of graphene opened up new perspectives in the nanotechnology field. Graphene and GBMs have been employed in various biomedical applications, including antimicrobial research. Following the GBMs, scientists explored new members of the 2D materials family.19 In order to better understand the current situation of 2D materials in antimicrobial research, we performed a systematic review of the literature on nanomaterials and 2D materials studied in antimicrobial applications, according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The electronic databases (Scopus and Web of Science) were used as data sources. Fig. 2A shows the analysis of the literature concerning nanomaterials and 2D materials directed towards antimicrobial, antibacterial, and antiviral applications. There is a clear difference between the number of studies involving nanomaterials or 2D materials. This analysis suggests that 2D materials showed promise in antimicrobial or antibacterial research only recently. More importantly, antiviral research for both nanomaterials and 2D materials has gained attention, possibly following the COVID-19 pandemic. Therefore, this literature survey shows a clear need to explore further the potential of 2D material-based antibacterial and antiviral applications. Fig. 2B, on the other hand, shows the distribution of antibacterial or antiviral studies among different 2D materials. It is clear that the majority of the publications involving antibacterial or antiviral applications involve graphene and other GBMs. However, it is also noteworthy that there is an increasing interest for other 2D materials, especially MXenes and gCN.I. 2D materials for air filtering and personal protective equipment
Conventional face masks and other PPEs such as gowns and medical aprons, filters of air conditioning systems, and medical respiratory devices do not have enough intrinsic antimicrobial actions. For this reason, they are prone to microbial or viral colonization. An efficient antimicrobial technology on air filtering is crucial for maintaining a safe air environment and protecting human health, especially during pandemics.30 For filtering applications, antimicrobial 2D materials including graphene and its derivatives, MXene, foam forms of MXene, and MXene-graphene composites can offer excellent potential.2,25 MXene foams with tunable porosities and pore size could be used in healthcare filtration systems to make advanced face masks or filters. Other than current high-quality masks like N95, advanced options will stop the transmission of viral or bacterial contaminants adhered onto the surface of the mask, and inactivate these contaminants locally.2 In addition, advanced face masks will reduce the usage of disposable face masks and other PPEs made up of plastics through the application of MXenes, graphene, or its derivatives on these plastic surfaces.31,32 These coatings also provide advantages for environmental and economic issues such as pollution and high cost.In 2019, Bhattacharjee et al. developed PPEs made up of polymers such as polyamide and textile fabrics including cotton, polyester, and nylon that were modified with graphene and its derivatives. The authors suggested that since these advanced PPEs can be recycled and reused, they can overcome the limitations of current PPEs.32 Such an approach will certainly allow the development of different PPEs with desired properties. For instance, Hasani and Montazer treated GO onto cellulosic/polyamide fabric, reduced the fabric to rGO via reducing agents like Na2S2O4, NaOH, and reported antimicrobial activity of rGO on the fabric against E. coli and P. aeruginosa, S. aureus, E. faecalis, and eukaryotic fungus C. albicans. As a result, the reducing agent Na2S2O4 with NaOH showed the optimum match with rGO and showed the best reduction for low-level electrical resistivity and antibacterial effects. In addition, rGO-coated fabrics provided 100% UV protection.33 Thanks to the antimicrobial, antistatic, and electrical properties of graphene, Zhong et al. developed a graphene-based face mask with higher superhydrophobic and photothermal properties allowing self-cleaning capabilities.34 In contrast to masks without graphene, they showed that the superhydrophobic effect of graphene grants better protection against viral droplets. In addition, graphene shows high absorption, which is 95%, across the solar spectrum from 300 to 2500 nm and a static angle of over 140°, allowing for the self-sterilization of remaining viruses. They also stated that the surface temperature of graphene-based masks could quickly go above 70 °C following solar illumination, which results in highly efficient photothermal performance against SARS-CoV-2.25,34
Furthermore, some companies like Flextrapower have developed a graphene mask blocking the viral droplets via its graphene-infused filter.35 ZEN Graphene Solution Ltd and Graphene Composite Ltd have developed silver nanoparticle–functionalized GO ink for face masks and other PPEs that can possess virucidal activity on influenza viruses and SARS-CoV-2.36 PlanarTECH and Ideati 2AM™ has developed cotton fabric–based reusable face masks that are coated with both graphene and carbon nanomaterials and were reported to be highly resistant to bacteria. While the graphene coating gives thermal distribution across the mask, the inner cotton layer has a water-repellent ability.37 LIGC Applications Ltd has developed a graphene-based respirator mask named as Guardian G-Volt that can trap pathogens. It is antistatic, reusable up to 10 times, has a LED light system to alert replacement of the mask, and can sterilize itself from a portable battery.38 In another report, Tiwary et al. developed a transparent antiviral surface coating based on a copper–graphene (Cu–Gr) nanocomposite in order to achieve potential antiviral activity against influenza A virus (IAV). They concluded that 2D graphene nanosheets and copper could synergistically interfere with the viral replication cycle by roughly 80%, thus blocking the viral particle entry within 30 minutes to the untreated one. They further showed that a 5 μM Cu–Gr composite with 10 mM polyvinyl alcohol (PVA) could be used to generate a solid transparent coating with higher antiviral activity leading to a 70% decrease in the activity of reporter virus, and maybe later implemented on face masks to minimize the virus transmission even more.3,39 Despite the remarkable advances in the development of PPEs, many challenges remain to be improved like heavyweight, bulky nature, lack of mobility and breathability, heat and physical stress, and lower protection against pathogens. Therefore, developing new 2D material–based PPE technologies with improved antimicrobial properties is critical to overcoming these shortcomings.32,40,41
II. 2D materials for disinfection
Pathogens like bacteria and viruses as well as organic and inorganic wastes are considerable environmental pollutants causing health problems. For example, illnesses caused by Escherichia coli (E. coli) and transmission via contaminated water brings about many childhood deaths worldwide.42–44 Conventional bacterial and viral disinfection approaches such as ultraviolet (UV) light, chlorination, and ozonation reagents can usually be ineffective due to environmental pollution, toxic byproduct generation, high energy consumption, and related cost.42,45–47 Besides, it is not practical to directly use natural enzymes like peroxidase as antimicrobials because of poor stability, high production cost and complicated purification.48–50 Thus, 2D materials such as GO, MoS2, and MXenes with much more favorable intrinsic material properties were explored by researchers, and their innate antimicrobial behaviors have been investigated over the last decade.17,19,20,42,51,52 They exhibit large specific surface area, remarkable stability, good catalytic activity, minimal cytotoxicity, and unique energy-conversion ability.48 Several groups have suggested that 2D materials possess photo-induced antimicrobial activity by inducing oxidative stress on pathogens via the generation of reactive oxygen species (ROS) and photothermal effect via the activation