Visible-light responsive electrospun nanofibers based on polyacrylonitrile-dispersed graphitic carbon nitride†
Tiefeng Xu,
Fei Wu,
Yan Gu,
Yi Chen,
Jinting Cai,
Wangyang Lu*,
Hongguang Hu,
Zhexin Zhu and
Wenxing Chen*
National Engineering Lab for Textile Fiber Materials & Processing Technology (Zhejiang), Zhejiang Sci-Tech University, Hangzhou 310018, China. E-mail: luwy@zstu.edu.cn; wxchen@zstu.edu.cn
First published on 30th September 2015
Abstract
A visible-light responsive photocatalyst, polyacrylonitrile-dispersed graphitic carbon nitride nanofibers (g-C3N4/PAN nanofibers), was synthesized by electrospinning. The g-C3N4 is dispersed uniformly in the nanofibers, which helps it overcome the defects of easy aggregation and difficult recycling of powder catalysts. The model substrate, rhodamine B (RhB), could be adsorbed rapidly into the PAN nanofibers and decomposed efficiently in situ simultaneously in the presence of the g-C3N4 over a wide pH range under visible light irradiation. As a fibrous catalyst, the g-C3N4/PAN nanofibers were quite simple to recycle, and the catalytic activity maintained a high level without obvious decline after being reused several times. In addition, based on the intermediates detected by ultra performance liquid chromatography-mass spectrometry and gas chromatography-mass spectrometry, N-de-ethylation chromophore cleavage and ring-opening mineralization are the main processes in RhB degradation. Finally, a possible mechanism was proposed, in which the hole along with the superoxide radical mainly contribute to the oxidative degradation of RhB.
1 Introduction
The increasingly serious energy crisis and environmental pollution are currently the two most important issues.1 Photocatalysis is a promising technology to degrade pollutants completely and efficiently by utilizing solar energy.2,3 During the past few decades, various photocatalysts have been developed. TiO2 is the most commonly used catalyst in the treatment of wastewater because of its non-toxicity, reasonable cost, high availability, photochemical stability and relatively high photocatalytic activity.4 However it can only absorb 3–5% of sunlight in the UV region, which limits its application greatly.5 Therefore it’s still a challenge to find a visible-light responsive photocatalyst to improve sunlight utilization.With the continuous research in semiconductor photocatalytic materials, the excellent performance of graphitic carbon nitride (g-C3N4) in many aspects has been discovered, such as thermal stability, chemical stability,6,7 and optical and photoelectrochemical properties.8–12 As an inorganic semiconductor material, compared to TiO2, g-C3N4 has a more suitable band gap (the former is 3.2 eV, the latter is 2.7 eV), which makes it absorb visible light better.5 Because of the outstanding performance described above, g-C3N4 has received much attention.13,14 Great progress in g-C3N4 has been made in the research in photochemical water splitting,15,16 bio-imaging,17–19 catalytic oxidation,20,21 catalytic hydrogenation,22,23 catalytic NO decomposition,24 photocatalytic degradation1,25,26 and as a basic catalyst.27 The photocatalytic degradation of pollutants into water, carbon dioxide and some other environmentally harmless small molecule compounds is an effective energy-saving and environmentally friendly method. Some common organic pollutants have been degraded by using modified g-C3N4 with enhanced photocatalyst properties.28 As a metal-free photocatalyst, g-C3N4 has also been used to activate H2O2 to produce hydroxyl radical (˙OH) to degrade organic pollutants under visible light irradiation.29 However, there are still some drawbacks of g-C3N4 which limit its future application: (a) poor dispersion in water and easy aggregation, which lead to a high photogenerated electron–hole recombination rate, and (b) difficulty in recycling, as a powder material.5 Researchers have made many attempts to improve their performance. For example, synthesising heterojunction composite photocatalysts is a good process to enhance the separation efficiency of photoexcited electron–hole pairs,30–32 and doping with some other elements or magnetic oxides could improve its performance and make it easy to recycle.5,33–35 Electrospinning has been recognized as an efficient technique for the preparation of polymer nanofibers. PAN-based electrospun nanofibers are hydrophobic with a too-low density, which makes them float easily on liquid. This point would maximize the photodegradation ability and optimize the irradiation efficiency of light by avoiding the hindrance of light source penetration.36,37
In this work, we report a novel visible-light responsive photocatalyst, polyacrylonitrile-dispersed graphitic carbon nitride nanofibers (g-C3N4/PAN nanofibers), which was prepared by electrospinning. This simple and effective method could overcome the aggregation of g-C3N4 in water, and recycle g-C3N4 from the reaction system. In addition, rhodamine B (RhB) was selected as a probe compound to evaluate the catalytic activity of g-C3N4/PAN nanofibers. The PAN nanofibers can adsorb RhB quickly and degrade it into small molecules in the presence of g-C3N4 in a wide pH range under visible light irradiation. By observing the effect of the active species capture agents on the degradation efficiency, and detecting the degradation products by ultra-performance liquid chromatography/high-definition mass spectrometry (UPLC Synapt G2-S HDMS) and gas chromatography/mass spectrometry (GC-MS), the possible mechanism and degradation process of RhB were proposed in the presence of g-C3N4/PAN nanofibers under visible light irradiation.
