DOI:
10.1039/C4RA13984D
(Paper)
RSC Adv., 2015,
5, 14103-14113
The role and activity of various adsorbed ammonia species on Cu/SAPO-34 catalyst during passive-SCR process†
Received
6th November 2014
, Accepted 9th January 2015
First published on 12th January 2015
Abstract
In this work, the adsorption and reaction performance of various adsorbed ammonia species during the passive-SCR process were investigated by temperature-programmed desorption (TPD), temperature-fixed surface reaction (TFSR), in situ diffuse reflectance infrared Fourier transform spectroscopy (in situ DRIFTS) and kinetic tests. The NH3-TPD and DRIFTS-NH3 adsorption results showed that the number of weak and strong acid sites in the Cu/SAPO-34 sample increased due to Cu species incorporation, which formed new Lewis acid sites. Further, a decrease in the number of moderate acid sites resulted from the replacement of hydroxyls by Cu2+. The TFSR results revealed that the adsorbed NH3 species presented different SCR activity, which could be divided into active NH3 species and inactive NH3 species. In the low temperature range, a large amount inactive NH3 resulted in lower activity and resulted in a long equilibrium process. However, the performance of inactive NH3 was distinctly different at high temperatures. Furthermore, the in situ DRIFTS results illustrated the activity difference and migration between the two adsorbed NH3 species. It was proved that the NH3 migration rate from the Brønsted acid sites to Lewis acid sites was slower than the SCR rate at low temperatures, which might determine the SCR reaction rates. However, at high temperatures, the NH3 migration rate was faster than the SCR rate of active NH3. In other words, NH3 migration from the Brønsted acid sites to Lewis acid sites may be the rate determining step of passive-SCR lean period at low temperatures, while NH3 adsorption possibly was the rate determining step of the passive-SCR rich period at high temperatures. On the basis of the above results, the present work gives an insight into the potential of Cu/SAPO-34 for passive-SCR applications in the future.
1. Introduction
A lean burn engine exhibits significant potential for improving fuel efficiency, increasing economy and savings, and reducing greenhouse gas emissions.1,2 However, with more and more strict emission regulations, lean period NOx generation beyond the regulations has restricted its wide applications.3–5 The existing de-NOx strategy requires a combination of various technologies, such as two of the most promising solutions under oxygen-rich condition: lean NOx trap (LNT) and ammonia selective catalytic reduction (NH3-SCR).1,6 Nevertheless, both technologies suffer considerable drawbacks because they demand supporting facility and increased cost of the after-treatment system.7,8 Recently, an efficient and cheaper lean NOx after treatment technology was reported by Li et al. that included passive selective catalytic reduction (passive-SCR),1 which could replace the complex combinations of various catalysts. A passive-SCR system itself contains two catalyst sections: TWC catalyst and NH3-SCR catalyst. During the rich period, NH3 is produced on the upstream TWC catalyst and stored on the downstream SCR catalyst. Subsequently, the generated NOx during the lean condition could be consumed by the adsorbed NH3 on the downstream SCR catalyst.3 Considering the dual role of SCR catalyst in the passive-SCR system, strong NH3 adsorption and storage capacity is necessary to trap ammonia during the rich operation avoiding NH3-slip. Subsequently, a high SCR activity may ensure NOx elimination from the lean period. On the basis of the above two requirements, Cu/SAPO-34 catalyst shows potential advantages, compared with traditional ZSM-5 and beta zeolite catalysts, due to its acidity, excellent SCR activity, high N2 selectivity and hydrothermal stability in a passive-SCR system.9–16
Brønsted and Lewis acid sites existing in Cu/SAPO-34 catalysts (chabazite structure) are necessary and important for NH3 adsorption.17 The Brønsted acid sites were generated by the isomorphous substitution of P by Si in AlPOs, which played an important role in binding and activating NH3 to react with NOx.18 Lewis acid sites were generated by exchanging the hydroxyls (Al–OH–Si) by Cu2+ ions in the Cu/SAPO-34 sample.19,20 Previous NH3-TPD results have been reported to assign the acidity of SAPO-34 and were compared with different silica–alumina zeolites.20–24 It provides us important information about the contribution of different zeolite structures to acid sites and metal ion incorporation effect on acidity. In addition, the titration experiments of the adsorbed NH3 were performed to speculate the mechanism of NH3-SCR reaction and Lewis acid sites were proposed to be SCR reaction active sites.21 Furthermore, Wang et al. discovered the NH3 migration issue and conjectured the existence of an NH3 migration phenomenon.22 Combining the above evolution, they formulated useful information regarding the basic acid sites and their properties. With respect to the passive-SCR process, there is no systematical research about NH3 adsorption behavior on various acid sites and their reaction performance. Whether Cu/SAPO-34 catalyst can be applied and if it meets the requirements needs to be verified. Especially, the reaction mechanism and rate-determining step of the passive-SCR process have not been proposed up to now. Consequently, it is worthy to study the adsorption and reaction behavior of ammonia species on Cu/SAPO-34 catalyst and investigate the guidelines for passive-SCR research.
In the present research, H/SAPO-34 support and Cu/SAPO-34 sample were synthesized to investigate the reaction performance of NH3, NO and O2 on a Cu/SAPO-34 catalyst. NH3-TPD and DRIFTS tests were performed to clarify the adsorption sites of NH3 species and the acid sites variation for Cu incorporation. Furthermore, TFSR and in situ DRIFTS experiments were applied to probe into the reactivity of active and inactive ammonia species on various adsorption sites. The NH3 migration between different acid sites was also researched. Finally, a kinetic test was employed to explore the possible rate determining step of passive-SCR reaction and provide an in-depth understanding about future passive-SCR applications.
2. Experiments
2.1 Catalyst preparation
H/SAPO-34 was synthesized via hydrothermal method with mole composition of 0.2 morpholine (MA): 0.1Al2O3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1P2O5
0.1P2O5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06SiO2
0.06SiO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.4H2O. The sources of Si, P and Al were silica sol, 85% phosphoric acid and pseudoboehmite, respectively. Firstly, the pseudoboehmite and phosphoric acid were mixed with water. The mixture was fiercely stirred for 1 h. Secondly, the MA and silica sol were mixed well and added dropwise into the former mixture. Thirdly, the blend was sealed in 200 mL Teflon-lined stainless steel pressure vessels and heated in an oven at 200 °C under autogenic pressure for 48 h. Finally, the H/SAPO-34 was obtained through centrifugal separation, washed, dried at 100 °C overnight and calcined at 650 °C in air for 6 h.
6.4H2O. The sources of Si, P and Al were silica sol, 85% phosphoric acid and pseudoboehmite, respectively. Firstly, the pseudoboehmite and phosphoric acid were mixed with water. The mixture was fiercely stirred for 1 h. Secondly, the MA and silica sol were mixed well and added dropwise into the former mixture. Thirdly, the blend was sealed in 200 mL Teflon-lined stainless steel pressure vessels and heated in an oven at 200 °C under autogenic pressure for 48 h. Finally, the H/SAPO-34 was obtained through centrifugal separation, washed, dried at 100 °C overnight and calcined at 650 °C in air for 6 h.