by near-infrared light (NIR).42,51,53To illustrate, GO is an intrinsically biocompatible nanomaterial with a high surface area that could be good for the adsorption of bacteria and viruses. It interacts effectively with the surfaces of viruses via hydrogen bonding and electrostatic interactions. It can be coupled with antimicrobial metals such as Ag and Cu nanoparticles for practical functionalization. The size and surface roughness of GO are important determinants of the efficiency of disinfection.13,42,54–56 In 2015, Ye et al. demonstrated the antiviral activity of GO against both DNA and RNA viruses even at low concentrations (1.5 μg mL−1) that may be explained by its negative surface charge and sharp-edged nanosheet structure. They have used GO at a concentration of 6 mg mL−1 in order to not have cytotoxic effects (>90% of cell viability). They have obtained that the plaque formation units of both viruses are prominently decreased and GO has shown time- and concentration-dependent antiviral properties. They have also found that GO mainly blocks the infection prior to the viral entry.42,55,57 In the same year, Song et al. reported the development of a GO-based label-free methodology to rapidly detect and disinfect environmental enteric viruses EV71 and H9N2. They reported that 95.6% of EV71 and 83.2% of H9N2 are captured on GO at 10 μg ml−1 and the capturing efficiency is found to be 100% for EV71 and 92.9% for H9N2 when the GO concentration is increased to 50 μg ml−1 at room temperature. They have also reported that the capture efficiency for H9N2 is increased at higher temperatures (56 °C). It has prominently degraded within only 5 min while EV71 is degraded only at 30 min in the presence of GO.54,58 In 2016, Zou et al. reviewed the antibacterial activities of graphene-derived nanomaterials based on their extraordinary properties and antibacterial mechanisms. According to this report, larger GO sheets usually show more robust antimicrobial activity because they wrap and isolate the cells completely.17,59–61 In 2018, Vi et al. investigated the antibacterial activity of Ag–GO nanocomposites grafted through thiol groups covalently without using reducing agents. They stated that GO wraps bacterial cell membrane and Ag kills bacteria via its toxicity.53,56 Alimohammadi et al. studied 2D MoS2 and MnO2 nanosheets for physical contact disinfection because of their sharp edges that could distinctly destroy the cell wall of Gram + B. subtilis and Gram-E. coli. This study vertically aligned and randomly oriented MoS2/MnO2 on GO/Ti3C2Tx MXene nanosheets. They concluded that Gram + B. subtilis show a higher loss in membrane integrity and that vertically aligned 2D nanosheets exhibit higher antibacterial activity against both bacteria classes than randomly oriented 2D nanosheets.59 Similarly, sharp-edged tungsten disulfide (WS2) nanosheets, bismuth selenide (Bi2Se3) nanodiscs, and plate-like molybdenum trioxide (MoO3) were found to be highly effective against specific bacterial infections through physical contact destruction.17,20,62–64 More recently, in 2021, Unal et al. performed in silico and in vitro analyses to evaluate whether GO nanosheets can interact with specific SARS-CoV-2 surface proteins and receptors for inhibition. They found that GO nanosheets have a strong affinity toward both the viral spike and ACE2 and they can significantly reduce the infection of three different viral clades, GR, GS, and S, even if there are mutated viral spike proteins.65
As mentioned above, 2D materials have also attracted tremendous attention in disinfection technologies owing to their photothermal activity under light irradiation due to their strong NIR photothermal conversion efficiency.66–72 To exemplify, Pal et al. have studied with MoS2–TiO2 and demonstrated its synergistic performance. They concluded that this nanocomposite possesses high adsorption capacity at about 364.56 mg g−1 for methylene blue at room temperature and antibacterial efficiency against S. aureus and E. coli. The nanocomposite strongly interacts with the bacterial cell membrane surface leading to better inhibition of S. aureus growth compared to that of E. coli. They have also found that the minimal inhibitory concentration is 25–50 mg mL−1, which is a superior antibacterial assessment upon comparing the previous antimicrobial studies of different 2D materials in the literature.73 Navale et al. reported that reduced GO (rGO)-WS2 nanosheets had a significant bacterial inhibitory effect than pure WS2 or reduced GO alone.74 Bismuth oxybromide (BiOBr) and graphitic carbon nitride (g-C3N4) nanosheets may inactivate bacteria efficiently through the photocatalytic disinfection process under visible light irradiation.17,75–80 For photodynamic disinfection, Tan et al. designed a novel antibacterial system based on BP nanosheets as a photosensitizer. They combined BP nanosheets with poly 4-pyridone methyl-styrene and peroxide (PPMS-EPO) to form an antibacterial PPMS-EPO/BPS film. This system showed superior ROS generation leading to an effective antibacterial activity with rates of 99.3% and 99.2% against E. coli and S. aureus after 10 min of light irradiation, respectively.81 Graphene also shows good photodynamic activity against Semliki Forest virus (SFV), vesicular stomatitis virus (VSV), HSV-1, HIV-1, IAV, and mosquito iridovirus (MIV).11,82,83 Moreover, several studies have concluded that different phases of 2D MoS2 nanosheets can selectively generate ROS on the bacterial cell surface.17,84 In 2018, Eke et al. studied whether membrane embedded with P can mimic water treatment and has the self-cleaning ability. They obtained that the average recovered flux of a P-modified membrane was four times higher than that of an unmodified membrane and the methylene blue coverage was four times lower than that of the unmodified membrane after filtering methylene blue through the membrane under UV irradiation. These results displayed that studies with monoelemental materials could be used for the control of fouling and pathogen inactivation.85 In addition to the study of Eke et al., Liu et al. have developed a promising strategy for synergistic capture of bacteria and elimination of its infection recently. They constructed a composite hydrogel via the combination of antimonene nanosheets (AM, 2D form of Sb element) with a chitosan (Cs) network to investigate both in vivo wound healing and in vitro antibacterial activity. As a result, they found predominant actions against E. coli, S. aureus, and bacteria-infected skin wounds under NIR irradiation on account of the good photothermal performance of AM and capture capacity of Cs.86 LDHs are also used in photocatalytic disinfection owing to their alternative metals. They are chemically inert. Zhao et al. synthesized Ti3+ doped zinc–titanium (ZnTi) LDH nanosheets with different lateral sizes, ranging from 40 to 80 nm, to investigate its antimicrobial activity against S. cerevisiae, E. coli, and S. aureus under visible light irradiation. They found higher inhibition of bacteria growth than conventionally prepared ZnTi LDH bulk. They also obtained the size-dependent activity of ZnTi LDHs, and 40 nm ones gave the best result which is 95% cell death.87 In another study by Moaty et al., zinc–iron (Zn–Fe) LDH has been successfully prepared and its durable antimicrobial activities have been evaluated against various types of microbes including fungi, Gram-positive, and Gram-negative bacteria. According to this study, Zn–Fe LDH is highly crystalline and has a homogeneous and rough surface with several pores. Zn–Fe LDH has also been found as a durable antimicrobial agent having the minimum inhibitory concentration between 0.49–15.60 μg mL−1 similar to some standard drugs like Ampicillin.88
In summary, thanks to their physiochemical properties and excellent antimicrobial activities, 2D materials are promising for disinfection strategies. However, there are several important issues to delineate, such as the suitability of 2D material–based disinfection on different surfaces or environments, biosafety consideration of 2D materials, and deeper understanding of the antimicrobial effect.