2 Experimental
2.1 Synthesis and characterization of photocatalysts
The g-C3N4 was synthesized by a facile and efficient method as reported previously with a few modifications.38 The g-C3N4/PAN nanofibers were prepared by ultrasound and electrostatic spinning. Firstly, the g-C3N4 powder was dispersed in DMF, and then PAN powder was added into the dispersion liquid. By ultrasonic treatment, the g-C3N4 powder was exfoliated into nanosheets and uniformly dispersed in the PAN solution to obtain the spinning solutions. Finally the electrospinning apparatus was employed to collect the g-C3N4/PAN nanofiber film. The detailed procedures and different samples with g-C3N4 (Table S1) are given in ESI.†2.2 Equipment
The morphology of the samples was observed on a transmission electron microscope (JEOL, JEM-2010) and a field emission scanning electron microscope (FESEM, ZEISS, ULTRA-55). The nitrogen adsorption and desorption isotherms were measured on a 3H-2000PS1 instrument in static measurement mode. The specific area was calculated using Brunauer–Emmett–Teller (BET) mode. The crystal structure of the samples was analyzed by X-ray diffraction spectroscopy (DX-2700) with Cu-Kα radiation at 2θ angles ranging from 5 to 70°. Fourier transform infrared spectroscopy (FTIR) spectra were obtained on a Thermo Nicolet 5700 spectrometer with KBr as the reference sample. A UV-vis spectrophotometer (UV-2550, Hitachi) was used to investigate the optical absorption properties of the samples. Thermogravimetric analysis (TGA) was evaluated on a 1100LF (Mettler-Toledo) from 25 to 800 °C at a heating rate of 10 K min−1 under air flow. UPLC Synapt G2-S HDMS (Waters) and GC-MS (7890B/5977A, Agilent) were employed to identify the intermediates. Electron paramagnetic resonance (EPR) signals of radicals trapped by 5,5-dimethylpyrroline-oxide (DMPO) were recorded at ambient temperature on a Bruker A300 spectrometer with settings as follows: center field, 3507 G; sweep width, 100 G; microwave frequency, 9.77 GHz; modulation frequency, 100 kHz; power, 20 mW.2.3 Photocatalytic experiments
The RhB dyes degradation experiments were conducted in the quartz glass reactors to test the photocatalytic activity of the as-prepared samples under visible light irradiation. g-C3N4/PAN nanofibers mats (1.5 mm × 1.5 mm, 1 g L−1) were dispersed in RhB aqueous solution (30 mL, 2 × 10−5 mol L−1). A 100 W lamp (LOT-oriel GmbH & Co. KG) was served as the visible light source, and a glass filter was used to block UV light (λ < 400 nm). During the photoreaction, the solution was continually taken from the reactor and filtered at given time intervals, and the concentration of RhB was determined by UV-vis spectrophotometer at 554 nm.3 Results and discussion
3.1 Characterization
Fig. 1 shows TEM images of bulk g-C3N4, the dispersion of g-C3N4 in PAN polymer solution and g-C3N4/PAN nanofibers, respectively. With its large size, the bulk g-C3N4 shows more serious aggregation (Fig. 1a). The g-C3N4 could be dispersed in PAN/DMF solution after ultrasound, and became very thin sheets (Fig. 1b). In Fig. 1c, the nanosheets of g-C3N4 have been embedded in the PAN nanofibers by electrostatic spinning. Moreover, the size of the g-C3N4 further reduced after the stripping and drawing process. In addition, it was found that the catalyst aggregated easily with increasing the amount of g-C3N4 (Fig. S1†) and the g-C3N4 was embedded in the 10% g-C3N4/PAN nanofibers uniformly (Fig. S1c†). | ||
| Fig. 1 TEM images of bulk g-C3N4 (a), the dispersion of g-C3N4 in PAN polymer solution (b), and 10% g-C3N4/PAN nanofibers (c). | ||
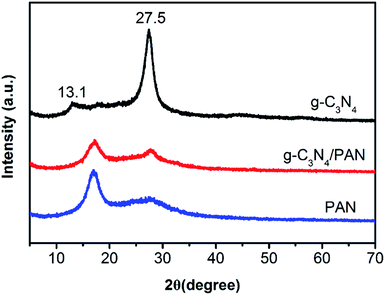
The X-ray diffraction (XRD) patterns for the as-prepared bulk g-C3N4, pure PAN nanofibers and g-C3N4/PAN nanofibers are shown in Fig. 2. The results indicated that the two peaks for g-C3N4 are in good agreement with the previous report.9,39 The strong XRD peak at 27.5° is indicative of interlayer stacking of the conjugated aromatic segment with distance of 0.324 nm, and the peak at 13.1° corresponds to in-plane ordering of tri-s-triazine units with an in-planar repeat period of 0.682 nm.22 For the pure PAN nanofibers, a strong diffraction peak centered at 17.1° and a weak diffusion diffraction peak centered at 27.9° were found, almost the same as in the previous report.40 The sharp diffraction peaks at 27.7° and 17.1° were observed in the g-C3N4/PAN nanofibers, however the intensities of the diffraction peaks were decreased due to the coupling with PAN. The surface area of the as-prepared samples was 12.07 m2 g−1 (Fig. S2†).
