Open Access Article
Open Access ArticleBiomedical materials for wound dressing: recent advances and applications
Hien Minh Nguyen *a,
Tam Thi Ngoc Lea,
An Thanh Nguyenb,
Han Nguyen Thien Lea and
Thi Tan Pham*b
*a,
Tam Thi Ngoc Lea,
An Thanh Nguyenb,
Han Nguyen Thien Lea and
Thi Tan Pham*b
aSchool of Medicine, Vietnam National University Ho Chi Minh City, Ho Chi Minh City, Vietnam. E-mail: nmhien@medvnu.edu.vn
bHo Chi Minh City University of Technology (HCMUT), Vietnam National University Ho Chi Minh City, Ho Chi Minh City, Vietnam. E-mail: ptthi@hcmut.edu.vn
First published on 13th February 2023
Abstract
Wound healing is vital to maintain the physiological functions of the skin. The most common treatment is the use of a dressing to cover the wound and reduce infection risk and the rate of secondary injuries. Modern wound dressings have been the top priority choice for healing various types of wounds owing to their outstanding biocompatibility and biodegradability. In addition, they also maintain temperature and a moist environment, aid in pain relief, and improve hypoxic environments to stimulate wound healing. Due to the different types of wounds, as well as the variety of advanced wound dressing products, this review will provide information on the clinical characteristics of the wound, the properties of common modern dressings, and the in vitro, in vivo as well as the clinical trials on their effectiveness. The most popular types commonly used in producing modern dressings are hydrogels, hydrocolloids, alginates, foams, and films. In addition, the review also presents the polymer materials for dressing applications as well as the trend of developing these current modern dressings to maximize their function and create ideal dressings. The last is the discussion about dressing selection in wound treatment and an estimate of the current development tendency of new materials for wound healing dressings.
1. Introduction
A wound is a type of injury that causes a loss of continuity in the skin, tissues, and mucous membranes. Based on the time and characteristics of the healing process, wounds are classified as acute wounds and chronic wounds. While acute wounds are skin wounds or surgical wounds, chronic wounds are pressure ulcers, leg ulcers, severe burns, and diabetic ulcers.1 Acute wounds often heal completely and without complications within four weeks, following the expected or predictable rate of healing.2 Compared to acute wounds, chronic wounds are more difficult to control the wound condition with slow healing time, persistence, and abnormal healing progress and can cause serious complications that require tissue removal.1 Both acute and chronic wounds have a severely detrimental impact on the world's healthcare systems and economies.3,4 In the United States, injury treatment costs about 50 billion dollars annually.5 According to Medicare, a health insurance program, the cost of wound care for beneficiaries ranges from 28.1 to 96.8 billion dollars per year, with surgical wounds and diabetic ulcers the most expensive, accounting for 38.3 and 18.7 billion dollars, respectively.6 Chronic wounds are common in elders over 65 years old.7 Therefore, with the aging population, the number of patients with chronic wounds is expected to increase and continues to be a long-lasting problem in this population.8 Modern dressings are commonly used to cover wounds and generate a moist environment for wound healing. Traditional wound dressings are often used in clinical practice because they are economical, yet fibers stick to the granulation tissue, causing pain when removing the dressing. On the other hand, modern dressings maintain ideal temperature and humidity for the wound to stimulate wound healing and protect the wound from external bacteria and prevent cross-infection.9 Moreover, some types of dressing including alginate or hydrogel dressing have the property non-adhesive to tissues, causing less pain during dressing changes for patients, overcoming the limitation of traditional dressing.10,11 With their advantages, many studies on modern dressings such as foams, hydrogels, alginates, hydrocolloids, and films are carried out to solve clinical problems in treating wounds.Here, we provide an overview of the modern dressings that have been developed for wound healing applications. We look over studies on the effectiveness of different dressings and consider the advantages and limits of wound treatment. In addition, the review also mentions new materials that are of interest to researchers for dressings applications in the future. Finally, we discuss the selection of modern dressings, and the trend of developing advanced materials to develop appropriate dressings in clinical treatment.