The Cu/SAPO-34 catalyst was prepared by the ion-exchange method over H/SAPO-34 molecular sieve as follows. Firstly, NH4/SAPO-34 was obtained by exchanging H/SAPO-34 in ammonium nitrate solution at 80 °C for 3 h. Secondly, NH4/SAPO-34 was added into the copper sulfate solution and then stirred at 70 °C for 4 h. The slurry was filtered, washed and dried at 90–100 °C for 16 h. The dried Cu/SAPO-34 was calcined at 500 °C for 5 h. Further, the aged Cu/SAPO-34 was obtained by the 750 °C, 10% H2O, 12 h hydrothermal treatment.
2.2 NH3 adsorption performance and acid sites characterization
NH3-TPD experiments were performed to evaluate the amount of acid sites under different temperatures. Prior to each of the following experiments, the catalysts were pretreated at 500 °C for 30 min in 5% O2/N2. Thereafter, they were cooled to the test temperatures (80, 150, 200, 250, 300, 350, 400 °C) in N2. At the different test temperatures, NH3 was introduced in 500 ppm NH3/N2 until the outlet concentration of NH3 was stable. The catalysts were purged with N2 to remove any weakly adsorbed NH3. Finally, the catalysts were heated from the test temperature to 550 °C at a ramp rate of 10 °C min−1.
Diffuse reflectance infrared Fourier transform spectra (DRIFTS) were obtained on a Nicolet 6700 FTIR equipped with a MCT detector at a resolution of 1 cm−1. Three scans were performed for each spectrum. The total flow rate was 150 mL min−1. Prior to every experiment, the samples were pretreated with 2% O2 in He at 500 °C for 30 min and the background spectra were collected under He at the test temperature.
For the NH3 adsorption experiments, the samples were exposed to 500 ppm NH3 at 80, 150, 200, 250, 300, 350, and 400 °C, respectively. For the adsorbed NH3-SCR experiments, NH3 (500 ppm) was firstly introduced into the catalyst until the spectra were stable. Then, the catalyst was exposed to (500 ppm) NO and (5%) O2 until equilibrium was attained. These experiments were performed at 150, 200, 250, 300 and 350 °C, respectively.
2.3 NOx removal performance
2.3.1 de-NOx efficiency. The SCR activity was tested in a quartz reactor (20 mm inner diameter), in which 0.1 g sample (60–80 mesh) was mixed with 0.9 g quartz (60–80 mesh) at atmospheric pressure. The sample was sealed in a reactor tube with quartz wool. A K type thermocouple was inserted into the center of the catalyst to control the temperature. A Fourier Transform Infrared (FTIR) spectrometer (MKS-2030) equipped with 5.11 m gas cell was used to measure the concentrations of NO, NO2, N2O and NH3. The flow rates in all the experiments were controlled at 500 mL min−1. After pretreatment, SCR tests were conducted with feed gas compositions of 500 ppm NO, 500 ppm NH3 and 5% O2. The test temperature range was from 100 °C to 550 °C with an interval of 50 °C. The NO conversion was calculated using the following equation:| |
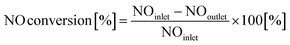 | (1) |
2.3.2 Lean NOx reaction performance with adsorbed ammonia. The sample particles size was 80–100 mesh and volume hourly space velocity was 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600
600![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1. The reaction temperatures were 150 °C, 200 °C, 250 °C, 300 °C, 350 °C and 400 °C. After the sample was pretreated, NH3 adsorption was performed with 500 ppm NH3 until its outlet concentration was stable. The catalyst purged with N2 after saturating by NH3. Finally, the titration reaction was performed by introducing it in 500 ppm NO and 5% O2.
000 h−1. The reaction temperatures were 150 °C, 200 °C, 250 °C, 300 °C, 350 °C and 400 °C. After the sample was pretreated, NH3 adsorption was performed with 500 ppm NH3 until its outlet concentration was stable. The catalyst purged with N2 after saturating by NH3. Finally, the titration reaction was performed by introducing it in 500 ppm NO and 5% O2.
2.4 Kinetic experiment
2.4.1 Reaction kinetic experiment. The NH3-SCR kinetics experiments were performed in a quartz reactor, mixing 25 mg catalyst with 125 mg quartz sand. A relatively small particles size (80–100 mesh) and volume hourly space velocity (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600
600![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1) was used in the kinetic tests. The inlet gas consisted of 500 ppm NO, 500 ppm NH3 and 5% O2, with N2 as the balance gas. The kinetics steady-state measurements were obtained in two temperature ranges: low temperature (160–205 °C) and high temperature (360–390 °C). The NH3-SCR reaction rates can be calculated from the NOx conversion by using the following equations:
000 h−1) was used in the kinetic tests. The inlet gas consisted of 500 ppm NO, 500 ppm NH3 and 5% O2, with N2 as the balance gas. The kinetics steady-state measurements were obtained in two temperature ranges: low temperature (160–205 °C) and high temperature (360–390 °C). The NH3-SCR reaction rates can be calculated from the NOx conversion by using the following equations:| |
 | (2) |
| | |
XNOx = NOx conversion [%]
| (3) |
| | |
FNOx = NOx flow rate [L(NOx) min−1]
| (4) |
2.4.2 NH3 adsorption kinetics experiment. The sample particles size and space velocity were the same as those in the NH3-SCR kinetics experiments. At each test temperature, 500 ppm NH3 was introduced into the reactor until the outlet concentration was steady and then purged with N2. At last, the sample was heated from the test temperature to 550 °C at a ramp rate of 10 °C min−1.
3. Results
3.1 NH3 adsorption performance and adsorption sites over Cu/SAPO-34 catalyst
3.1.1 NH3-TPD. NH3-TPD is used to determine the numbers and strengths of acid sites over catalysts. Fig. 1 is the deconvolution of NH3-TPD profiles as a function of temperature under different adsorption temperatures. As shown in Fig. 1(a), there are three acid sites existing on the H/SAPO-34 support, denoted as A, B and C. Low temperature area around 150 °C (peak A) is ascribed to NH3 desorption from weak Brønsted acid sites at the surface hydroxyl. Medium and high temperature ranges (peaks B and C) are assigned to the moderate and strong structural Brønsted acid sites. Compared with the TPD results of the Cu-SAPO-34 catalyst in Fig. 1(b), more than three acid sites residing in Cu-SAPO-34 catalyst have been discovered. Peak A around 150 °C is assigned to the weak Brønsted and Lewis acid sites related to Cu species. Peak B is ascribed to the residual structural Brønsted acid sites. Peaks C and D above 200 °C are considered as strong Brønsted acid sites and the new Lewis acid sites created by the Cu2+ species.