III. 2D materials as nanotherapeutics against pathogens
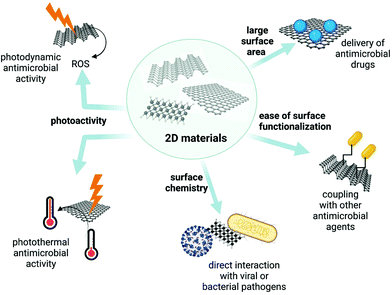
The design of new drug delivery systems for antimicrobial therapy focuses on being affordable and lowering the adverse side effects of antimicrobial drugs when patients take high drug doses. In order to achieve such a goal, nanotechnology-based drug delivery systems represent an essential option to achieve because 2D materials may offer unique physicochemical properties such as high surface area, ease of chemical functionalization and photoactivity. Thanks to these properties, 2D materials show antimicrobial activity through different mechanisms (Table 1 and Fig. 3).| 2D material | Mechanisms of antimicrobial activity | Ref. | |
|---|---|---|---|
| GBMs | Gr | ⇒ Lower affinity towards pathogens due to hydrophobicity | 34, 82, 120 and 121 |
| ⇒ Photodynamic and photothermal activities | |||
| Gr–Cu | ⇒ İnterference with viral proteins | 3 and 39 | |
| ⇒ Blockage of viral replication | |||
| Thiol grafted GO–Ag | ⇒ İnteraction with bacterial cell membrane and cell wall | 56 | |
| ⇒ Oxidative stress | |||
| GO | ⇒ Electrostatic interactions with microbial proteins | 57, 58 and 65 | |
| ⇒ İnterference with viral or bacterial proteins | |||
| ⇒ Blockage of viral entry | |||
| rGO | ⇒ Coating of cellulosic/polyamide fabric surface | 33 | |
| ⇒ İmproved electrical resistance | |||
| ⇒ İnhibition of antibacterial growth and superior UV protection | |||
| rGO–WS2 | ⇒ Direct contact and oxidative stress on bacterial cells | 74 | |
| GO-Hypericin | ⇒ Physical adsorption leading to inhibition of infection | 102 | |
| ⇒ Drug delivery platform for viral diseases | |||
| gCN | g-C3N4 | ⇒ Photocatalytic activity under visible light | 75, 118 and 119 |
| MXenes | T3C2Tx | ⇒ Size- and exposure time–dependent direct physical interactions | 22, 52, 107, 122 and 123 |
| ⇒ İnterference with viral uptake mechanisms | |||
| ⇒ İnterference with bacterial cell wall | |||
| Mo2Ti2C3Tx | ⇒ İnterference with viral uptake mechanisms | 107 | |
| Xenes | P | ⇒ Potential UV photocatalysis activity | 85 |
| ⇒ Self-cleaning membrane coverage | |||
| AM | ⇒ NIR photothermal activity in combination with Cs | 86 | |
| ⇒ Physical interactions with bacteria | |||
| ⇒ Treatment of bacteria-infected wound | |||
| TMDs | MoS2 | ⇒ Physical contact leading to disinfection | 59 and 84 |
| ⇒ Targeting bacterial peptidoglycan | |||
| ⇒ Oxidative stress | |||
| WS2 | ⇒ Time- and concentration-dependent direct contact damage | 62 | |
| ⇒ Oxidative stress | |||
| MoS2–TiO2 | ⇒ Adsorption via strong nanocomposite-bacteria interactions | 73 | |
| ⇒ NIR photothermal activity | |||
| TMOs | MoO3 | ⇒ Direct physical contact with bacteria | 63 |
| ⇒ Photocatalysis activity | |||
| MnO2 | ⇒ Direct physical interactions with bacterial surface | 59 | |
| BP | BP | ⇒ Disinfection through ROS generation | 81 and 124 |
| BP – PPMS | ⇒ Superior photodynamic activity | 81 | |
| BP – Ag nanoparticles | ⇒ Photothermal activity of the BP in combination with Ag-induced oxidative stress | 125 | |
| LDHs | ZnTi | ⇒ Size-dependent visible light photochemical activity | 87 |
| ZnFe | ⇒ Strong pH-dependent adsorption to heavy metal ions | 88 | |
| ⇒ İnhibition of bacteria through ROS and positive charge |
Several studies have already reported efficacious antiviral therapy against a series of common viruses such as Hepatitis B (HBV), herpes simplex virus (HSV), and HIV.89–93 Surface recognition and nanoscale interactions between such materials are crucial for successful drug delivery.11,94 Their small size, large surface to volume ratio, and tunable surface charge can favor efficient drug delivery and entry through the cell membrane.94–100 In particular, graphene and GO have been studied for antiviral drug delivery and have successfully shown a broad-spectrum antiviral activity.57 They may treat antiviral diseases and act effectively by mimicking the cell surface as an antiviral drug platform. For example, an antiviral Hypericin which is generally used against retroviruses causing hepatitis B was loaded on GO through physical adsorption in order to achieve slow drug release with no cytotoxicity. As a result, this GO-Hypericin system showed effectiveness in inhibiting the viral activity of retroviruses.101–103
In a study by Sametband et al., HSV attachment on cells was inhibited by GO and its functionalized derivative rGO–SO3 at concentrations up to 100 μg mL−1 and without affecting cell-to-cell spread. They were shown to mimic cell surface receptor, heparan sulfate. The study has also concluded that GO and rGO–SO3 have almost the same antiviral potential, and the materials with large lateral dimensions cannot inhibit the viral infection. The study has also concluded that GO and rGO–SO3 have almost the same antiviral potential, and the materials with large lateral dimensions cannot inhibit the viral infection.104 Deokar et al. used another approach in which superior (approximately 99.9%) photothermal destruction of HSV-1 was achieved by sulfonated magnetic nanoparticles of size ranging from 5 to 25 nm functionalized with reduced graphene oxide under 808 nm of light for 7 minutes.105 In another study, further functionalization of GO with a broad range of alkyl chains resulted in antiviral activity through synergistic electrostatic and hydrophobic interactions.106 In addition to graphene-based materials, MXenes have also been recently shown to present antiviral activity. Unal et al. reported that Ti3C2Tx and Mo2Ti2C3Tx MXene could inhibit the infection of SARS-CoV-2 in Vero E6 cells in vitro. They concluded that Ti3C2Tx has no cytotoxicity and it particularly inhibits the infection caused by the viral clade GR rather than other clades (>99% inhibition at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3125 dilution), and Mo2Ti2C3Tx are able to inhibit >95% viral copies at 100 μg mL−1. Thus, they suggested that it is highly essential to take into consideration genotypes and mutations of viruses.107 While using such materials directly for their antiviral properties is a potential option, engineering them to deliver antiviral or antimicrobial drugs could also be another option. Still, to carry these 2D materials a step forward towards clinical studies, several factors need to be considered, including biodegradation profiles, permeability through biological membranes, non-specific cellular uptake, aggregation profile during physical processes.94,108,109 Therefore, improvements of currently available antivirals should be explored rigorously using advances in nanotechnology for future pathogenic infections causing pandemics.
3125 dilution), and Mo2Ti2C3Tx are able to inhibit >95% viral copies at 100 μg mL−1. Thus, they suggested that it is highly essential to take into consideration genotypes and mutations of viruses.107 While using such materials directly for their antiviral properties is a potential option, engineering them to deliver antiviral or antimicrobial drugs could also be another option. Still, to carry these 2D materials a step forward towards clinical studies, several factors need to be considered, including biodegradation profiles, permeability through biological membranes, non-specific cellular uptake, aggregation profile during physical processes.94,108,109 Therefore, improvements of currently available antivirals should be explored rigorously using advances in nanotechnology for future pathogenic infections causing pandemics.
IV. 2D materials for future pandemics
As the world is coping with the worst pandemic in our century, it is inevitable to think about possible future pandemics. The current COVID-19 fight continues as new variants of the virus are emerging across different countries. Since the first report of SARS-CoV-2, numerous mutations have been identified, and currently, the virus is classified with 10 distinct clades by the GISAID platform.110 The evolution and rise of pathogens need close attention, and therefore, it is crucial to monitor the effectiveness of current nanomaterials against them and tailor the properties according to specific needs. One tool that can potentially help and allow preliminary screening of antiviral or antibacterial properties is in silico computational analysis. By performing docking analysis between important viral or bacterial pathogens and 2D materials, we can gain insight into how they can interfere and inhibit infections. For example, through in silico docking analysis, GO has been shown to interfere with SARS-CoV-2 spike protein and its cellular receptor ACE2.11 Such an approach could be implemented for future applications in order to screen the effectiveness of newly synthesized 2D materials.While this review discusses currently available applications of 2D materials against pathogens, it is noteworthy to have a projection towards other possible applications. The success of mRNA-lipid nanoparticle vaccines against COVID-19 has emphasized the importance of developing nanotechnology-based delivery vectors.15,111 Considering that lipid nanoparticles were first discovered in the 1990s, research efforts investigating the biomedical applications of 2D materials will undoubtedly contribute to our battles against infectious diseases in the future. Supporting this idea, GO has already been reported to deliver mRNA molecules into human cells efficiently. In a study by Choi et al., rat or human adipose–derived fibroblasts (rADFs or hADFs) have been transfected with mRNAs encoding for reprogramming factors which were complexed to functionalized GO nanosheets. Transfection efficiencies of GO alone and PEI alone have been found to be around 20% which can be considered as a significantly low transfection efficiency compared to the efficiency of GO-PEI-RNAs (about 50%). In addition, they have achieved high reprogramming efficiency (0.12%) with rADFs.112 Complexation with GO has successfully protected mRNA from degradation. In addition to the delivery potential, GO is known to modulate immune responses.11,113 GO and other potential 2D materials can work as vaccine adjuvants for future applications by boosting adaptive responses.
Since the discovery of graphene, 2D materials have gained much attention and are currently used in various disciplines, including biomedical sciences. Over the last years, their photocatalytic and photoactive properties have become more critical.114,115 For example, in cancer therapy, MXenes, BPs or g-C3N4 have shown great potential in photodynamic and photothermal therapies.116,117 Furthermore, catalytic activities of monoelemental materials have been tested for photocatalytic fouling control of membrane filtration, and P has achieved self-cleaning via photocatalysis action. Hence, their structures similar to BP nanosheets could be candidates for pathogen inactivation, and they hope to be studied further to provide a safe environment.23 As discussed above, the same properties allowed the successful development of 2D material-based masks and disinfection technologies (Fig. 3). As scientists develop much more efficient photoactive materials, their involvement in antimicrobial systems will become even more inevitable. In addition, the wavelength at which the material becomes active plays an essential role in the antimicrobial mechanism. For example, visible light–induced photocatalytic activity is the most applicable option for current technologies.43,78,79,118,119 However, for future applications, when reuse of masks or self-disinfection is needed at a specific time point, 2D materials that can be activated by infrared or near-infrared light would offer an advantage over the others.
In conclusion, scientists have been studying the biomedical applications of 2D materials and their opportunities in the field since the discovery of graphene and they have demonstrated potential usage in biosensors, imaging, cancer, and antimicrobial technologies. They mostly achieved high levels of antimicrobial efficiencies, given their appropriate size, dimension, surface area, functionalization, biosafety, self-cleaning, photothermal, and photodynamic activities. Some studies have also shown that the combined forms of them with other materials may have better activities against microbes. On the other hand, some challenges of Xenes such as lack of a standard synthesis technique controlling its properties, small scale production, retention time, and the actual mechanism of disinfection remain to be further solved. In addition, we have foreseen that more studies will be carried out not only in the antibacterial field but also in the antiviral field shortly soon. Similar mechanisms may work for antiviral and antibacterial applications, especially when we consider material characteristics. Therefore, it can be said that developments in the antibacterial studies of 2D materials may hold promise for antiviral studies of the same materials and it might help scientists to discover their potential for both applications. For example, considering that it would be best to have a surface or air filtering system consisting of 2D materials with both antibacterial and antiviral properties, it is crucial to discuss both antimicrobial properties at the same time. Furthermore, it is very well known that silver or gold nanoparticles harbor both antibacterial and antiviral properties, therefore discussing research efforts that are aimed to evaluate both of these properties for 2D materials will be beneficial in the field. For these reasons, we believe this review will provide new inspirations to develop and design novel antimicrobial agents in combination with 2D materials for combating global infectious diseases in the future.