2. Physiology of wound healing and treatment
2.1. Physiology of wound healing
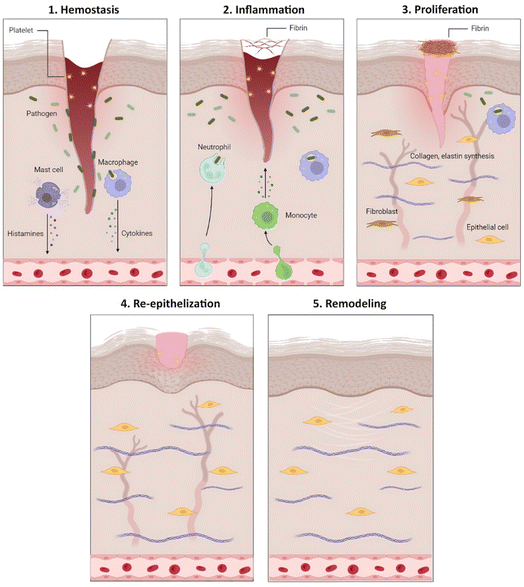
Wound healing is a complicated biological process that restores tissue integrity. Acute wounds usually heal in a relatively short time frame from four to six weeks, depending on the size, depth, and extent of damage in the epidermis and dermis of the skin and the operation of growth factors, cytokines, and matrix proteins.12 Physiologically, an acute wound healing is divided into five stages: hemostasis, inflammation, proliferation, re-epithelialization, and remodeling (Fig. 1).13Hemostasis is the first stage of wound healing. Clotting factors are activated in hemostasis and form a platelet knot to reduce blood loss from the injuries.14 When bleeding is under control, growth factors such as vascular endothelial growth factor (VEGF), epithelial growth factor (EGF), and cytokines are released to recruit neutrophils, monocytes, and lymphocytes to reach the injured tissue and promote the inflammatory stage.15 The inflammatory stage involves a series of responses involving neutrophils and cytokines.16 The cells participate in clearing away cells debris, and pathogens as well as releasing cytokines such as tumor necrosis factor α (TNF-α), interleukin 6, 1β (IL-6, IL-1β) to thrombolysis. The third stage is proliferation, in which platelets and leukocytes release cytokines, stimulate angiogenesis, fibroblast proliferation, collagen, and elastin synthesis to restore the dermis, leading to scar formation.15,17 The re-epithelialization stage is the re-establishment of intact epidermis over the newly formed tissue. Cells enhance collagen and elastin synthesis to increase skin elasticity and stability. Keratocytes migrate into the wound site while proteases released by macrophages remove excess extracellular matrix (ECM).18 The last stage of wound healing process is remodeling. During this stage, recently formed capillaries regress and most macrophages and fibroblasts undergo apoptosis.19
Chronic wounds last more than 12 weeks and heal at a much lower rate than acute wounds.20 Chronic wounds have abnormal sequences of epithelial regeneration due to poor tissue blood supply, tissue necrosis, infection, persistent trauma, cancer, and a high amount of matrix metalloproteinase (MMP). In addition, chronic wounds have a low rate of cell division and high levels of pro-inflammatory cytokines and proteases.21 Other factors affecting wound healing include nutritional deficiency, vitamin-C, zinc deficiency, and hormone deficiency such as insulin in diabetics.22,23 These factors prolong the inflammatory stage and interfere with wound healing by continually attracting macrophages and neutrophils to the wound.24–26 Due to abnormalities in the successive stages of chronic wound healing, tissue and epithelial regeneration are disrupted, and the wound becomes a severe ulcer.
2.2. Wound treatment
In wound treatment, wound dressings cover damaged skin, maintain a moist environment and appropriate temperature required for healing, provide physical and microbiological protection, absorb excessive tissue fluid, and provide pain relief.9 In the 1960s, medical experts recommended that humidifying the wound environment dramatically affects the wound healing process.27,28 Dried wounds with eschar require extra moisture to optimize healing and soften the eschar.29 On the other hand, excessive moisture leads to excessive hydration and damages the skin normal barrier function, causing ulceration.30 Since then, various dressings have been designed to optimize moisture levels and create the ideal wound healing environment. Moreover, an ideal dressing not only protects a wound and maintains wound humidity but is also biocompatible, biodegradable, non-toxic, and non-allergenic while promoting gas exchange, granulation, and re-epithelialization.31 Modern wound dressings may also contain pharmacologically active substances such as antibiotics, non-steroidal anti-inflammatory, analgesic, and local anesthetics medicines or natural extracts with anti-inflammatory, epithelializing, antioxidant, and antimicrobial properties.32,33 Different wounds have different physiological conditions, depth, location, and extent of the wound, the amount of discharge, infection, and wound adhesion. Therefore, it is necessary to choose the proper dressing to promote the healing process or make it worse. Therefore, healthcare workers have to understand the condition of the wound and the characteristics of each dressing to choose appropriately and promote the healing rate and quality of healing.3. Classification of modern wound dressings
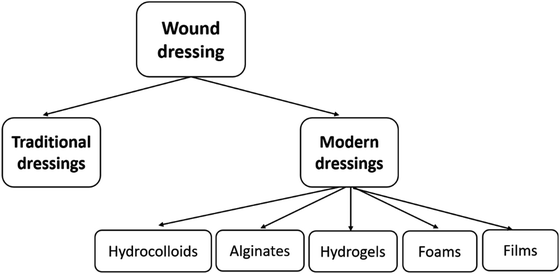