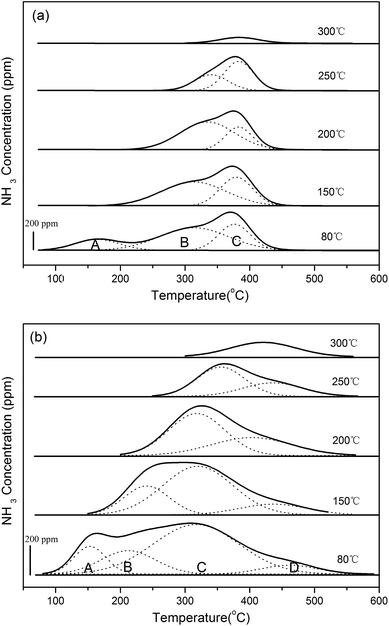 |
| | Fig. 1 NH3-TPD profiles of (a) H/SAPO-34 and (b) Cu/SAPO-34 catalysts under different temperatures. | |
The peak quantification results of NH3-TPD are shown in Table 1. It is seen that the number of NH3 adsorption sites decreases with an increase of temperature, while the acidity becomes stronger. Meanwhile, when compared with the acid sites of the H/SAPO-34 support, the number of weak and strong acid sites in the Cu/SAPO-34 sample increases, while the number of moderate acid sites decreases. This is concomitant with the assignment of acid sites according to the NH3-TPD profiles.
Table 1 Fitting results of NH3-TPD under different temperatures
| Temperature |
Sample |
Weak acid sites (mmol g−1) |
Moderate acid sites (mmol g−1) |
Strong acid sites (mmol g−1) |
| 80 °C |
H/SAPO-34 |
0.18 |
0.57 |
0.30 |
| Cu/SAPO-34 |
0.22 |
0.13 |
0.87 |
| 150 °C |
H/SAPO-34 |
|
0.62 |
0.28 |
| Cu/SAPO-34 |
|
0.21 |
0.70 |
| 200 °C |
H/SAPO-34 |
|
0.56 |
0.22 |
| Cu/SAPO-34 |
|
— |
0.63 |
| 250 °C |
H/SAPO-34 |
|
0.38 |
0.11 |
| Cu/SAPO-34 |
|
— |
0.36 |
| 300 °C |
H/SAPO-34 |
|
|
0.08 |
| Cu/SAPO-34 |
|
|
0.15 |
| 350 °C |
H/SAPO-34 |
|
|
0.009 |
| Cu/SAPO-34 |
|
|
0.014 |
3.1.2 DRIFTS of NH3 adsorption. The acid sites and their acidities can be further determined using ammonia adsorption DRIFTS.25 The spectra of the adsorbed ammonia on H/SAPO-34 and Cu/SAPO-34 catalyst under different temperatures are shown in Fig. 2. The negative bands at 3680, 3624, and 3590 cm−1 are assigned to the OH vibrations of P–OH, Si–OH, and Al–OH–Si, respectively.22 These bands at 3356, 3275, and 3180 cm−1 are due to the N–H stretching vibration of ammonia ions and coordinated ammonia species. In the N–H bending vibration region, 1628 and 1459 cm−1 are attributed to the adsorbed ammonia ion species.21 Further, the peak positions and assignments in the DRIFTS spectra are listed in Table 2. Compared with H/SAPO-34 results in Fig. 2(a), new bands at 885 and 858 cm−1 appear (Fig. 2(b)) over the Cu/SAPO-34 sample and they are related to the internal asymmetric framework vibrations perturbed by copper cations to form NH3–Cu2+.19,20,23,26 With respect to the similarity in the DRIFTS results of H/SAPO-34 support and Cu/SAPO-34 catalyst, the bands at 3680, 3624, and 3590 cm−1 related to the Brønsted acid sites are shown in Fig. 2(a) and (b). Furthermore, the intensities of these bands decrease with the temperature increasing, indicating that the ammonia is desorbed from the weak and moderate Brønsted sites. However, the strengths of 3624 and 3590 cm−1 bands at each temperature in Fig. 2(b) is lower than that in Fig. 2(a), which means that the number of moderate Brønsted acid sites decreases in the Cu/SAPO-34 catalyst. Moreover, the band at 3180 cm−1 distinctly decreases above 200 °C, which suggests that new weak acid sites are formed on the Cu/SAPO-34 sample. Additionally, the bands at 885 and 858 cm−1 assigned to the Lewis acid sites have been detected above 300 °C only on Cu/SAPO-34. This could result in an increase in the number of strong acid sites. Meanwhile, this is consistent with the NH3-TPD results that the number of weak and strong sites increases, but the number of moderate sites decreases. In addition, the IR bands at 3624 and 3590 cm−1 obviously decrease at 300 °C, while the intensity of the band at 885 cm−1 is still stable with the temperature rising above 300 °C. Therefore, the ammonia adsorbed on the Lewis acid sites is much stronger than that on the Brønsted acid sites, easily desorbing up to 300 °C.
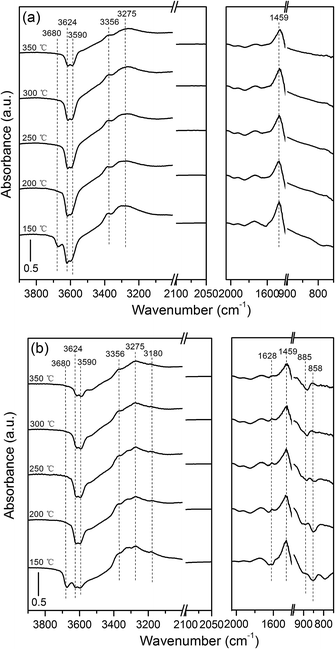 |
| | Fig. 2 DRIFTS spectra of NH3 chemisorbed on H/SAPO-34 (a) and Cu/SAPO-34 catalyst (b) at different temperatures. | |
Table 2 Peak positions and assignments in the DRIFTS spectra
| Wavenumber (cm−1) |
Species |
Reference |
| 3680, 3624, 3590 |
P–OH, Si–OH, and Al–OH–Si groups |
22 |
| 3356, 3275, 3180 |
N–H stretching vibration |
21 |
| 1628, 1459 |
N–H bending vibration |
21 |
| 885, 858 |
Internal asymmetric framework vibrations perturbed by copper cations |
19, 20, 23 and 26 |
3.2 NOx removal performance
3.2.1 Catalyst characterization. Fig. 3(a) shows the XRD patterns of H/SAPO-34 and Cu/SAPO-34. The diffraction peaks of the samples in Fig. 3(a) are consistent with typical SAPO-34 zeolite, indicating that the prepared SAPO-34 exhibits a CHA structure and the process of Cu loading does not destroy the crystalline structure. Further, the chemical composition of H/SAPO-34 support and Cu/SAPO-34 catalyst are listed in Table 3. Meanwhile, as shown in Fig. 3(b), it shows that the catalyst retains the CHA structure after the 750 °C, 10% H2O, 12 h hydrothermal treatment, which suggests its excellent hydrothermal stability. Furthermore, the activity test was performed to probe the de-NOx performance of the Cu/SAPO-34 catalyst before and after the hydrothermal treatment, as illustrated in Fig. 3(c). It displays that the aged Cu/SAPO-34 shows higher NO conversion than the fresh one, which could result from the increase of the isolated Cu2+ ions of Cu/SAPO-34 catalyst after the hydrothermal treatment. Therefore, with regard to either the structural integrity or SCR activity, the Cu/SAPO-34 catalyst presents excellence performance after hydrothermal treatment.
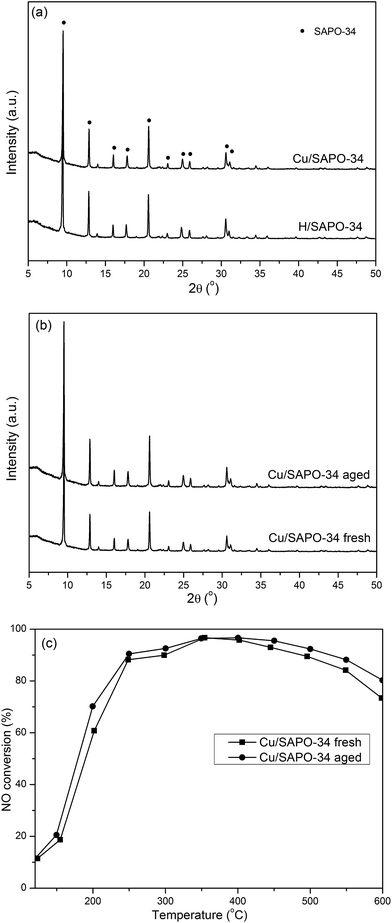 |
| | Fig. 3 (a) XRD patterns of H/SAPO-34 and Cu/SAPO-34, (b) XRD patterns of fresh Cu/SAPO-34 and aged Cu/SAPO-34, (c) de-NOx activity of fresh Cu/SAPO-34 and aged Cu/SAPO-34. | |
Table 3 Chemical composition of H/SAPO-34 support and Cu/SAPO-34 catalyst
| Sample |
Concentration (wt%) (ICP) |
| Al |
Si |
P |
Cu |
| H/SAPO-34 |
21.6 |
8.6 |
12.3 |
— |
| Cu/SAPO-34 |
21.4 |
8.6 |
12.2 |
1.7 |
3.2.2 de-NOx efficiency. In order to examine the efficiency of NOx elimination, the SCR activity was evaluated over the Cu/SAPO-34 catalyst from 120 °C to 600 °C. The NO and NH3 conversion results are shown in Fig. 4(a). The NO conversion continuously increases and the maximum NO conversion occurs at 350 °C. With the temperature increasing above 350 °C, NO conversion decreased due to the competitive NH3 oxidation at high temperatures. Approximately 100% NH3 conversion has been achieved above 350 °C. In addition, Fig. 4(b) shows that NO2 and N2O were marginally detected during the tests, suggesting high N2 selectivity. Consequently, the homemade Cu/SAPO-34 catalyst with representative SCR performance could be used to study the behavior of various reactants, which has definite applications in current passive-SCR research.
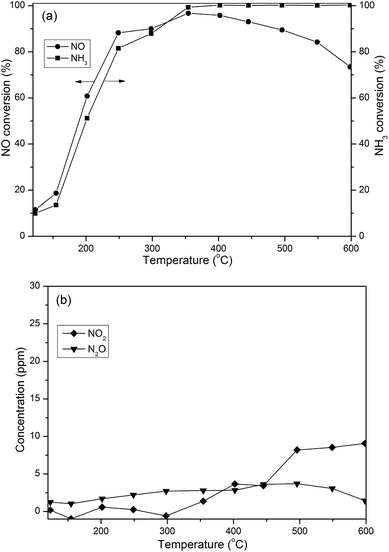 |
| | Fig. 4 SCR performance of Cu/SAPO-34 catalyst. Reaction conditions: 500 ppm NO, 500 ppm NH3, 10% O2, 7% CO2, 5% H2O, and N2 as balance gas. (a) NO conversion and NH3 conversion; (b) NO2 and N2O concentration. Flow rate: 500 mL min−1. GHSV: 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1. 000 h−1. | |
3.2.3 Lean NOx reaction performance with adsorbed ammonia. During the lean period in the passive-SCR process, NH3 saturated catalyst is actually titrated by generated NOx. In order to evaluate the activity of adsorbed NH3 species, they were titrated by NO + O2 under a series of temperatures in Fig. 5. During the first dozens of seconds, NO conversion rates rapidly decrease at all the temperatures. Further, with the proceeding of the reaction, NO reaction rates show distinctly different performances with the former. Moreover, the lower the reaction temperature, the longer is the time required for equilibrium. When the temperature is above 300 °C, it is shown that the adsorbed NH3 could be thoroughly consumed within 100 s. However, reaction equilibrium needs a long time at temperatures below 300 °C. Consequently, different activities of adsorbed NH3 along with time could imply that there are diverse mechanisms under the corresponding temperatures.
 |
| | Fig. 5 SCR rates as a function of time at different temperatures over Cu-SAPO-34 catalyst. | |
3.2.4 DRIFTS spectra of lean NOx reaction with adsorbed ammonia. The DRIFTS spectra of the adsorbed NH3 titration by NO + O2 at a series of temperatures (150, 200, 250, 300, 350 °C) are shown in Fig. 6. Particularly, the blank experiment was carried out to rule out the effect of residual NH3 in the system and verify their reliability. The results are shown in Fig. S2 and S3.† As reported previously,22 the doublet bands at 3624 cm−1 and 3590 cm−1 were assigned to the stretching model of the bridged Brønsted OH groups Al–(OH)–Si, which could represent the Brønsted acid sites. Meanwhile, the bands at 885 cm−1 and 858 cm−1 were related to the Lewis acid sites of Cu2+ ions. Accordingly, the peak intensity at 3624 cm−1 and 885 cm−1 in the DRIFTS spectra are chosen to study the behaviors of the adsorbed ammonia species on the Cu-SAPO-34 catalyst. The peak intensities, representing the ammonia contents, are plotted in Fig. 6(b), (d), (f), (h) and (j). As shown in Fig. 6(b) and (d), the peak intensities of the Brønsted and Lewis acid sites simultaneously decreases with the proceeding of the titration reaction at relatively low temperatures (150 °C and 200 °C). However, when the reaction temperature increases, the Brønsted acid sites intensity still decreases along with time. However, the Lewis acid sites intensity increases for the initial two minutes, followed by an abrupt decrease. Moreover, since the Cu2+ cations (Lewis acid sites) are SCR reaction active sites, the adsorbed NH3 could be directly consumed on the Cu2+ cations of Cu/SAPO-34 catalyst. This results in the decrease of the peak intensity of the Lewis acid sites. Subsequently, NH3 adsorbed on the Brønsted acid sites has to migrate from the Brønsted acid sites to the Lewis acid sites for participation in the SCR reaction. Consequently, an increase in the adsorbed ammonia on the Lewis sites can be attributed to NH3 migration from the Brønsted sites. In view of the above results, it is implied that the NH3 migrates between the two acid sites, potentially enabling NH3 to be transferred to the Lewis acid sites, resulting in a reaction. The different variation trends of the adsorbed ammonia contents on the Lewis sites at different temperature ranges is related to the speed of NH3 consumption and NH3 migration, which will be discussed later. In other words, the rate differences of NH3 migration and reaction induce the difference in performances of the adsorbed NH3 in the titration reaction.
 |
| | Fig. 6 NO + O2 titration experiment was performed at 150 °C (a), 200 °C (c), 250 °C (e), 300 °C (g), 350 °C (i). Left-hand sections: DRIFTS spectra of adsorbed NH3 titration by NO + O2 on Cu-SAPO-34 at different times (0 min, 1 min, 2 min, 3 min, 5 min and 7 min, 9 min, 11 min). Right-hand sections: the change of peak intensity of the representative Lewis and Brønsted acid sites during the NO + O2 titration (b), (d), (f), (h), and (j). | |
3.3 Kinetic results
3.3.1 Reaction kinetics results. In order to distinguish between the difficulty of NH3 migration and reaction over the Cu/SAPO-34 catalyst, it is possible to compare the reaction activity energy with the adsorption energy. Consequently, the SCR activity energy and NH3 adsorption energy on the Cu/SAPO-34 sample are calculated in the following section. Further, the NH3-SCR reaction rate equation could be expressed by the following equation:On the basis of our previous results, the SCR reaction order for NH3, NO, and O2 is approximately 0, 1, and 0.5, respectively.27 As a result, α = 0, β = 1, and θ = 0.5 are taken into the rate equation and simplified as follows:
| |
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) r = ln r = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k + ln k + ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CNO + 0.5 CNO + 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2 CO2
| (6) |
Because the reactant concentrations in the present research are CNO = 500 ppm and CO2 = 5%, the Arrhenius equation can be introduced into eqn (6). Further, the simplified eqn (6) was obtained through the above calculation. Consequently, the Arrhenius plots of the SCR reaction rates are attained from the simplified eqn (6), as shown in Fig. 7. Thereupon, the SCR reaction Ea can be calculated through the Arrhenius plots, from which the Ea values at low temperature and high temperature can be obtained as 52.78 kJ mol−1 and 7.26 kJ mol−1, respectively, for the Cu/SAPO-34 catalyst.28
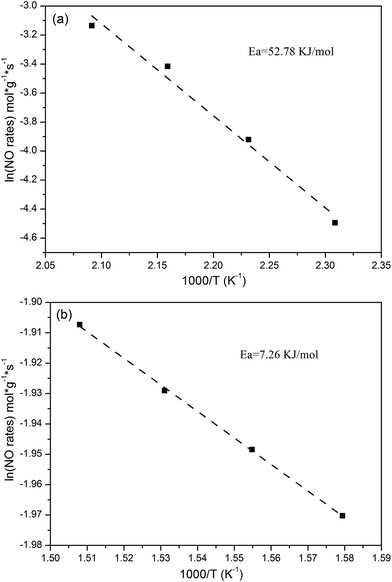 |
| | Fig. 7 Arrhenius plots of the SCR reaction rates per gram of Cu/SAPO-34 at 160–205 °C (a) and 360–390 °C (b). Conditions: 500 ppm NO, 500 ppm NH3, 5% O2, and N2 as balance gas. Flow rate: 1.5 l min−1. GHSV: 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1. 000 h−1. | |
3.3.2 NH3 adsorption kinetics results. The adsorption energy of NH3 can be obtained from the Langmuir isotherm model incorporating temperature dependency. The concentration of NH3 (QS) present in the fully filled micro-pores of a material can be calculated as below:where Mp (cm3 g−1) is the micropore volume of the material, and V* (cm3 mol−1) is the mole volume of NH3 at the adsorption temperature. The molar volume can be calculated from the virial equation of state, as described previously.29 The Langmuir model equation, considering temperature dependency of Ka, is expressed as follows:| | |
Q = (QSKaP)/(1 + KaP)
| (8) |
where Ka = Ka0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(q/RT), Q (mmol g−1) is the amount of adsorbate. Ka (1/Pa) is an equilibrium parameter. Ka0 (1/Pa) is an equilibrium constant. q (kJ mol−1) is the adsorption energy. R is the ideal gas constant. P (Pa) is the gas pressure. T (K) is the adsorption temperature.
exp(q/RT), Q (mmol g−1) is the amount of adsorbate. Ka (1/Pa) is an equilibrium parameter. Ka0 (1/Pa) is an equilibrium constant. q (kJ mol−1) is the adsorption energy. R is the ideal gas constant. P (Pa) is the gas pressure. T (K) is the adsorption temperature.Fig. 8 displays the NH3 breakthrough curves for the Cu-SAPO-34 catalyst at different temperatures (80, 150, 200, 250, 300, 330, 360 and 390 °C). The relative NH3 concentration, the ratio of outlet concentration (Cout) to inlet concentration (Cin), as a function of time is plotted in Fig. 8. A relative concentration value of 1 implies NH3 adsorption saturation on the catalyst. The breakthrough times are shortened with the increase of the adsorption temperature, indicating that the number of NH3 adsorption sites decreases.30 The amount of adsorbed NH3 is determined by the temperature program desorption from the test temperature to 600 °C: the results are listed in Table 4. Fig. 9 depicts the temperature dependence of the calculated adsorption equilibrium parameter (Ka), from which the adsorption energy of 12.22 and 87.54 kJ mol−1 are obtained.
 |
| | Fig. 8 NH3 breakthrough curves at different temperatures on Cu-SAPO-34 catalyst. | |
Table 4 Adsorption amounts of NH3 on Cu/SAPO-34 catalyst at saturation
| Catalyst |
Adsorbate |
Adsorption temperatures (°C) |
Amount of adsorbed NH3 (mmol g−1) |
| Cu/SAPO-34 |
NH3 |
80 |
1.316 |
| NH3 |
150 |
0.944 |
| NH3 |
200 |
0.587 |
| NH3 |
250 |
0.288 |
| NH3 |
300 |
0.101 |
| NH3 |
330 |
0.048 |
| NH3 |
360 |
0.013 |
| NH3 |
390 |
0.009 |
 |
| | Fig. 9 Adsorption equilibrium parameter of NH3 under Langmuir isotherm model on the Cu/SAPO-34 sample. | |
4. Discussion
4.1 NH3 adsorption performance and assignment of various adsorption sites on Cu/SAPO-34 catalyst
In order to understand the NH3 behavior during the passive-SCR process, firstly, the acid type and Cu influence on the acid sites of the catalyst should be clearly separated. Comparing the NH3-TPD results of H/SAPO-34 support and Cu/SAPO-34 catalyst in Fig. 1, it is evident that the number of weak and strong acid sites in the latter one increases, but the number of moderate acid sites decreases. This is also verified by the peak quantification results of NH3-TPD under each temperature. For a Cu/SAPO-34 sample, the increase in the number of weak and strong acid sites in Table 1 can be attributed to the Cu species incorporation into the H/SAPO-34 support. As illustrated in Fig. 2(b), the band at 3180 cm−1 is due to the N–H stretching vibration of the coordinated ammonia species (NH3–Cu+), which desorbs at a low temperature and is attributed to the weak Lewis acid sites. Meanwhile, the bands at 885 and 858 cm−1 are related to NH3–Cu2+, which are assigned to strong Lewis acid sites. Conversely, the number of moderate acid sites decreases on the Cu/SAPO-34 sample compared with the H/SAPO-34 support. This is induced by the replacement of Brønsted hydroxyls in the Si–OH–Al groups by Cu species. The bands at 3624 and 3590 cm−1 are attributed to the OH vibrations of the Al–OH–Si structure. Further, the strengths of these two negative peaks decrease in the Cu/SAPO-34 sample. This confirms the speculation about the decrease in the number of moderate acid sites. Furthermore, comparing the NH3-TPD profiles at different temperatures in Fig. 1(b), the quantity and species of the adsorbed NH3 on the Cu/SAPO-34 sample gradually decrease with increasing temperature. Specifically, the amount of adsorbed NH3 on the Brønsted acid sites quickly decreases; however, NH3 adsorbed on the Lewis acid sites seldom desorbs at high temperatures. NH3 adsorption DRIFTS shown in Fig. 2(b) illustrates that the bands at 3624 and 3590 cm−1 related to the Brønsted acid sites significantly decrease; nevertheless, the band at 885 cm−1 attributed to the Lewis acid sites scarcely changes above 300 °C. Consequently, strong Lewis acid sites related to Cu2+ species could play a more important role in the NH3 adsorption process at high temperatures. Furthermore, in the passive-SCR process, the generated NH3 in the rich period requires the maximum possible adsorption, especially at high temperatures. Accordingly, for fulfilling the different NH3 adsorption requirements of passive-SCR in the future, it is possible to formulate a specific catalyst formula by modulating the Cu content, Si content and templates. After the rich period of the passive-SCR process, in order to examine the performance of the Cu/SAPO-34 catalyst in the lean period, it is necessary to discuss the reaction behavior of the adsorbed NH3.
4.2 Activity of various adsorbed NH3 species over Cu/SAPO-34
By combining the NH3-TPD results, it is evident that only Brønsted sites exist on H/SAPO-34, while both Brønsted and Lewis sites exist on Cu/SAPO-34. It is known that H/SAPO-34 exhibits almost no SCR activity, and a Lewis site is an SCR active site. Therefore, a Lewis site is active in the SCR reaction and NH3 adsorbed on the Lewis sites is denoted as active NH3 species, while those adsorbed on a Brønsted site are labeled as inactive NH3 species. Further, inactive NH3 should transform into active NH3 by NH3 migration to the Lewis site, subsequently participating in the SCR reaction. Furthermore, active and inactive NH3 species present different reaction behaviors at low and high temperatures in the titration reaction. Consequently, it is important to discuss the problem of NH3 migration and SCR reaction rate of the different adsorbed NH3 species. As shown in Fig. 6(b) and (d), the amounts of active and inactive NH3 decline at low temperatures, thereby suggesting that the migration rate of inactive NH3 is slower than the reaction rate of active NH3. Moreover, the reaction equilibrium requiring a long time at low temperatures can be attributed to the above phenomenon, as shown in Fig. 5. However, the amount of active NH3 shows a short increase before decreasing with the inactive NH3 at high temperatures, as shown in Fig. 6(f), (h) and (j). Further, this illustrates that the migration rate of inactive NH3 is faster than the reaction rate of active NH3. Therefore, adsorbed NH3 is rapidly consumed at high temperatures, as shown in Fig. 5. Furthermore, it is necessary to combine the NH3 adsorption strength on Cu/SAPO-34 and systematically discuss its application potential in lean and rich operation of the passive-SCR technology. Meanwhile, a possible mechanism of passive-SCR could be proposed.
4.3 Reaction mechanism of passive-SCR process
Considering the efficiency of the passive-SCR process, the discussion about controlling factors and rate-determining steps during different periods is essential for Cu/SAPO-34 application and modification in the future. It is known that the front TWC catalyst and the back SCR catalyst satisfy the exhaust elimination in different periods. In this study, we focus on the role of the back SCR catalyst and the potential of the Cu/SAPO-34 sample. During the rich period, NH3 is generated over the front TWC catalyst and stored on the back SCR catalyst. Further, the acid content and acidity of the Cu/SAPO-34 sample are the main factors related to the adsorption of NH3, which ensures the prevention of the secondary pollution by NH3 slip. During the lean period, the generated NO from the front TWC could be consumed by the NH3 saturated Cu/SAPO-34 sample. Consequently, the activity of the adsorbed NH3 species and the NH3 consumption rate-determining step are the key points to investigate.
For the Cu/SAPO-34 catalyst, the NH3 adsorption only happens during the rich period. The NH3-TPD results shown in Fig. 1 and Table 1 show its excellent NH3 adsorption capability. Since the NH3 adsorption sites decrease with increasing temperature, our previous study found that Cu/SAPO-34 acidity could be adjusted to modify the Si contents, Cu contents and templates for the detailed engine requirement. In addition, the NH3 consumption mechanism over the Cu/SAPO-34 sample during the lean period is shown in Scheme 1. Fig. 5 and 6 show that the inactive NH3 species on the Brønsted sites have to become active NH3 species on the Lewis sites in order to participate in the NO consumption reaction, since the Cu2+ species are the only active sites for SCR over the Cu/SAPO-34 sample. Due to the variation of the NH3 adsorption energy and apparent activation energy for different temperature ranges, the rate-determining step changes at the lean period. At low temperatures, Fig. 6 shows that the NH3 migration is slower than NH3 consumption on active sites and NH3 could be easily adsorbed on the acid sites. Compared with the NO consumption rate, the passive-SCR reaction is controlled by the NH3 migration, as shown in Scheme 1a. In addition, the NH3 adsorption energy shown in Fig. 9 could be approximately ascribed to the NH3 adsorption on the Brønsted acid sites (12.22 kJ mol−1) and Lewis acid sites (87.54 kJ mol−1), since the latter show the strongest acidity (Fig. 1) at high temperatures and NH3 mainly adsorbs on the Brønsted sites for its overwhelming contents at low temperature. Therefore, their differences (75.32 kJ mol−1) is the apparent energy of migration from the Brønsted acid sites to Lewis acid sites, which is higher than NO consumption apparent Ea value in Fig. 7(a). This also proves that NH3 migration is the rate-determining step at low temperatures. The increase in the number of Lewis acid sites could improve the passive-SCR efficiency at low temperatures. In the high temperature range, Fig. 6 reveals that NH3 migration occurs at a higher speed than NO consumption on Cu2+ sites and the apparent Ea value in Fig. 7(b) also shows the fast rate of NO consumption at high temperatures. As shown in Scheme 1(b), the efficient adsorption of NH3 could ensure the NO consumption over the Cu/SAPO-34 sample, while the decreasing NH3 adsorption sites are the main factor to the passive-SCR process at high temperatures. Consequently, NH3 adsorption may be the rate-determining step of the passive-SCR rich period under high temperatures.
 |
| | Scheme 1 Mechanism of passive-SCR process over Cu/SAPO-34 catalyst in the low temperature range (a) and high temperature range (b). | |
Furthermore, compared with standard NH3-SCR mechanism,22,31 the distinction of the passive-SCR process is its different rate-determining step. In view of the NH3-SCR process, the orders of the NH3 and NO reactants over Cu/SAPO-34 were 0 and 0.75, respectively. Further, this suggests that NH3 species adsorption is easy and strong, but this is difficult for NO species.27 Therefore, it is proposed that the NH3-SCR reaction mechanism tends to be the E–R mechanism. Further, our previous works proved that isolated Cu2+ are the active sites for NH3-SCR reaction under 200 °C.32 Furthermore, it is reported that ammonia nitrites are the intermediates and decompose to nitrogen and water.22,23 Moreover, the rate-determining step of the NH3-SCR mechanism is still controversial up to now. Actually, both NH3-SCR and passive-SCR process are similar in terms of the essence of the NH3-SCR reaction. The passive-SCR process is deemed as stepwise NH3-SCR, in which the effect of NH3 adsorption is ruled out. This is because after the catalyst is saturated by NH3 in the rich period, the adsorbed NH3 reacts through the static consumption process during the lean period without NH3 supplement. Moreover, it could present the same adsorption species and intermediates as standard NH3-SCR, while it shows different rate-determining steps. The NH3 migration plays a vital role in the passive-SCR process, which could be the rate-determining step of low temperature in the lean period. In short, the significance of NH3 migration in the NH3-SCR reaction could be distinctly reflected in the passive-SCR research.
5. Conclusion
The acid sites assignment and activity of various adsorbed NH3 species over the Cu/SAPO-34 catalyst have been systematically studied. The NH3-TPD and DRIFTS results illustrate that SAPO-34 support contains different Brønsted acid sites and Cu incorporation creates Lewis acid sites in the Cu/SAPO-34 sample. The number of weak and strong acid sites over the Cu/SAPO-34 sample increases, which results from the Cu incorporation forming Lewis sites. However, due to the replacement of Brønsted hydroxyls in the Si–OH–Al groups by Cu2+ species, the number of moderate acid sites decreases. Furthermore, a possible reaction mechanism and rate-determining step for the passive-SCR process are proposed. First, the active NH3 adsorbed on the Lewis sites presents high activity, but the inactive NH3 species have to migrate and transform into active NH3 for NO consumption. During the rich period of the passive-SCR, only NH3 adsorption takes place and the increase in the number of acid sites can satisfy different technical requirements. During the lean period, the inactive NH3 migration rate from the Brønsted acid sites to Lewis acid sites is slower than the NO consumption rate by active NH3, which confines the SCR reaction rate at low temperatures. Therefore, NH3 migration could be the rate-determining step of the passive-SCR lean period at low temperatures. Nevertheless, at high temperatures, NH3 migration exhibits higher speed than the NO consumption rate and the adsorbed NH3 species reveal enough activity for NO consumption. However, the decreasing number of NH3 adsorption sites exerts the primary impact on the passive-SCR process at high temperatures. Accordingly, the rate-determining step of the passive-SCR rich period in the high temperature range is the adsorption of NH3.
Acknowledgements
The authors are grateful to the financial support from the GM Global Research and Development (RD-07-312-NV487). This work was supported by the program of the National High Technology Research and Development Program of China (863 Program, 2011AA03A405).
Notes and references
- W. Li, K. Perry, K. Narayanaswamy, C. Kim and P. Najt, SAE Int., 2010, 01, 0366 Search PubMed
 .
. - O. Guralp, G. Qi, W. Li and P. Najt, SAE Int., 2011, 01, 0307 Search PubMed
 .
. - C. Kim, K. Perry, M. Viola, W. Li and K. Narayanaswamy, SAE Int., 2011, 01, 0306 Search PubMed
 .
. - J. Wang, Y. Ji, G. Jacobs, S. Jones, D. Kim and M. Crocker, Appl. Catal., B, 2014, 148–149, 51–61 CrossRef CAS PubMed
 .
. - B. Shakya, M. Harold and V. Balakotaiah, Chem. Eng. J., 2014, 237, 109–122 CrossRef CAS PubMed
 .
. - Y. Liu, Y. Zheng, M. Harold and D. Luss, Top. Catal., 2013, 56, 104–108 CrossRef CAS
 .
. - F. Rohr, D. Peter, E. Lox, E. Kögel, A. Sassi, L. Juste, C. Rigaudeau, G. Belot, P. Gélin and M. Primet, Appl. Catal., B, 2005, 56, 201–212 CrossRef CAS PubMed
 .
. - S. Matsumoto, Y. Ikeda, H. Suzuki, M. Ogai and N. Miyoshi, Appl. Catal., B, 2000, 25, 115–124 CrossRef CAS
 .
. - I. Bull, W. Xue, P. Burk, S. Boorse, W. Jaglowski, G. Koermer, A. Moini, J. Patchett, J. Dettling and M. Caudle, US Pat., US 7,601,662 B2, 2009
 .
. - T. Ishihara, M. kagawa, F. Hadama and Y. Takita, J. Catal., 1997, 169, 93–102 CrossRef CAS
 .
. - D. Fickel, E. D'Addio, J. Lauterbach and R. Lobo, Appl. Catal., B, 2011, 102, 441–448 CrossRef CAS PubMed
 .
. - Q. Ye, L. Wang and R. Yang, Appl. Catal., A, 2012, 427–428, 24–34 CrossRef CAS PubMed
 .
. - R. Martínez-Franco, M. Moliner, C. Franch, A. Kustov and A. Corma, Appl. Catal., B, 2012, 127, 273–280 CrossRef PubMed
 .
. - S. Gómez, A. Campero, A. Hernández and G. Fuentes, Appl. Catal., A, 2000, 197, 157–164 CrossRef
 .
. - J. Kryca, M. Iwaniszyn, M. Piatek, P. Jodlowski, J. Thomas, A. Kolodziej and J. Lojewska, Catal. Today, 2013, 216, 135–141 CrossRef PubMed
 .
. - J. Wang, Z. Peng, H. Qiao, L. Han, W. Bao, L. Chang, G. Feng and W. Liu, RSC Adv., 2014, 4, 42403 RSC
 .
. - J. Wang, T. Yu, X. Wang, G. Qi, J. Xue, M. Shen and W. Li, Appl. Catal., B, 2012, 127, 137–147 CrossRef CAS PubMed
 .
. - X. Hou, S. Schmieg, W. Li and W. Epling, Catal. Today, 2012, 197, 9–17 CrossRef CAS PubMed
 .
. - P. Fanning and M. Vannice, J. Catal., 2002, 207, 166–182 CrossRef CAS
 .
. - L. Wang, G. Qi and D. Weng, J. Catal., 2012, 289, 21–29 CrossRef CAS PubMed
 .
. - L. Ma, Y. Cheng, G. Cavataio, R. McCabe, L. Fu and J. Li, Appl. Catal., B, 2014, 156–157, 428–437 CrossRef CAS PubMed
 .
. - D. Wang, L. Zhang, K. Kamasamudram and W. Epling, ACS Catal., 2013, 3, 871–881 CrossRef CAS
 .
. - L. Shi, T. Yu, X. Wang, J. Wang and M. Shen, Acta Phys.–Chim. Sin., 2013, 29(7), 1550–1557 CAS
 .
. - T. Yu, J. Wang, M. Shen and W. Li, Catal. Sci. Technol., 2013, 3, 3234–3241 CAS
 .
. - L. Ma, J. Li, R. Ke and L. Fu, J. Phys. Chem. C, 2011, 115, 7603–7612 CAS
 .
. - G. Lei, B. Adelman, J. Sárkány and W. Sachtler, Appl. Catal., B, 1995, 5, 245–256 CrossRef CAS
 .
. - T. Yu, T. Hao, D. Fan, J. Wang, M. Shen and W. Li, J. Phys. Chem. C, 2014, 118(13), 6565–6575 CAS
 .
. - I. Centeno, S. Castillo, R. Camposeco and G. Fuentes, Catal. Commun., 2013, 31, 11–15 CrossRef PubMed
 .
. - D. Kim, J. Wang and M. Crocker, Catal. Today, 2014, 231, 83–89 CrossRef CAS PubMed
 .
. - G. Wang, B. Dou, J. Wang, W. Wang and Z. Hao, RSC Adv., 2013, 3, 20523 RSC
 .
. - C. Paolucci, A. A. Verma, S. A. Bates, V. F. Kispersky, J. T. Miller, R. Gounder, W. N. Delgass, F. H. Ribeiro and W. F. Schneider, Angew. Chem., Int. Ed., 2014, 53, 1–7 CrossRef PubMed
 .
. - J. Xue, X. Wang, G. Qi, J. Wang, M. Shen and W. Li, J. Catal., 2013, 297, 56–64 CrossRef CAS PubMed
 .
.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra13984d |
|
| This journal is © The Royal Society of Chemistry 2015 |
Click here to see how this site uses Cookies. View our privacy policy here. ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1P2O5
0.1P2O5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06SiO2
0.06SiO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.4H2O. The sources of Si, P and Al were silica sol, 85% phosphoric acid and pseudoboehmite, respectively. Firstly, the pseudoboehmite and phosphoric acid were mixed with water. The mixture was fiercely stirred for 1 h. Secondly, the MA and silica sol were mixed well and added dropwise into the former mixture. Thirdly, the blend was sealed in 200 mL Teflon-lined stainless steel pressure vessels and heated in an oven at 200 °C under autogenic pressure for 48 h. Finally, the H/SAPO-34 was obtained through centrifugal separation, washed, dried at 100 °C overnight and calcined at 650 °C in air for 6 h.
6.4H2O. The sources of Si, P and Al were silica sol, 85% phosphoric acid and pseudoboehmite, respectively. Firstly, the pseudoboehmite and phosphoric acid were mixed with water. The mixture was fiercely stirred for 1 h. Secondly, the MA and silica sol were mixed well and added dropwise into the former mixture. Thirdly, the blend was sealed in 200 mL Teflon-lined stainless steel pressure vessels and heated in an oven at 200 °C under autogenic pressure for 48 h. Finally, the H/SAPO-34 was obtained through centrifugal separation, washed, dried at 100 °C overnight and calcined at 650 °C in air for 6 h.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600
600![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1. The reaction temperatures were 150 °C, 200 °C, 250 °C, 300 °C, 350 °C and 400 °C. After the sample was pretreated, NH3 adsorption was performed with 500 ppm NH3 until its outlet concentration was stable. The catalyst purged with N2 after saturating by NH3. Finally, the titration reaction was performed by introducing it in 500 ppm NO and 5% O2.
000 h−1. The reaction temperatures were 150 °C, 200 °C, 250 °C, 300 °C, 350 °C and 400 °C. After the sample was pretreated, NH3 adsorption was performed with 500 ppm NH3 until its outlet concentration was stable. The catalyst purged with N2 after saturating by NH3. Finally, the titration reaction was performed by introducing it in 500 ppm NO and 5% O2.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600
600![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1) was used in the kinetic tests. The inlet gas consisted of 500 ppm NO, 500 ppm NH3 and 5% O2, with N2 as the balance gas. The kinetics steady-state measurements were obtained in two temperature ranges: low temperature (160–205 °C) and high temperature (360–390 °C). The NH3-SCR reaction rates can be calculated from the NOx conversion by using the following equations:
000 h−1) was used in the kinetic tests. The inlet gas consisted of 500 ppm NO, 500 ppm NH3 and 5% O2, with N2 as the balance gas. The kinetics steady-state measurements were obtained in two temperature ranges: low temperature (160–205 °C) and high temperature (360–390 °C). The NH3-SCR reaction rates can be calculated from the NOx conversion by using the following equations:


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) r = ln
r = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k + ln
k + ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CNO + 0.5
CNO + 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2
CO2
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(q/RT), Q (mmol g−1) is the amount of adsorbate. Ka (1/Pa) is an equilibrium parameter. Ka0 (1/Pa) is an equilibrium constant. q (kJ mol−1) is the adsorption energy. R is the ideal gas constant. P (Pa) is the gas pressure. T (K) is the adsorption temperature.
exp(q/RT), Q (mmol g−1) is the amount of adsorbate. Ka (1/Pa) is an equilibrium parameter. Ka0 (1/Pa) is an equilibrium constant. q (kJ mol−1) is the adsorption energy. R is the ideal gas constant. P (Pa) is the gas pressure. T (K) is the adsorption temperature.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.