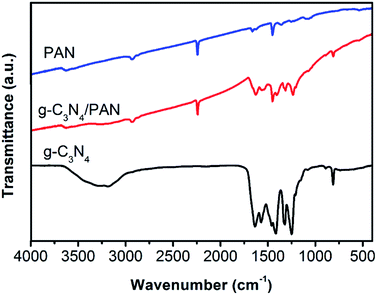
The FTIR spectra of the bulk g-C3N4, pure PAN nanofibers and g-C3N4/PAN nanofibers are shown in Fig. 3. For the pure PAN nanofibers, the peaks at 2928 and 2240 cm−1 are ascribed to stretch vibration of –CH2 and C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N, and the characteristic peak at 1448 cm−1 corresponds to the bend vibration of –CH2. As can be seen in the FTIR spectra, the original g-C3N4 exhibits characteristic absorption peaks similar to those in the previous literature.15 The intense band at 810 cm−1 is ascribed to the breathing mode of triazine rings.41 The bands at 1323 and 1250 cm−1 are the stretching vibration of C–N–C. The peak at 1635 cm−1 can be ascribed to the C–N stretching vibration mode, while the four strong peaks at 1416, 1460, and 1570 cm−1 correspond to the CN heterocycle stretching of g-C3N4. Additionally, the 3000–3600 cm−1 broad band can be attributed to the stretching vibration of terminal –NH groups at the defect sites of the aromatic ring. For the g-C3N4/PAN nanofibers, the characteristic peaks of g-C3N4 could be seen clearly.
N, and the characteristic peak at 1448 cm−1 corresponds to the bend vibration of –CH2. As can be seen in the FTIR spectra, the original g-C3N4 exhibits characteristic absorption peaks similar to those in the previous literature.15 The intense band at 810 cm−1 is ascribed to the breathing mode of triazine rings.41 The bands at 1323 and 1250 cm−1 are the stretching vibration of C–N–C. The peak at 1635 cm−1 can be ascribed to the C–N stretching vibration mode, while the four strong peaks at 1416, 1460, and 1570 cm−1 correspond to the CN heterocycle stretching of g-C3N4. Additionally, the 3000–3600 cm−1 broad band can be attributed to the stretching vibration of terminal –NH groups at the defect sites of the aromatic ring. For the g-C3N4/PAN nanofibers, the characteristic peaks of g-C3N4 could be seen clearly.
The optical absorption properties of the as-prepared samples were studied using UV-vis diffuse reflectance spectra. As can be seen from Fig. 4, the UV-vis diffuse reflectance spectrum of g-C3N4 shows typical semiconductor optical characteristics. The absorption edge of the g-C3N4 is around 450 nm, which signifies that the g-C3N4 can absorb visible light with a wavelength shorter than 450 nm. In comparison with the pure g-C3N4, the absorption edge of the g-C3N4/PAN nanofibers has no significant change.
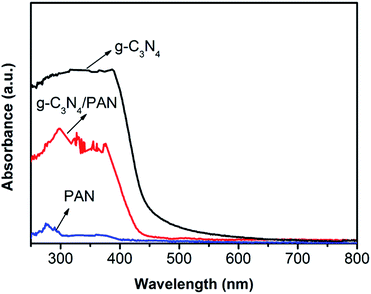 | ||
| Fig. 4 UV-vis diffuse reflectance absorption spectra of PAN nanofibers, bulk g-C3N4, and 10% g-C3N4/PAN nanofibers. | ||
Fig. 5 shows the TGA curves of the bulk g-C3N4, pure PAN nanofibers and g-C3N4/PAN nanofibers. It can be seen that there was no weight loss for the g-C3N4 until 500 °C and no residue of the sample left when heated to 750 °C, indicating that the as-prepared g-C3N4 shows high thermal stability. The pure PAN nanofibers have one significant weight loss at 300 °C, which is attributed to the release of small molecular gases such as NH3 and HCN. The PAN nanofibers started to degrade quickly above 300 °C, generating many gaseous compounds (e.g. HCN, CO and CO2) from the cyclization reaction. Due to the low quantity of g-C3N4 in the nanofibers, the thermal stability of the g-C3N4/PAN nanofibers presents a slight increase compared with the pure PAN nanofibers.
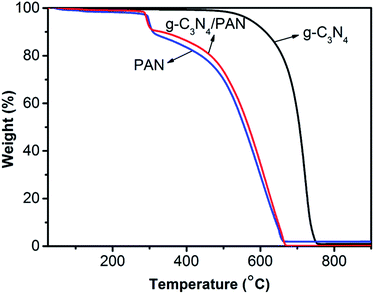 | ||
| Fig. 5 Thermogravimetric analysis for bulk g-C3N4, PAN nanofibers, and 10% g-C3N4/PAN nanofibers in air. | ||
3.2 Photocatalytic performance
The photocatalytic performance of the g-C3N4/PAN nanofibers was evaluated by the degradation of RhB in aqueous solution under visible light irradiation (λ > 400 nm) at room temperature, and the adsorption of RhB by the photocatalytic nanofibers in the dark served as a control experiment (Fig. 6). As can be seen, in the presence of g-C3N4/PAN nanofibers, the RhB in the solution was completely decomposed after 180 min under visible light irradiation. On the contrary, the degradation of RhB was negligible under visible light irradiation without g-C3N4/PAN nanofibers, indicating the high stability of RhB under visible light irradiation. To investigate the adsorption effect of the nanofibers on RhB, the RhB solution was stirred for 180 min in the dark to achieve adsorption/desorption equilibration. When it achieved adsorption/desorption equilibration, almost 41% of RhB was adsorbed on the photocatalytic nanofibers, suggesting that the g-C3N4/PAN nanofibers exhibited a high RhB adsorption capacity. The absorption maximum of the degraded solution exhibited a marked hypsochromic shift before complete decolorization (Fig. S3†). The above results indicated that the presence of the catalyst and light is essential for the efficient degradation of RhB under visible light irradiation. Futhermore, the g-C3N4/PAN nanofibers performed photocatalytic activity in the degradation of 4-CP (Fig. S4†), which indicated that the degradation of RhB was not caused by the sensitization effect.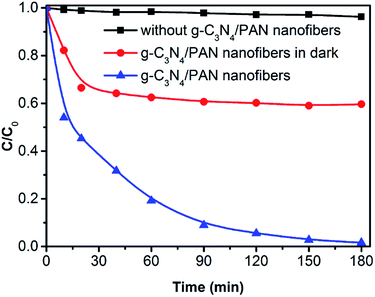 | ||
| Fig. 6 The adsorption and photocatalytic degradation of RhB under visible light irradiation (λ > 400 nm). (RhB: 2 × 10−5 M, pH 5.6, 10% g-C3N4/PAN: 1 g L−1). | ||
The pH value determines the surface charge properties of the photocatalyst, and influences the photocatalytic degradation and the adsorption behavior of pollutants. Fig. 7 displays the effect of pH on the adsorption and photocatalytic degradation of RhB over the g-C3N4/PAN nanofibers. Here, both the adsorption and photocatalytic degradation rate of RhB increased gradually with the decrease of pH. In Fig. 7b, the degradation rate of RhB was beyond 90% within 40 minutes at pH 3. Under neutral conditions, RhB can also be degraded completely within 3 h. However, in an alkaline environment, the degradation efficiency of RhB decreased significantly and the degradation rate of RhB was only 60% after 3 h. Therefore, the adsorption of RhB on the photocatalytic nanofibers played an important role in the photocatalytic degradation of RhB. In addition, the positive holes are more easily produced and are considered as the major oxidation species at low pH. In order to determine the optimal amount of g-C3N4, we studied the influence of the amount of g-C3N4 in the g-C3N4/PAN nanofibers on the photocatalytic activity under the same conditions. As can be seen in Fig. S5,† 10% g-C3N4/PAN nanofibers performed the highest photocatalytic activity. This is because the catalyst became aggregated more easily with the increasing amount of g-C3N4 (Fig. S1†).