The COVID-19 pandemic has shown that knowledge and expertise developed in the field of nanotechnology were crucial for our fight against it. This review article highlighted how 2D materials can combat different pathogens, including bacteria or viruses. Their intrinsic antimicrobial properties will certainly make 2D materials important players for combating current or future pandemics.
Conflicts of interest
There are no conflicts of interest to declare.Acknowledgements
CG, OB, MAU and AY would like to thank the funding from the Scientific and Technological Research Council of Turkey (TUBITAK) under grant number 18AG020. AY would like to acknowledge the Turkish Academy of Sciences (TUBA) for financial support under the young investigator programme. Fig. 1 and 3 were created with BioRender.com.References
- M. W. Taylor, in Viruses and Man: A History of Interactions, Springer, 2014, pp. 23–40 Search PubMed.
- N. Dwivedi, C. Dhand, P. Kumar and A. Srivastava, Mater. Adv., 2021, 2, 2892–2905 RSC.
- I. Das Jana, P. Kumbhakar, S. Banerjee, C. C. Gowda, N. Kedia, S. K. Kuila, S. Banerjee, N. C. Das, A. K. Das and I. Manna, ACS Appl. Nano Mater., 2020, 4, 352–362 CrossRef.
- Infectious diseases kill over 17 million people a year: WHO warns of global crisis, https://www.who.int/whr/1996/media_centre/press_release/en/(accessed August 17, 2021).
- The Top 10 Causes of Death, https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death, (accessed August 16, 2021).
- H. c. Editors, Pandemics That Changed History, https://www.history.com/topics/middle-ages/pandemics-timeline, (accessed August 17, 2021).
- Y. Wu, W. Ho, Y. Huang, D.-Y. Jin, S. Li, S.-L. Liu, X. Liu, J. Qiu, Y. Sang and Q. Wang, Lancet, 2020, 395, 949–950 CrossRef CAS.
- P. Vasickova, I. Pavlik, M. Verani and A. Carducci, Food Environ. Virol., 2010, 2, 24–34 CrossRef.
- P. R. Saunders-Hastings and D. Krewski, Pathogens, 2016, 5, 66 CrossRef PubMed.
- A. Fehr, S. Perlman, H. Maier, E. Bickerton and P. Britton, Methods Mol. Biol., 2015, 1282, 1–23 CrossRef CAS PubMed.
- C. Weiss, M. Carriere, L. Fusco, I. Capua, J. A. Regla-Nava, M. Pasquali, J. A. Scott, F. Vitale, M. A. Unal and C. Mattevi, ACS Nano, 2020, 14, 6383–6406 CrossRef CAS PubMed.
- M. C. Sportelli, M. Izzi, E. A. Kukushkina, S. I. Hossain, R. A. Picca, N. Ditaranto and N. Cioffi, Nanomaterials, 2020, 10, 802 CrossRef CAS PubMed.
- H. Huang, C. Fan, M. Li, H.-L. Nie, F.-B. Wang, H. Wang, R. Wang, J. Xia, X. Zheng and X. Zuo, ACS Nano, 2020, 14, 3747–3754 CrossRef CAS PubMed.
- Y.-N. Chen, Y.-H. Hsueh, C.-T. Hsieh, D.-Y. Tzou and P.-L. Chang, Int. J. Environ. Res. Public Health, 2016, 13, 430 CrossRef PubMed.
- T. Kisby, A. Yilmazer and K. Kostarelos, Nat. Nanotechnol., 2021, 1–8, DOI:10.1038/s41565-021-00946-9.
- A. Srivastava, N. Dwivedi, C. Dhand, R. Khan, N. Sathish, M. K. Gupta, R. Kumar and S. Kumar, Mater. Today Chem., 2020, 100385, DOI:10.1016/j.mtchem.2020.100385.
- L. Mei, S. Zhu, W. Yin, C. Chen, G. Nie, Z. Gu and Y. Zhao, Theranostics, 2020, 10, 757 CrossRef CAS PubMed.
- Y. Gogotsi and B. Anasori, ACS Nano, 2019, 13(8), 8491–8494 CrossRef CAS PubMed.
- W. Sun and F. G. Wu, Chem. – Asian J., 2018, 13, 3378–3410 CrossRef CAS PubMed.
- R. Kurapati, K. Kostarelos, M. Prato and A. Bianco, Adv. Mater., 2016, 28, 6052–6074 CrossRef CAS PubMed.
- H. Zhang, ACS Nano, 2015, 9, 9451–9469 CrossRef CAS PubMed.
- K. Rasool, M. Helal, A. Ali, C. E. Ren, Y. Gogotsi and K. A. Mahmoud, ACS Nano, 2016, 10, 3674–3684 CrossRef CAS PubMed.
- R. Hu, G. Liao, Z. Huang, H. Qiao, H. Liu, Y. Shu, B. Wang and X. Qi, J. Hazard. Mater., 2020, 124179 Search PubMed.
- Q.-Z. Yang, Y.-Y. Chang and H.-Z. Zhao, Water Res., 2013, 47, 6712–6718 CrossRef CAS PubMed.
- I. Maqbool, F. Rehman, F. Soomro, Z. Bhatti, U. Ali, A. H. Jatoi, B. Lal, M. Iqbal, S. Phulpoto and A. Ali, ChemBioEng Rev., 2021, 8, 67–77 CrossRef CAS.
- Z. Xie, Y.-P. Peng, L. Yu, C. Xing, M. Qiu, J. Hu and H. Zhang, Sol. RRL, 2020, 4, 1900400 CrossRef.
- H. E. Karahan, C. Wiraja, C. Xu, J. Wei, Y. Wang, L. Wang, F. Liu and Y. Chen, Adv. Healthcare Mater., 2018, 7, 1701406 CrossRef PubMed.
- D. V. Andreeva and K. S. Novoselov, Natl. Sci. Rev., 2021, nwab095 CrossRef.
- M. Khedri, R. Maleki, M. Dahri, M. M. Sadeghi, S. Rezvantalab, H. A. Santos and M.-A. Shahbazi, Drug Delivery Transl. Res., 2021, 1–15 Search PubMed.
- C. Balagna, S. Perero, E. Percivalle, E. V. Nepita and M. Ferraris, Open Ceram., 2020, 1, 100006 CrossRef.
- Q.-S. Rao, S.-Y. Liao, X.-W. Huang, Y.-Z. Li, Y.-D. Liu and Y.-G. Min, Polymers, 2020, 12, 2192 CrossRef CAS PubMed.
- S. Bhattacharjee, R. Joshi, A. A. Chughtai and C. R. Macintyre, Adv. Mater. Interfaces, 2019, 6, 1900622 CrossRef CAS PubMed.
- M. Hasani and M. Montazer, J. Text. Inst., 2017, 108, 1777–1786 CrossRef CAS.
- H. Zhong, Z. Zhu, J. Lin, C. F. Cheung, V. L. Lu, F. Yan, C.-Y. Chan and G. Li, ACS Nano, 2020, 14, 6213–6221 CrossRef CAS PubMed.
- Graphene Mask, https://www.bonbouton.com/graphene-mask, (accessed September 19, 2021).
- . A COVID-19 Virucidal Graphene-based Composite Ink for More Effective PPE, https://statnano.com/news/67657/A-COVID-19-Virucidal-Graphene-based-Composite-Ink-for-More-Effective-PPE, (accessed August 18, 2021).
- . GRAPHENE-ENHANCED FACE MASK, PACK OF 3, https://www.planartech.com/store/p1/graphene-enhanced-face-mask-pack-of-3.html, (accessed September 19, 2021).
- . Guardian G-Volt masks use graphene and electrical charge to repel viruses, https://grapheneindustry.org.au/2020/05/g-volt-mask/, (accessed August 18, 2021).
- R. K. Matharu, H. Porwal, B. Chen, L. Ciric and M. Edirisinghe, Med. Devices Sens., 2020, 3, e10107 CAS.
- T. LaTourrette, D. Peterson, J. T. Bartis, B. A. Jackson and A. Houser, Protecting emergency responders volume 2: community views of safety and health risks and personal protection needs, Rand Corporation, 2003 Search PubMed.
- H. Honda and K. Iwata, Curr. Opin. Infect. Dis., 2016, 29, 400–406 CrossRef PubMed.
- H. Miao, Z. Teng, C. Wang, H. Chong and G. Wang, Chem. – Eur. J., 2019, 25, 929–944 CrossRef CAS PubMed.
- J. Manna, S. Goswami, N. Shilpa, N. Sahu and R. K. Rana, ACS Appl. Mater. Interfaces, 2015, 7, 8076–8082 CrossRef CAS PubMed.
- M. Santosham, A. Chandran, S. Fitzwater, C. Fischer-Walker, A. H. Baqui and R. Black, Lancet, 2010, 376, 63–67 CrossRef.
- E. M. Anastasi, T. D. Wohlsen, H. Stratton and M. Katouli, Water Res., 2013, 47, 6670–6679 CrossRef CAS PubMed.
- Y. Gilboa and E. Friedler, Water Res., 2008, 42, 1043–1050 CrossRef CAS PubMed.
- S. W. Krasner, H. S. Weinberg, S. D. Richardson, S. J. Pastor, R. Chinn, M. J. Sclimenti, G. D. Onstad and A. D. Thruston, Environ. Sci. Technol., 2006, 40, 7175–7185 CrossRef CAS PubMed.
- X. Fan, F. Yang, C. Nie, L. Ma, C. Cheng and R. Haag, Adv. Mater., 2021, 33, 2100637 CrossRef CAS PubMed.
- F. Vatansever, W. C. de Melo, P. Avci, D. Vecchio, M. Sadasivam, A. Gupta, R. Chandran, M. Karimi, N. A. Parizotto and R. Yin, FEMS Microbiol. Rev., 2013, 37, 955–989 CrossRef CAS PubMed.
- Q. Tang, S. Cao, T. Ma, X. Xiang, H. Luo, P. Borovskikh, R. D. Rodriguez, Q. Guo, L. Qiu and C. Cheng, Adv. Funct. Mater., 2021, 31, 2007475 CrossRef CAS.
- F. Hossain, O. J. Perales-Perez, S. Hwang and F. Román, Sci. Total Environ., 2014, 466, 1047–1059 CrossRef PubMed.
- A. Arabi Shamsabadi, M. Sharifian Gh, B. Anasori and M. Soroush, ACS Sustainable Chem. Eng., 2018, 6, 16586–16596 CrossRef CAS.
- Q. Li, S. Mahendra, D. Y. Lyon, L. Brunet, M. V. Liga, D. Li and P. J. Alvarez, Water Res., 2008, 42, 4591–4602 CrossRef CAS PubMed.
- T. Yun, G. H. Jeong, S. Padmajan Sasikala and S. O. Kim, APL Mater., 2020, 8, 070903 CrossRef CAS.
- A. M. Díez-Pascual, Nanomaterials, 2018, 8(6), 359 CrossRef PubMed.
- T. T. T. Vi, S. Rajesh Kumar, B. Rout, C.-H. Liu, C.-B. Wong, C.-W. Chang, C.-H. Chen, D. W. Chen and S. J. Lue, Nanomaterials, 2018, 8, 163 CrossRef PubMed.
- S. Ye, K. Shao, Z. Li, N. Guo, Y. Zuo, Q. Li, Z. Lu, L. Chen, Q. He and H. Han, ACS Appl. Mater. Interfaces, 2015, 7, 21571–21579 CrossRef CAS PubMed.
- Z. Song, X. Wang, G. Zhu, Q. Nian, H. Zhou, D. Yang, C. Qin and R. Tang, Small, 2015, 11, 1171–1176 CrossRef CAS PubMed.
- F. Alimohammadi, M. Sharifian Gh, N. H. Attanayake, A. C. Thenuwara, Y. Gogotsi, B. Anasori and D. R. Strongin, Langmuir, 2018, 34, 7192–7200 CrossRef CAS PubMed.
- F. Perreault, A. F. De Faria, S. Nejati and M. Elimelech, ACS Nano, 2015, 9, 7226–7236 CrossRef CAS PubMed.
- X. Zou, L. Zhang, Z. Wang and Y. Luo, J. Am. Chem. Soc., 2016, 138, 2064–2077 CrossRef CAS PubMed.
- X. Liu, G. Duan, W. Li, Z. Zhou and R. Zhou, RSC Adv., 2017, 7, 37873–37880 RSC.
- K. Krishnamoorthy, M. Veerapandian, K. Yun and S. J. Kim, Colloids Surf., B, 2013, 112, 521–524 CrossRef CAS PubMed.
- J. Ouyang, M. Wen, W. Chen, Y. Tan, Z. Liu, Q. Xu, K. Zeng, L. Deng and Y.-N. Liu, Chem. Commun., 2019, 55, 4877–4880 RSC.
- M. A. Unal, F. Bayrakdar, H. Nazir, O. Besbinar, C. Gurcan, N. Lozano, L. M. Arellano, S. Yalcin, O. Panatli and D. Celik, Small, 2021, 2101483, DOI:10.1002/smll.202101483.
- Z. Miao, L. Fan, X. Xie, Y. Ma, J. Xue, T. He and Z. Zha, ACS Appl. Mater. Interfaces, 2019, 11, 26664–26673 CrossRef CAS PubMed.
- X. Wang, F. Lv, T. Li, Y. Han, Z. Yi, M. Liu, J. Chang and C. Wu, ACS Nano, 2017, 11, 11337–11349 CrossRef CAS PubMed.
- W. Yin, L. Yan, J. Yu, G. Tian, L. Zhou, X. Zheng, X. Zhang, Y. Yong, J. Li and Z. Gu, ACS Nano, 2014, 8, 6922–6933 CrossRef CAS PubMed.
- S. S. Chou, B. Kaehr, J. Kim, B. M. Foley, M. De, P. E. Hopkins, J. Huang, C. J. Brinker and V. P. Dravid, Angew. Chem., Int. Ed., 2013, 52, 4160–4164 CrossRef CAS PubMed.
- X. Huang, X. Qi, F. Boey and H. Zhang, Chem. Soc. Rev., 2012, 41, 666–686 RSC.
- J. Yu, W. Yin, X. Zheng, G. Tian, X. Zhang, T. Bao, X. Dong, Z. Wang, Z. Gu and X. Ma, Theranostics, 2015, 5, 931 CrossRef CAS PubMed.
- K. Ma, Y. Li, Z. Wang, Y. Chen, X. Zhang, C. Chen, H. Yu, J. Huang, Z. Yang and X. Wang, ACS Appl. Mater. Interfaces, 2019, 11, 29630–29640 CrossRef CAS PubMed.
- A. Pal, T. K. Jana, T. Roy, A. Pradhan, R. Maiti, S. M. Choudhury and K. Chatterjee, ChemistrySelect, 2018, 3, 81–90 CrossRef CAS.
- G. R. Navale, C. S. Rout, K. N. Gohil, M. S. Dharne, D. J. Late and S. S. Shinde, RSC Adv., 2015, 5, 74726–74733 RSC.
- P. Murugesan, J. Moses and C. Anandharamakrishnan, J. Mater. Sci., 2019, 54, 12206–12235 CrossRef CAS.
- D. Wu, S. Yue, W. Wang, T. An, G. Li, H. Y. Yip, H. Zhao and P. K. Wong, Appl. Catal., B, 2016, 192, 35–45 CrossRef CAS.
- S. P. Adhikari, G. P. Awasthi, J. Lee, C. H. Park and C. S. Kim, RSC Adv., 2016, 6, 55079–55091 RSC.
- D. Wu, B. Wang, W. Wang, T. An, G. Li, T. W. Ng, H. Y. Yip, C. Xiong, H. K. Lee and P. K. Wong, J. Mater. Chem. A, 2015, 3, 15148–15155 RSC.
- J. Huang, W. Ho and X. Wang, Chem. Commun., 2014, 50, 4338–4340 RSC.
- S. Cao, J. Low, J. Yu and M. Jaroniec, Adv. Mater., 2015, 27, 2150–2176 CrossRef CAS PubMed.
- L. Tan, J. Li, X. Liu, Z. Cui, X. Yang, K. W. K. Yeung, H. Pan, Y. Zheng, X. Wang and S. Wu, Small, 2018, 14, 1703197 CrossRef PubMed.
- A. Wiehe, J. M. O'Brien and M. O. Senge, Photochem. Photobiol. Sci., 2019, 18, 2565–2612 CrossRef CAS PubMed.
- Q. Jia, Q. Song, P. Li and W. Huang, Adv. Healthcare Mater., 2019, 8, 1900608 CrossRef PubMed.
- D. K. Ji, Y. Zhang, Y. Zang, J. Li, G. R. Chen, X. P. He and H. Tian, Adv. Mater., 2016, 28, 9356–9363 CrossRef CAS PubMed.
- J. Eke, K. Elder and I. C. Escobar, Membranes, 2018, 8, 79 CrossRef PubMed.
- Y. Liu, Y. Xiao, Y. Cao, Z. Guo, F. Li and L. Wang, Adv. Funct. Mater., 2020, 30, 2003196 CrossRef CAS.
- Y. Zhao, C. J. Wang, W. Gao, B. Li, Q. Wang, L. Zheng, M. Wei, D. G. Evans, X. Duan and D. O'Hare, J. Mater. Chem. B, 2013, 1, 5988–5994 RSC.
- S. A. Moaty, A. Farghali and R. Khaled, Mater. Sci. Eng., C, 2016, 68, 184–193 CrossRef PubMed.
- A. M. Schrand, S. A. C. Hens and O. A. Shenderova, Crit. Rev. Solid State Mater. Sci., 2009, 34, 18–74 CrossRef CAS.
- Z. P. Xu, Q. H. Zeng, G. Q. Lu and A. B. Yu, Chem. Eng. Sci., 2006, 61, 1027–1040 CrossRef CAS.
- S. Kango, S. Kalia, A. Celli, J. Njuguna, Y. Habibi and R. Kumar, Prog. Polym. Sci., 2013, 38, 1232–1261 CrossRef CAS.
- J. Ahlawat and M. Narayan, in Intelligent Nanomaterials for Drug Delivery Applications, Elsevier, 2020, pp. 1–16, DOI:10.1016/B978-0-12-817830-0.00001-1.
- F.-D. Cojocaru, D. Botezat, I. Gardikiotis, C.-M. Uritu, G. Dodi, L. Trandafir, C. Rezus, E. Rezus, B.-I. Tamba and C.-T. Mihai, Pharmaceutics, 2020, 12, 171 CrossRef PubMed.
- L. Singh, H. G. Kruger, G. E. Maguire, T. Govender and R. Parboosing, Ther. Adv. Infect. Dis., 2017, 4, 105–131 CAS.
- R. Parboosing, G. E. Maguire, P. Govender and H. G. Kruger, Viruses, 2012, 4, 488–520 CrossRef CAS PubMed.
- A. Kumar, H. Ma, X. Zhang, K. Huang, S. Jin, J. Liu, T. Wei, W. Cao, G. Zou and X.-J. Liang, Biomaterials, 2012, 33, 1180–1189 CrossRef CAS PubMed.
- S. E. McNeil, in Characterization of nanoparticles intended for drug delivery, Springer, 2011, pp. 3–8, DOI:10.1007/978-1-60327-198-1_1.
- R. A. Petros and J. M. DeSimone, Nat. Rev. Drug Discovery, 2010, 9, 615–627 CrossRef CAS PubMed.
- J. Caron, L. H. Reddy, S. Lepêtre-Mouelhi, S. Wack, P. Clayette, C. Rogez-Kreuz, R. Yousfi, P. Couvreur and D. Desmaële, Bioorg. Med. Chem. Lett., 2010, 20, 2761–2764 CrossRef CAS PubMed.
- S. Mukherjee, P. Mazumder, M. Joshi, C. Joshi, S. V. Dalvi and M. Kumar, Environ. Res., 2020, 191, 110119 CrossRef CAS PubMed.
- P. Innocenzi and L. Stagi, Chem. Sci., 2020, 11, 6606–6622 RSC.
- X. Du, R. Xiao, H. Fu, Z. Yuan, W. Zhang, L. Yin, C. He, C. Li, J. Zhou and G. Liu, Mater. Sci. Eng., C, 2019, 105, 110052 CrossRef CAS PubMed.
- H. Roy, S. Bhanja, U. P. Panigrahy and V. K. Theendra, in Characterization and Biology of Nanomaterials for Drug Delivery, Elsevier, 2019, pp. 77–111, DOI:10.1016/B978-0-12-814031-4.00004-0.
- M. Sametband, I. Kalt, A. Gedanken and R. Sarid, ACS Appl. Mater. Interfaces, 2014, 6, 1228–1235 CrossRef CAS PubMed.
- A. R. Deokar, A. P. Nagvenkar, I. Kalt, L. Shani, Y. Yeshurun, A. Gedanken and R. Sarid, Bioconjugate Chem., 2017, 28, 1115–1122 CrossRef CAS PubMed.
- I. S. Donskyi, W. Azab, J. L. Cuellar-Camacho, G. Guday, A. Lippitz, W. E. Unger, K. Osterrieder, M. Adeli and R. Haag, Nanoscale, 2019, 11, 15804–15809 RSC.
- M. A. Unal, F. Bayrakdar, L. Fusco, O. Besbinar, C. E. Shuck, S. Yalcin, M. T. Erken, A. Ozkul, C. Gurcan and O. Panatli, Nano Today, 2021, 38, 101136 CrossRef CAS PubMed.
- T. Kelf, V. Sreenivasan, J. Sun, E. Kim, E. Goldys and A. Zvyagin, Nanotechnology, 2010, 21, 285105 CrossRef CAS PubMed.
- L. Singh, R. Parboosing, H. G. Kruger, G. E. Maguire and T. Govender, Adv. Nat. Sci.: Nanosci. Nanotechnol., 2016, 7, 045013 Search PubMed.
- https://www.gisaid.org , (accessed September 20, 2021).
- N. Chaudhary, D. Weissman and K. A. Whitehead, Nat. Rev. Drug Discovery, 2021, 1–22, DOI:10.1038/s41573-021-00283-5.
- H. Y. Choi, T.-J. Lee, G.-M. Yang, J. Oh, J. Won, J. Han, G.-J. Jeong, J. Kim, J.-H. Kim and B.-S. Kim, J. Controlled Release, 2016, 235, 222–235 CrossRef CAS PubMed.
- A. Gazzi, L. Fusco, M. Orecchioni, S. Ferrari, G. Franzoni, J. S. Yan, M. Rieckher, G. Peng, M. A. Lucherelli and I. A. Vacchi, J. Phys.: Mater., 2020, 3, 034009 CAS.
- B. Luo, G. Liu and L. Wang, Nanoscale, 2016, 8, 6904–6920 RSC.
- P. Ganguly, M. Harb, Z. Cao, L. Cavallo, A. Breen, S. Dervin, D. D. Dionysiou and S. C. Pillai, ACS Energy Lett., 2019, 4, 1687–1709 CrossRef CAS.
- L. Fusco, A. Gazzi, G. Peng, Y. Shin, S. Vranic, D. Bedognetti, F. Vitale, A. Yilmazer, X. Feng and B. Fadeel, Theranostics, 2020, 10, 5435 CrossRef CAS PubMed.
- A. Gazzi, L. Fusco, A. Khan, D. Bedognetti, B. Zavan, F. Vitale, A. Yilmazer and L. G. Delogu, Front. Bioeng. Biotechnol., 2019, 7, 295 CrossRef PubMed.
- C. Zhang, Y. Li, D. Shuai, W. Zhang, L. Niu, L. Wang and H. Zhang, Chemosphere, 2018, 208, 84–92 CrossRef CAS PubMed.
- C. Zhang, Y. Li, W. Zhang, P. Wang and C. Wang, Chemosphere, 2018, 195, 551–558 CrossRef CAS PubMed.
- H. Mohammed, A. Kumar, E. Bekyarova, Y. Al-Hadeethi, X. Zhang, M. Chen, M. S. Ansari, A. Cochis and L. Rimondini, Front. Bioeng. Biotechnol., 2020, 8, 465 CrossRef PubMed.
- F. De Maio, V. Palmieri, G. Babini, A. Augello, I. Palucci, G. Perini, A. Salustri, P. Spilman, M. De Spirito and M. Sanguinetti, Iscience, 2021, 24, 102788 CrossRef CAS PubMed.
- R. P. Pandey, K. Rasool, V. E. Madhavan, B. Aïssa, Y. Gogotsi and K. A. Mahmoud, J. Mater. Chem. A, 2018, 6, 3522–3533 RSC.
- A. M. Jastrzębska, E. Karwowska, T. Wojciechowski, W. Ziemkowska, A. Rozmysłowska, L. Chlubny and A. Olszyna, J. Mater. Eng. Perform., 2019, 28, 1272–1277 CrossRef.
- C. Zhang, Y. Wang, J. Ma, Q. Zhang, F. Wang, X. Liu and T. Xia, Sci. Total Environ., 2020, 721, 137740 CrossRef CAS PubMed.
- J. Ouyang, R.-Y. Liu, W. Chen, Z. Liu, Q. Xu, K. Zeng, L. Deng, L. Shen and Y.-N. Liu, J. Mater. Chem. B, 2018, 6, 6302–6310 RSC.
| This journal is © The Royal Society of Chemistry 2022 |