Joseph A. Izzo, Yaroslaw Myshchuk, Jennifer S. Hirschi and Mathew J. Vetticatt
Org. Biomol. Chem., 2019,17, 3934-3939
DOI:
10.1039/C9OB00072K,
Paper
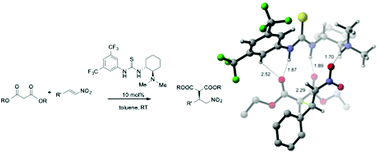
The mechanism of the enantioselective Michael addition of diethyl malonate to trans-β-nitrostyrene catalyzed by a tertiary amine thiourea organocatalyst is explored using experimental 13C kinetic isotope effects and density functional theory calculations. Large primary 13C KIEs on the bond-forming carbon atoms of both reactants suggest that carbon–carbon bond formation is the rate-determining step in the catalytic cycle. This work resolves conflicting mechanistic pictures that have emerged from prior experimental and computational studies.