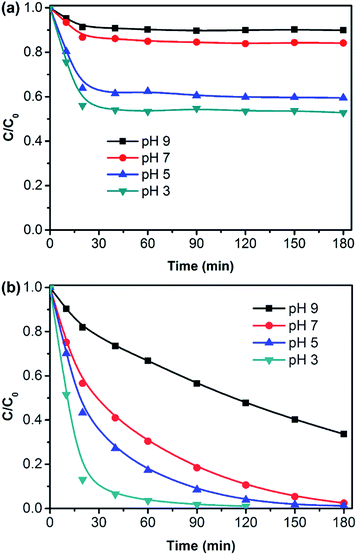 | ||
| Fig. 7 Effect of pH on adsorption of RhB in the dark (a) and photocatalytic degradation of RhB under visible light irradiation (λ > 400 nm) (b). (RhB: 2 × 10−5 M, 10% g-C3N4/PAN: 1 g L−1). | ||
To further investigate the reusability of the g-C3N4/PAN nanofibers, five repeated experiments of RhB degradation were carried out under the same conditions. Each cycle the fibers were separated from the solution, rinsed with ultrapure water and then dried at 70 °C. The results of the cycling experiments for the degradation of RhB driven by visible light irradiation are shown in Fig. 8, and there was no evident deactivation of the photocatalyst after five cycles. Therefore, it can be speculated that the g-C3N4/PAN nanofibers exhibited excellent self-reliant regeneration and recycling activity, indicating that the polyacrylonitrile-dispersed g-C3N4 nanofibers have broad application prospects for removing organic pollutants.
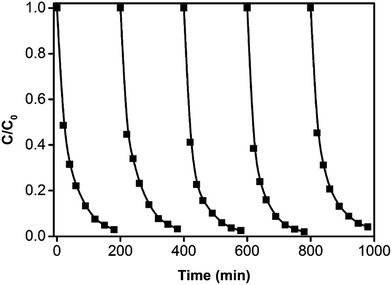 | ||
| Fig. 8 The cyclic photocatalytic degradation of RhB under visible light irradiation (λ > 400 nm). (RhB: 2 × 10−5 M, pH 5.6, 10% g-C3N4/PAN: 1 g L−1). | ||
In view of the practical application of the g-C3N4/PAN nanofibers in the degradation of organic pollutants, direct sunlight was used to degrade RhB. The g-C3N4/PAN nanofiber membrane can float on the liquid surface, however, the majority of the g-C3N4 powder settled to the bottom of the solution in the case of no stirring (Fig. S7†). As shown in Fig. 9, no photocatalytic degradation of RhB under solar irradiation was observed in the absence of catalysts, and the removal rate of RhB based on the g-C3N4/PAN nanofibers was higher than that of the pure g-C3N4 after irradiation for 7 h in the sunlight. As for the g-C3N4 powder, the agglomeration and subsidence of the powder catalyst led to decreased photocatalytic performance. In addition, the dye solution can also absorb a part of sunlight, resulting in a reduced intensity of light on the bottom of the catalyst. Compared with the g-C3N4 powder, the g-C3N4/PAN nanofiber membrane floated on the surface of the solution to overcome the disadvantageous agglomeration and subsidence, resulting in significantly enhanced photocatalytic activity.
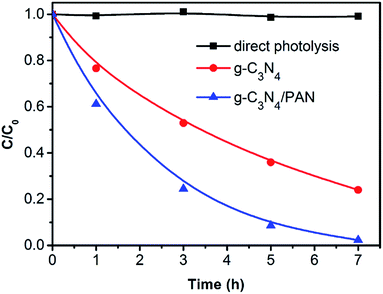 | ||
Fig. 9 Removal of RhB under solar irradiation. (Average sunlight intensity: 46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 760 lux, RhB: 2 × 10−5 M, g-C3N4: 0.03 g L−1, 10% g-C3N4/PAN: 0.3 g L−1). 760 lux, RhB: 2 × 10−5 M, g-C3N4: 0.03 g L−1, 10% g-C3N4/PAN: 0.3 g L−1). | ||
3.3 Degradation pathway and photocatalytic mechanism
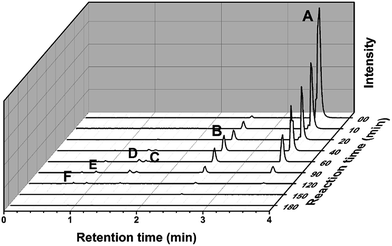
A detailed characterization of the intermediates generated during degradation is helpful to elucidate the decolorization route and possible mechanism, and is also useful for the environmental safety assessment.42 The molecular weights and structural information of the dye intermediates are helpful for proposing the degradation pathway and providing strong evidence for the degradation mechanism. To identify the products, UPLC Synapt G2-S HDMS was employed to analyze the intermediates, and GC-MS was used to detect small molecule products. Fig. 10 shows the UPLC Synapt G2-S HDMS chromatograms of the reaction intermediates. Five N-de-ethylated intermediates of the sample solution were examined easily in the positive mode. Based on the retention times and UPLC Synapt G2-S HDMS spectra peaks, A–F have been identified (Table S2†), namely as the parent compound RhB, N,N-diethyl-N′-ethylrhodamine (DER), N-ethyl-N′-ethylrhodamine (EER), N,N-diethylrhodamine (DR), N-ethylrhodamine (ER), and rhodamine (R), respectively, which is consistent with the sequential removal of the N-ethyl groups from the parent RhB molecule. The peak (A) of RhB gradually declines, while the peak (B), the peak (C), the peak (D), the peak (E) increased in the initial stage of the reaction, and then decreased gradually. After visible light irradiation for 180 min, these formed intermediate products almost disappeared. Additionally, another two products were detected in the negative mode. They were identified as benzoic acid (G) and phthalic acid (H).To further identify the intermediates, the GC-MS technique was employed to detect the small molecule products. Five aliphatic acids were identified, including oxalic acid, malonic acid, maleic acid, succinic acid and malic acid (Table S3†). In general, it was inferred that N-de-ethylation chromophore cleavage and ring-opening mineralization were the main processes in RhB degradation. Based on the results of the UPLC Synapt G2-S HDMS and GC-MS, the possible degradation pathway of RhB is shown in Fig. 11. It’s very similar to the previous reports.43,44
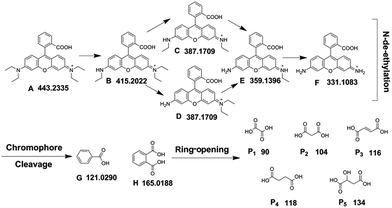 | ||
| Fig. 11 Photocatalytic degradation pathway of RhB over g-C3N4/PAN nanofibers (λ > 400 nm). (RhB: 2 × 10−5 M, pH 5.6, 10% g-C3N4/PAN: 1 g L−1). | ||
In order to evaluate the degradation mechanism of RhB over g-C3N4/PAN nanofibers, different active species capture agents such as isopropanol (IPA) benzoquinone (BQ)45,46 and potassium iodide (KI)25 were added to observe the effect on the degradation efficiency of dyes, as shown in Fig. 12. When adding IPA as a hydroxyl radical (˙OH) capture agent, the photodegradation efficiency only declined slightly, implying ˙OH was not the major active species in this photocatalytic degradation process. Nevertheless, when BQ as a superoxide radical (˙O2−) capture agent was added or N2 was continuously bubbled into the reaction solution, the photodegradation activity of RhB declined obviously, suggesting that ˙O2− plays a key role in this photodegradation system. With the addition of the hole (h+) scavenger KI, a significant inhibition effect of photocatalytic activity was observed, which confirms the important role of h+ in the photodegradation process.
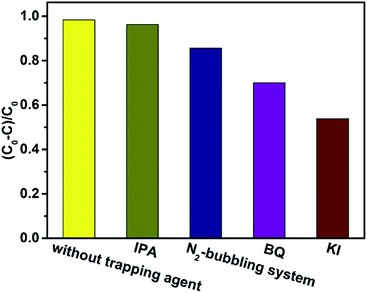 | ||
| Fig. 12 Effect of trapping agents on photocatalytic degradation of RhB under visible light irradiation (λ > 400 nm). (RhB: 2 × 10−5 M, pH 5.6, 10% g-C3N4/PAN: 1 g L−1). | ||
In addition, EPR was utilized to probe the possible reactive oxygen species generated in the catalytic reaction, and 5,5-dimethylpyrroline-oxide (DMPO) was used as the spin trapping reagent. In aqueous solution (Fig. S8a†), DMPO–˙OH signals were detected weakly in the systems, which means that ˙OH is not the major active species with the g-C3N4/PAN nanofibers. In methanol solution (Fig. S8b†), the g-C3N4/PAN nanofibers can produce the active species of ˙O2−, which reveals that ˙O2− plays an important role in the g-C3N4/PAN nanofiber photocatalytic system. The results were consistent with the above capture experiment results. Thus, it can be concluded that the photogenerated h+ and ˙O2− played a dominant part in the degradation process. It is quite different from the photocatalytic system of Yan and coworkers’.47 In their boron-doped g-C3N4 catalytic system under visible light irradiation, the degradation of methyl orange (MO) originated mainly from the photogenerated electrons, whereas the photodegradation of RhB is attributed mainly to the photogenerated holes. In our catalytic system, the central carbon of RhB could be attacked by h+ and ˙O2− to decolorize the dye and further degraded via an N-de-ethylation process. The N-de-ethylation intermediates DER, EER, DR, ER and R could be further carboxylated into aromatic acids with m/z values of 165 and 121. Finally, these aromatic compounds can be degraded into small molecular biodegradable acids by ring-opening.
4 Conclusions
A facile synthetic strategy to prepare visible-light responsive electrospun nanofibers based on polyacrylonitrile-dispersed g-C3N4 was developed to disperse and reuse g-C3N4. This supported catalyst can overcome the disadvantageous agglomeration and subsidence of powder g-C3N4, resulting in significantly enhanced photocatalytic activity under solar irradiation. In addition, UPLC Synapt G2-S HDMS and GC-MS technologies were employed to identify the intermediates, and the main degradation pathway included the N-de-ethylation and ring-opening processes. Based on the different capture experiments for the active species and EPR results, the photogenerated h+ and ˙O2− dominated the oxidation degradation of RhB. Therefore, this work provides a practical method to improve the photocatalytic activity of the powder catalyst and has potential applications for water purification using sunlight.Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 51133006 and 51103133), Textile Vision Science & Education Fund, 521 Talent Project of ZSTU, Zhejiang Provincial Natural Science Foundation of China (No. LY14E030013), and the Public Welfare Technology Application Research Project of Zhejiang Province (No. 2015C33018).Notes and references
- H. Zhao, H. Yu, X. Quan, S. Chen, H. Zhao and H. Wang, RSC Adv., 2014, 4, 624 RSC.
- W. Zhao, W. Ma, C. Chen, J. Zhao and Z. Shuai, J. Am. Chem. Soc., 2004, 126, 4782–4783 CrossRef CAS PubMed.
- H. Shon, S. Vigneswaran, H. Ngo, J.-H. Kim and J. Kandasamy, Sep. Purif. Technol., 2007, 56, 388–391 CrossRef CAS PubMed.
- C.-Y. Kuo, C.-H. Wu and H.-Y. Lin, Desalination, 2010, 256, 37–42 CrossRef CAS PubMed.
- S. Zhang, J. Li, M. Zeng, G. Zhao, J. Xu, W. Hu and X. Wang, ACS Appl. Mater. Interfaces, 2013, 5, 12735–12743 CAS.
- E. G. Gillan, Chem. Mater., 2000, 12, 3906–3912 CrossRef CAS.
- Y. Zhang, A. Thomas, M. Antonietti and X. Wang, J. Am. Chem. Soc., 2009, 131, 50–51 CrossRef CAS PubMed.
- M. Deifallah, P. F. Mcmillan and F. J. Corà, J. Phys. Chem. C, 2008, 112, 5447–5453 CAS.
- X. Wang, K. Maeda, A. Thomas, K. Takanabe, G. Xin, J. M. Carlsson, K. Domen and M. Antonietti, Nat. Mater., 2009, 8, 76–80 CrossRef CAS PubMed.
- J. Zhang, X. Chen, K. Takanabe, K. Maeda, K. Domen, J. D. Epping, X. Fu, M. Antonietti and X. Wang, Angew. Chem., Int. Ed., 2010, 49, 441–444 CrossRef CAS PubMed.
- Y. Zhang and M. Antonietti, Chem.–Asian J., 2010, 5, 1307 CAS.
- X. Wang, K. Maeda, X. Chen, K. Takanabe, K. Domen, Y. Hou, X. Fu and M. Antonietti, J. Am. Chem. Soc., 2009, 131, 1680–1681 CrossRef CAS PubMed.
- Y. Wang, X. Wang and M. Antonietti, Angew. Chem., Int. Ed., 2012, 51, 68–89 CrossRef CAS PubMed.
- X. Wang, S. Blechert and M. Antonietti, ACS Catal., 2012, 2, 1596–1606 CrossRef CAS.
- X. Zhang, L. Yu, C. Zhuang, T. Peng, R. Li and X. Li, ACS Catal., 2014, 4, 162–170 CrossRef CAS.
- S. Yang, Y. Gong, J. Zhang, L. Zhan, L. Ma, Z. Fang, R. Vajtai, X. Wang and P. M. Ajayan, Adv. Mater., 2013, 25, 2452–2456 CrossRef CAS PubMed.
- X. Zhang, X. Xie, H. Wang, J. Zhang, B. Pan and Y. Xie, J. Am. Chem. Soc., 2013, 135, 18–21 CrossRef CAS PubMed.
- Y. Tang, H. Song, Y. Su and Y. Lv, Anal. Chem., 2013, 85, 11876–11884 CrossRef CAS PubMed.
- L.-S. Lin, Z.-X. Cong, J. Li, K.-M. Ke, S.-S. Guo, H.-H. Yang and G.-N. Chen, J. Phys. Chem. B, 2014, 2, 1031–1037 CAS.
- J. Jin, X. Fu, Q. Liu and J. Zhang, J. Phys. Chem. A, 2013, 1, 10538 CAS.
- X. Dai, M. Xie, S. Meng, X. Fu and S. Chen, Appl. Catal., B, 2014, 158–159, 382–390 CrossRef CAS PubMed.
- Y. Wang, J. Yao, H. Li, D. Su and M. Antonietti, J. Am. Chem. Soc., 2011, 133, 2362–2365 CrossRef CAS PubMed.
- Y. Li, X. Xu, P. Zhang, Y. Gong, H. Li and Y. Wang, RSC Adv., 2013, 3, 10973 RSC.
- T. Sano, S. Tsutsui, K. Koike, T. Hirakawa, Y. Teramoto, N. Negishi and K. Takeuchi, J. Mater. Chem. A, 2013, 1, 6489 CAS.
- C. Chang, L. Zhu, S. Wang, X. Chu and L. Yue, ACS Appl. Mater. Interfaces, 2014, 6, 5083–5093 CAS.
- H. Katsumata, T. Sakai, T. Suzuki and S. Kaneco, Ind. Eng. Chem. Res., 2014, 53, 8018–8025 CrossRef CAS.
- X. Jin, V. V. Balasubramanian, S. T. Selvan, D. P. Sawant, M. A. Chari, G. E. Q. Lu and A. Vinu, Angew. Chem., Int. Ed., 2009, 48, 7884–7887 CrossRef CAS PubMed.
- M. Tahir, C. Cao, N. Mahmood, F. K. Butt, A. Mahmood, F. Idrees, S. Hussain, M. Tanveer, Z. Ali and I. Aslam, ACS Appl. Mater. Interfaces, 2014, 6, 1258–1265 CAS.
- Y. Cui, Z. Ding, P. Liu, M. Antonietti, X. Fu and X. Wang, Phys. Chem. Chem. Phys., 2012, 14, 1455–1462 RSC.
- P. Madhusudan, J. Ran, J. Zhang, J. Yu and G. Liu, Appl. Catal., B, 2011, 110, 286–295 CrossRef CAS PubMed.
- P. Madhusudan, J. Ran, J. Zhang, J. Yu and G. Liu, Appl. Catal., B, 2011, 110, 286–295 CrossRef CAS PubMed.
- M. T. Uddin, Y. Nicolas, C. Olivier, T. Toupance, L. Servant, M. M. Muller, H. J. Kleebe, J. Ziegler and W. Jaegermann, Inorg. Chem., 2012, 51, 7764–7773 CrossRef CAS PubMed.
- S. Ye, L.-G. Qiu, Y.-P. Yuan, Y.-J. Zhu, J. Xia and J.-F. Zhu, J. Mater. Chem. A, 2013, 1, 3008 CAS.
- S. Kumar, T. Surendar, B. Kumar, A. Baruah and V. Shanker, J. Phys. Chem. C, 2013, 117, 26135–26143 CAS.
- Y. Ishida, L. Chabanne, M. Antonietti and M. Shalom, Langmuir, 2014, 30, 447–451 CrossRef CAS PubMed.
- J. S. Im, M. I. Kim and Y.-S. Lee, Mater. Lett., 2008, 62, 3652–3655 CrossRef CAS PubMed.
- J.-H. Park, Y.-W. Ju, S.-H. Park, H.-R. Jung, K.-S. Yang and W.-J. Lee, J. Appl. Electrochem., 2009, 39, 1229–1236 CrossRef CAS.
- F. Dong, L. Wu, Y. Sun, M. Fu, Z. Wu and S. C. Lee, J. Mater. Chem., 2011, 21, 15171 RSC.
- G. Liu, P. Niu, C. Sun, S. C. Smith, Z. Chen, G. Q. Lu and H.-M. Cheng, J. Am. Chem. Soc., 2010, 132, 11642–11648 CrossRef CAS PubMed.
- F. He, G. Chen, Y. Yu, S. Hao, Y. Zhou and Y. Zheng, ACS Appl. Mater. Interfaces, 2014, 6, 7171–7179 CAS.
- J. Hong, X. Xia, Y. Wang and R. Xu, J. Mater. Chem., 2012, 22, 15006–15012 RSC.
- L. Liu, J. Zhang, Y. Tan, Y. Jiang, M. Hu, S. Li and Q. Zhai, Chem.–Eng. J., 2014, 244, 9–18 CrossRef CAS PubMed.
- Z. He, C. Sun, S. Yang, Y. Ding, H. He and Z. Wang, J. Hazard. Mater., 2009, 162, 1477–1486 CrossRef CAS PubMed.
- T. S. Natarajan, M. Thomas, K. Natarajan, H. C. Bajaj and R. J. Tayade, Chem.–Eng. J., 2011, 169, 126–134 CrossRef CAS PubMed.
- W. Li, D. Li, Y. Lin, P. Wang, W. Chen, X. Fu and Y. Shao, J. Phys. Chem. C, 2012, 116, 3552–3560 CAS.
- J. Cao, B. Luo, H. Lin, B. Xu and S. Chen, Appl. Catal., B, 2012, 111–112, 288–296 CrossRef CAS PubMed.
- S. C. Yan, Z. S. Li and Z. G. Zou, Langmuir, 2010, 26, 3894–3901 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: The synthesis of g-C3N4, g-C3N4/PAN nanofibers, spectral changes during photocatalytic degradation of RhB under visible light irradiation, the photograph of photocatalytic degradation of RhB by g-C3N4 powder and g-C3N4/PAN nanofibers under solar irradiation, the detailed condition for UPLC Synapt G2-S HDMS and GC-MS. See DOI: 10.1039/c5ra15973c |
| This journal is © The Royal Society of Chemistry 2015 |