For many years, traditional dressings such as cotton wool, lint, gauze have been widely used to ensure the wound clean and prevent getting bacteria infection. However, the dressings easily stick to the wound and do not create a suitable moist environment. Modern dressings have been developed with better-improved biocompatibility, degradability, pain relief, and moisture retention. Rather than just covering wound itself, modern wound dressings also act as facilitation for the function of the wound.34 Several modern dressings currently used in clinical practice include hydrocolloid, alginate, hydrogel, foam, and film dressings (Fig. 2).3.1. Hydrocolloid dressings
Hydrocolloid dressings can absorb a relatively large amount of wound fluids38 as well as be virtually impermeable to water vapor, promoting the formation of a moist healing environment.39 Furthermore, they are impermeable to oxygen, which accelerates epithelialization and collagen synthesis and decreases the wound exudates' pH, thus reducing the number of bacteria.40 They also prevent contamination, promote autolysis to remove damaged or infected tissues, and do not require secondary dressings.35,37,41
Hydrocolloid dressings are often incorporated with active ingredients in treating pressure ulcers or lower-extremity ulcers. They are indicated for low to moderate exuding wounds,40 granular or necrotic wounds, and other acute wounds, including partial and full-thickness burns,35 surgical or postsurgical wounds in children.42 Some hydrocolloid dressing products are shown in Table 1. However, this dressing is unsuitable for high exudate because there might lead to accumulation around the wound site.43 To address the issues, the dressings need to be changed many times a week. Also, because of the adhesive nature that can damage the fragile surrounding skin, hydrocolloid dressings should not be used on infected wounds.35
| Type | Product name | Components | Main features | Applications | References |
|---|---|---|---|---|---|
| Hydrocolloid | DuoDerm® | Pectin, gelatin, CMC and PU | The outer layer provides a waterproof barrier | Managing wounds with light to moderate exudate: Stage I and Stage II pressure ulcers, burn/scald, abrasions, lacerations, and reducing infection | 50 |
| The matrix of hydrocolloid particles absorbs exudate | |||||
| They are keeping a moisture environment | |||||
| Comfeel®Plus | NaCMC and calcium alginate is added | Allows moisture to evaporate while maintaining a moist healing environment | Chronic wounds such as leg ulcers and pressure ulcers; acute wounds such as superficial burns, superficial partial-thickness burns, donor sites, postoperative wounds and skin abrasions | 51 | |
| Water and bacteria proof to protect from external factors | |||||
| Cutinova® Hydro | Absorbent colloid covered with semiocclusive polyurethane film covering | Selective absorption: water is taken into the matrix and locked away | Venous leg ulceration, diabetic ulcers, slough, and necrotic tissues | 52 | |
| Highly absorptive granules: absorb the fluid 10 times its own weight | |||||
| Does not form a gel substance | |||||
| Replicare® | Absorbent colloid covered with polyurethane film | Cohesive properties keep the wound free of dressing residue | Partial to full thickness wound: ulcers (venous, arterial, diabetic) pressure sores, donor sites, surgical incisions an excision, burns (grade 1st and 2nd) | 53 | |
| Creation and maintenance of a moist wound environment | |||||
| Alginate | Kaltostat® | Calcium alginate with guluronic acid | Supporting moist wound healing environment | Moderate to highly exuding wounds and for wounds with minor bleeding: leg ulcers, pressure ulcers, diabetic ulcers and fungating lesions, donor sites, abrasions, lacerations and post-surgical wounds | 54 |
| Promoting haemostatic, stop minor bleeding | |||||
| Algicell® Ag | Calcium alginate with 1.4% silver | Calcium alginate with 1.4% silver | Diabetic foot ulcer, leg ulcers, pressure ulcers, donor sites, and traumatic and surgical wounds | 55 | |
| Minimizing fibrous residue | |||||
| Maintaining a moist wound environment | |||||
| Antibacterial activity: Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Candida albicans | |||||
| Guardix-SG® | Containing sodium alginate and poloxamer | Prevents adhesion by forming a physical barrier on the surface of the wound tissue | Reduce the adhesion upon spine, thyroids, and abdominal surgery | 56 | |
| Algivon® | Alginate dressing impregnated with manuka honey | Anti-bacterial and anti-inflammatory | Eliminates odors | 57 | |
| Maintaining the ideal moist wound healing environment | Apply for pressure ulcers, leg ulcers, diabetic ulcers, surgical wounds, burns, graft sites, infected wounds | ||||
| Fibracol™ Plus | 90% collagen and 10% calcium alginate | