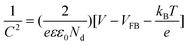DOI:
10.1039/C2RA21979D
(Paper)
RSC Adv., 2012,
2, 12455-12459
Hierarchical Cu2O photocathodes with nano/microspheres for solar hydrogen generation†
Received 30th August 2012, Accepted 17th October 2012
First published on 19th October 2012
Abstract
A hierarchical p-type Cu2O film with nano/microspheres on copper foil is successfully synthesized via a facile and cost-effective liquid reduction route through transformation of a lotus-like CuO/Cu(OH)2 nanosheet/nanowire structure. Various sizes of sphere-like Cu2O are transformed from CuO nanosheets and Cu(OH)2 nanowires by chemically reducing the oxides from Cu2+ to Cu1+ in a solution of ascorbic acid at a low temperature of 60 °C. Mott–Schottky analysis shows the flat band potential of the sphere-like Cu2O film to be −0.11 V and an acceptor concentration of 2.7 × 1020 cm−3. A direct band gap of 2.02 eV in Cu2O film is determined by incident photon-to-electron conversion efficiency measurements. Significantly, this hierarchical Cu2O photocathode exhibits remarkable photoelectrochemical activity in visible light. These results demonstrate that the Cu2O film with nano/microspheres has great potential in solar hydrogen applications.
1. Introduction
Of all the clean energy production methods, solar-powered splitting of water into hydrogen and oxygen is a favorable means of converting solar energy into a “solar fuel”, because both water and sunlight are vastly abundant.1–6 The generation of hydrogen by photoelectrochemical (PEC) water decomposition under solar illumination is much simpler and more environmentally friendly than the catalytic reforming of hydrocarbon fuels.7–10 Typically, solar conversion efficiency and the stability of PEC water decomposition are determined by the properties of the semiconductor materials that are used as photoelectrodes. The materials that are being developed for use in photoelectrodes, such as TiO2, GaN and ZnO, have been modified to shift their energy band gaps by substitutional element doping to extend their photoresponse to visible light.11–13 However, the inherently unavoidable trap centers, which are created by dopants, decrease their photoresponse significantly. Hence, cuprous oxide (Cu2O) with a direct band gap of 2.1 eV exhibits great potential applications in solar energy conversion. Besides the p-type photoactive materials, copper is abundant and the materials can be processed by industrially proven, low-cost methods.14,15 Recently, A. Paracchino et al. reported the highly active photocathode of a Cu2O thin film, which can achieve photocurrents of −7.6 mA cm−2 at a potential of 0 V and remained active for 1 h of testing.14 These results demonstrate the great potential of Cu2O for PEC water decomposition. Actually, the PEC activity can be improved by the use of nanometer-sized materials, which are particularly relevant on account of their high surface-to-volume ratio and short diffusion length for carrier transport compared with their bulk counterparts.15 Up to now, various Cu2O nanostructures, including nanowires, nanotubes, nanocubes, nanospheres, and so on, have been successfully fabricated using a variety of techniques.16–19 It is noticed that hierarchical Cu2O nano/microspheres have attracted more and more attention. From literature reports, such materials are expected to exhibit high light-collection efficiency and fast motion of charge carriers, owing to their hierarchical structures, closely packed interpenetrating networks, and large surface area.20 In fact, different approaches have been reported to prepare Cu2O nano/microspheres such as template-directed synthesis21 and hydrothermal synthesis.20 However, these Cu2O nano/microspheres exist in powder form, and have poor carrier transport paths. The best strategy for PEC photoelectrodes is to fabricate a continuity of Cu2O nano/microspheres within the film. To date, there has been little progress in preparing Cu2O films made up of hierarchical nano/microspheres as photoelectrodes.In this work, a facile liquid reduction route for directly grown Cu2O nano/microsphere films on copper foil as a photocathode is proposed by means of the chemical reduction of lotus-like CuO/Cu(OH)2 nanosheets/nanowires as templates. Typically, the formation of Cu2O from CuO reduction requires high temperature in vacuum and/or H2 atmosphere, and also restricts the initial CuO particle size to within 10 nm.22 To our knowledge, the transformation of CuO to Cu2O through chemical reduction at low temperature (below 100 °C), and the fabrication of a Cu2O film with a hierarchical nano/microsphere structure for PEC water decomposition has not been reported yet. Moreover, these Cu2O nano/microspheres films not only function as photocathodes for PEC hydrogen generation, but also offer a new opportunity to develop electronic and photoelectronic devices based on 3D hierarchical nanomaterials.
2. Experimental
2.1. Fabrication of Cu2O nano/microspheres
Firstly, the flower-like CuO and Cu(OH)2 nanowire arrays on copper foil were fabricated via chemical oxidation reaction without any template.23 An aqueous solution was prepared in a 100 mL glass bottle; a 2.5 mol L−1 NaOH and 0.125 mol L−1 (NH4)2S2O8 solution was mixed and the total solution volume was kept at 50 mL. A piece of copper foil (10 × 20 × 2 mm3), which had been ultrasonically cleaned in acetone and subsequently in de-ionized water, was immersed in the solution. A few minutes later, a faint blue color appeared on the copper foil surface, and the initial colorless solution became increasingly blue. In 10 min, a light-blue film covered the copper foil surface. When the color of the film became dark blue (reaction time of 30 min), the copper foil was extracted from the solution, rinsed with water and ethanol, and dried in air. In the second process of chemical reduction, a piece of as-prepared CuO/Cu(OH)2 was immersed in the solution of ascorbic acid (10 mM) and then the solution was kept at 60 °C for reaction times of 20, 40, 60, and 80 min. In this process, the dark blue CuO/Cu(OH)2 samples turned orange, while the surrounding solution still remained colorless. The product was carefully taken out of the solution, and rinsed with distilled water. The as-grown sample was prepared by bonding a copper wire onto the edge of approximately 1 × 1 cm2. The bonding was accomplished by using silver paste, cured for 20 min at 50 °C. The bonding pad was covered with epoxide to expose only the copper oxide surface to the test solutions.2.2. Characterization of materials
The morphology of the hierarchical Cu2O nano/microsphere arrays was examined by scanning electron microscopy (SEM, JEM-4000EX), and the structure of the samples was analyzed using an X-ray diffractometer (XRD, Bruker D8 Advance diffractometer) with Cu-Kα radiation (λ = 0.1506 nm). The chemical states of the elements were determined by X-ray photoelectron spectroscopy (XPS, Perkin-Elmer model PHI 1600). The photoelectrochemical behavior of the electrodes was measured in 0.5 M Na2SO4 solution by a CHI 627D potentiostat/galvanostat. A conventional three-electrode system consisting of the Cu2O nano/microsphere array as the working electrode, a square platinum sheet as the auxiliary electrode, and an Ag/AgCl reference electrode in 3 M KCl solution was implemented. All potentials reported in this article were referenced to Ag/AgCl (3 M KCl, 0.207 V vs. SHE). A 150 W Xe lamp light source with an AM 1.5 filter was used, and the intensity of the illumination at the sample position was determined to be 100 mW cm−2. In addition, a 150 W Xe lamp equipped with a monochromator was used as the excitation light source to obtain monochromatic light for incident photon-to-electron conversion efficiency measurements. The incident light was irradiated onto the Cu2O nano/microsphere electrodes from the front face through a quartz window and the electrolyte unless noted otherwise.3. Results and discussion
3.1. Structure and composition characterization
Fig. 1a shows the hierarchical structures of CuO and Cu(OH)2 on a copper substrate by chemical oxidation. The flower-like microspheres of CuO reveal a diameter of 3–5 μm uniformly with the thickness of the flower petals of approximately 50–100 nm. Apparently, the Cu(OH)2 nanowire arrays are formed under the microflowers, and the diameter of the nanowires is approximately 100 nm; in other words, the microflowers are standing on the nanowire arrays, similar to a lotus leaf. Under the chemical reduction for a reaction time of 20 min, the wire-like Cu(OH)2 dramatically converts into microspheres with a diameter of 1 μm, and the flower-like CuO seems not to change, as shown in Fig. 1b. However, the magnified image of a single micro-flower (Fig. 1c) reveals that, in fact, the partial petals transfer into nanospheres with a diameter of 100 nm. With increasing reaction time of 40, 60, and 80 min, each petal gradually forms nanospheres in serial, as shown in Fig. 1d, 1e, and 1f, respectively, and the complete transformation of the shape of the nanospheres can be achieved at a reaction time of 60 min. Thus, conversion of the lotus-like nanostructure into hierarchical micro- and nanosphere structures was carried out by the chemical reduction route. |
| | Fig. 1 (a) FESEM images of flower-like CuO standing on Cu(OH)2 nanowires; (b) FESEM image of Cu2O nano/microspheres with reaction time of 20 min; magnified FESEM images of Cu2O nano/microspheres for reaction time of (c) 20 min, (d) 40 min, (e) 60 min, and (f) 80 min. | |
The crystal phase of the samples with chemical oxidation and subsequent chemical reduction was determined using powder XRD, as shown in Fig. 2. The XRD pattern of copper foil is also displayed for comparison. After chemical oxidation, the XRD pattern of the lotus-like microflowers/nanowire sample shows that the diffraction peaks marked with reverse triangles, indexed to monoclinic-phase CuO (JCPDS card No. 80-0076), appear along with those marked with an asterisk of the copper substrate, and orthorhombic phase Cu(OH)2 marked with triangles. Under the chemical reduction of 20 min, the diffraction peaks from orthorhombic phase Cu(OH)2 disappear, and then appearance of Cu2O marked with circles are identified as cubic-phase (JCPDS card No. 78-2076). This evolution of structures is consistent with the findings of SEM, and also confirms that the microspheres belong to Cu2O. With increasing reaction time, the diffraction peaks from Cu2O become stronger; meanwhile the intensity of the CuO peaks disappear at a reaction time of 60 min. However, the diffraction peak of Cu2O slightly decreases at a reaction time of 80 min, because partial sphere-like Cu2O peels off from the Cu foil. It is worth noting that the formation of pure Cu2O phase can be accomplished by the complete transformation of lotus-like CuO/Cu(OH)2 under chemical reduction at low temperature.
 |
| | Fig. 2 X-Ray diffraction patterns of Cu2O nano/microspheres for various reaction times. | |
From the XPS measurements, the Cu 2p peaks were resolved, as shown in Fig. 3. The Cu 2p peaks of the lotus-like microflowers/nanowire sample at 934.4 eV and 954.2 eV, corresponding to Cu 2p3/2 and 2p1/2, respectively, reveal an oxidation state of +2. The occurrence of a satellite feature on the higher binding energy side (943.2 eV) of the Cu 2p main peak indicates the presence of CuO, because cupric oxide (Cu2+) has hole states in the Cu 3d band (Cu 3d9 configuration). The 3d band of cuprous oxide (Cu+) is filled (Cu 3d10), and the 4s band is unoccupied, thus no satellites are expected. Therefore, as the reaction time of chemical reduction increases, the Cu 2p3/2 and 2p1/2 peaks shift to the low-energy side of 932.2 eV and 952.1 eV, corresponding to an oxidation state of +1; meanwhile the intensity of the high energy peaks and satellites decrease dramatically with increasing reduction time. This result confirms the complete reduction of oxide species with Cu2+ to Cu2O, which agrees with the findings of XRD. Moreover, the observed amount of CuO is very tiny due to the slightly oxidized surface of Cu2O under their exposure to the atmosphere. All of these results indicate that the chemical reduction method can effectively and completely transfer CuO/Cu(OH)2 to Cu2O.
 |
| | Fig. 3 Cu 2p XPS spectra of Cu2O nano/microspheres for various reaction times. | |
3.2. Mechanism of sphere-like Cu2O formation
The formation of the Cu2O micro- and nanospheres via chemical oxidation and subsequent chemical reduction are schematically depicted in Scheme 1. The fabrication of a CuO/Cu(OH)2 nanosheet/nanowire structure by means of (NH4)2S2O8 oxidant in alkaline solution is reported elsewhere.23 The fundamental chemical oxidation reaction is expressed as follows:| | | Cu + 4NaOH + (NH4)2S2O8 → Cu(OH)2 + 2Na2SO4 + 2NH3 + 2H2O | (1) |
 |
| | Scheme 1 Schematic illustration of fabrication of the Cu2O nano/microspheres. | |
Importantly, the conversion of lotus-like CuO/Cu(OH)2 nanosheets/nanowires to Cu2O nano/microspheres is significant and is believed to be a result of the Kirkendall effect. The fundamental reactions involved in our experiment could be expressed as follows:17
| | | C6H8O6 + 2Cu(OH)2 → C6H6O6 + Cu2O + 3H2O | (2) |
| | | C6H8O6 + 2CuO → C6H6O6 + Cu2O + H2O | (3) |
Herein, the ascorbic acid functions as a reduction agent during the reaction. When the array of lotus-like copper oxide is immersed in the ascorbic acid solution, a thin layer of Cu2O is first nucleated and formed on the surface of each CuO nanosheet and Cu(OH)2 nanowire based on eqn (1) and (2), respectively.
This thin Cu2O layer acts as an interface, which separates the inner CuO and Cu(OH)2 from the ascorbic acid molecules outside. Hence, direct chemical reaction is hindered, and further reaction depends on the outward diffusion of copper ions and/or inward diffusion of ascorbic acid molecules through the interface. Meanwhile, there will be a concentration gradient of ascorbic acid molecules normal to the surface of the CuO and Cu(OH)2. This will drive the inward diffusion of ascorbic acid through the Cu2O layer. Copper ions and ascorbic acid molecules react to produce Cu2O crystallites, which aggregate into polycrystalline nanospheres around the interface, while the by-products of C6H6O6 and H2O can get out of the Cu2O layers. With the reaction going on, sphere-like Cu2O structures are yielded. According to the observations of chemical reduction, however, the conversion of Cu(OH)2 is accomplished within a reaction time of 20 min, but the conversion of CuO takes 60 min for complete reduction. The variation in reaction rate can be ascribed to the fact that the surface energy of Cu(OH)2 is lower than that of CuO.24 In addition, the difference of the reduction rate and the shape of the oxide precursors may result in the formation of Cu2O micro- and nanospheres.
3.3. PEC characterization
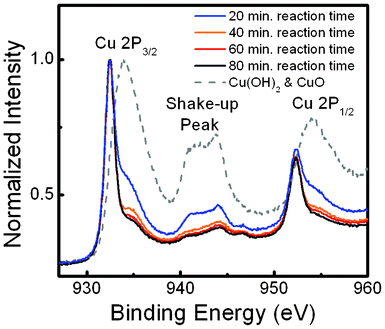
To investigate the PEC characteristics of the Cu2O nano/microsphere film as a photocathode for the solar generation of hydrogen by the decomposition water, a three-electrode electrochemical setup was adopted. Here, chopped light was periodically illuminated on the sample at a rate of 2 s exposure following every 2 s unilluminated, and the current was recorded while the voltage was changed from 0 to −0.5 V with a scan rate of 10 mV s−1. For all samples, a clear increase in the magnitude of the cathodic photocurrent was seen under light exposure, as shown in Fig. 4. These photo-induced cathodic currents result from the reduction of protons involving the photo-generation of electrons, revealing the p-type nature of the sphere-like Cu2O hierarchical photoelectrode. At a 20 min reaction time, a weak photocurrent and large dark-current can be ascribed to the lesser amount of Cu2O phase and poor crystallization of CuO caused by the pseudo-capacitive nature, respectively.23 As the reaction time increases, the appearance of Cu2O and reduction of CuO in the samples significantly enhance the photocurrent and diminish the dark current at the same time. Meanwhile, it is noteworthy that the optimum photocurrent for the Cu2O nano/microspheres can be achieved at a reduction time of 60 min. In comparison with Cu2O thin film electrodes,25 the Cu2O nano/microsphere electrode shows approximately 3 times the photocurrent density. As the reaction time exceeds 80 min, the partly peeled Cu2O from Cu foil results in a decreased photocurrent. Furthermore, the photocurrent action spectra (Fig. 5) plots the incident photon-to-electron conversion efficiency (IPCE) as a function of excitation wavelength at a potential of −0.4 V. Significantly, the photoresponse of Cu2O shows the onset wavelength for photocurrent generation at around 620 nm in the electrolyte. Furthermore, the gradual increase in the IPCE, η, with increasing photon energy is related to band gap energy by the following equation:10| | | ηhν = A(L + W)(hν − Eg)m | (4) |
where Eg is the bandgap energy while the exponent m equals 1/2 for a direct and 2 for an indirect electronic transition. W is the width of the space charge layer. L is the diffusion length. The inset of Fig. 5 shows that eqn (4) with m = 1/2 is closely followed in a range of photon energies close to the absorption threshold, indicating that the optical transition near the band gap is direct. From the intercept of the straight line with the abscissa, the band gap energy for the Cu2O nano/microspheres is derived as 2.02 eV. This result agrees with the literature reports.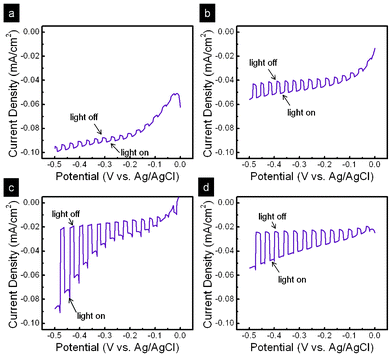 |
| | Fig. 4 Photocurrent–voltage responses of Cu2O nano/microspheres for reaction time of (a) 20 min, (b) 40 min, (c) 60 min, and (d) 80 min. | |
 |
| | Fig. 5 IPCE as a function of excitation wavelength at a potential of −0.4 V from Cu2O nano/microspheres under a reaction time of 60 min; inset, variation of the square root of IPCE (h) times hv with photon energy for Cu2O nano/microspheres. | |
Electrochemical impedance spectroscopy measurements were made on sphere-like Cu2O hierarchical photocathodes in the dark. The capacitances at the semiconductor–electrolyte-interface (SEI) with the use of an equivalent circuit are described by a Mott–Schottky plot, in order to estimate the flat-band position for Cu2O. Fig. 6 shows a Mott–Schottky plot of data analyzed by using the CNLS fitting method based on an equivalent circuit, as shown in the inset. However, the Mott–Schottky equation relates the capacitance of the semiconductor to the carrier concentration (Nd) and the other constants, such as the fundamental charge constant (e), dielectric constant (ε of Cu2O is 7.6), vacuum permittivity (εo), temperature (T), Boltzmann constant (kB), and the flat-band potential (VFB):26
| | 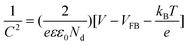 | (5) |
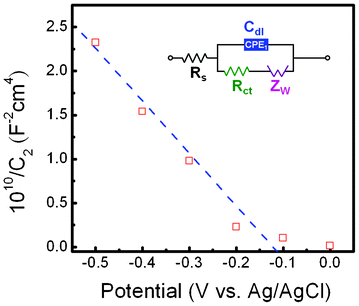 |
| | Fig. 6 Mott–Schottky plot of Cu2O nano/microspheres; inset, equivalent circuit. | |
The slope of the linear part of the curve in the Mott–Schottky plot is negative, indicating a p-type semiconductor. Furthermore, plotting 1/C2versus V allows the estimation of the flat-band and surface carrier concentration, with the flat-band being the x-intercept and the carrier concentration calculated from the slope. The value of the flat-band is located at −0.11 V vs. Ag/AgCl, which is close to the valence band. Estimation of the acceptor concentrations shows that in Cu2O the concentration is 2.7 × 1020 cm−3. The high value of Na can be attributed to a higher density of vacancies and the hydrous nature of oxide nano-materials, in agreement with previous reports.26,27
4. Conclusions
In summary, a hierarchical p-type Cu2O film with nano/microspheres on copper foil is successfully synthesized via a facile and cost-effective template route through transformation of lotus-like CuO/Cu(OH)2 nanosheets/nanowires. During chemical reduction, the Cu(OH)2 nanowires and CuO nanosheets transform to microspheres and nanospheres, respectively, based on the Kirkendall effect. As regards the evolution of structure and composition, CuO an Cu(OH)2 completely convert to Cu2O with ascorbic acid as the reducing agent at a low temperature of 60 °C. Furthermore, Mott–Schottky analysis reveals a flat-band potential of −0.11 V vs. Ag/AgCl and an acceptor concentration of 2.7 × 1020 cm−3 in the Cu2O nano/microspheres. The band gap energy, estimated at around 2.02 eV, of these sphere-like Cu2O films is suited for the efficient utilization of solar light. From the PEC measurements, the optimum active photocathode of Cu2O film with nano/microspheres can be achieved under a reaction time of 60 min. Significantly, our study suggests that these hierarchical Cu2O nano/microspheres are not only promising for solar hydrogen generation, but also offer a new opportunity to develop electronic and photoelectronic devices based on 3D hierarchical nanomaterials.Acknowledgements
The authors would like to thank the National Dong Hwa University and the National Science Council of the Republic of China, Taiwan, for financially supporting this research under Contract No. NSC 101-2221-E-259-011.References
- O. Khaselev and J. A. Turner, Science, 1998, 280, 425 CrossRef CAS.
- Y. K. Hsu, Y. G. Lin and Y. C. Chen, Electrochem. Commun., 2011, 13, 1383 CrossRef CAS.
- S. Martha, D. P. Das, N. Biswal and K. M. Parida, J. Mater. Chem., 2012, 22, 10695 RSC.
- K. M. Parida, S. Martha, D. P. Das and N. Biswal, J. Mater. Chem., 2010, 20, 7144 RSC.
- N. Biswal, D. P. Das, S. Martha and K. M. Parida, Int. J. Hydrogen Energy, 2011, 36, 13452 CrossRef CAS.
- K. M. Parida, N. Biswal, D. P. Das and S. Martha, Int. J. Hydrogen Energy, 2010, 35, 5262 CrossRef CAS.
- S. W. Boettcher, J. M. Spurgeon, C. Putnam, E. L. Warren, D. B. Turner-Evans, D. Kelzenberg, J. R. Maiolo, H. A. Atwater and N. S. Lewis, Science, 2010, 327, 185 CrossRef CAS.
- Y. G. Lin, Y. K. Hsu, S. Y. Chen, Y. K. Lin, L. C. Chen and K. H. Chen, J. Mater. Chem., 2011, 21, 324 RSC.
- A. Wolcott, W. A. Smith, T. R. Kuykendall, Y. P. Zhao and J. Z. Zhang, Adv. Funct. Mater., 2009, 19, 1849 CrossRef CAS.
- Y. K. Hsu, Y. C. Chen, Y. G. Lin, L. C. Chen and K. H. Chen, J. Mater. Chem., 2012, 22, 2733 RSC.
- I. M. Huygens, K. Strubbe and W. P. Gomes, J. Electrochem. Soc., 2000, 147, 1797 CrossRef CAS.
- S. Pedro, Prog. Surf. Sci., 2011, 86, 41 CrossRef.
- Y. K. Hsu and C. M. Lin, Electrochim. Acta, 2012, 74, 73 CrossRef CAS.
- A. Paracchino, V. Laporte, K. Sivula, M. Grätzel1 and E. Thimsen, Nat. Mater., 2011, 10, 456 CrossRef CAS.
- W. M. Jin, J. H. Kang and J. H. Moon, ACS Appl. Mater. Interfaces, 2010, 2, 2982 CAS.
- Y. Tan, X. Xue, Q. Peng, H. Zhao, T. Wang and Y. Li, Nano Lett., 2007, 7, 3723 CrossRef CAS.
- M. Cao, C. Hu, Y. Wang, Y. Guo, C. Guo and E. Wang, Chem. Commun., 2003, 1884 RSC.
- L. Gou and C. Murphy, Nano Lett., 2003, 3, 231 CrossRef CAS.
- J. Zhang, J. Liu, Q. Peng, X. Wang and Y. Li, Chem. Mater., 2006, 18, 867 CrossRef CAS.
- Y. Sui, Y. Zhang, W. Fu, H. Yang, Q. Zhao, P. Sun, D. Ma, M. Yuan, Y. Li and G. Zou, J. Cryst. Growth, 2009, 311, 2285 CrossRef CAS.
- W. Zhang, C. Luan, Z. Yang, X. Liu, D. Zhang and S. Yang, Appl. Surf. Sci., 2007, 253, 6063 CrossRef CAS.
- J. Pike, S. W. Chan, F. Zhang, X. Wang and J. Hanson, Appl. Catal., A, 2006, 303, 273 CrossRef CAS.
- Y. K. Hsu, C. Y. Chen and Y. G. Lin, J. Electroanal. Chem., 2012, 673, 43 CrossRef CAS.
- S. F. Cheah, G. E. Brown and G. A. Parks, Amer. Mineral., 2000, 85, 118 CAS.
- J. N. Nian, C. C. Hu and H. H. Teng, Int. J. Hydrogen Energy, 2008, 33, 2897 CrossRef CAS.
- Z. Zhang and P. Wang, J. Mater. Chem., 2012, 22, 2456 RSC.
- K. Nakaoka, J. Ueyama and K. Ogura, J. Electrochem. Soc., 2004, 151, C661 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: See DOI: 10.1039/c2ra21979d |
|
| This journal is © The Royal Society of Chemistry 2012 |
Click here to see how this site uses Cookies. View our privacy policy here.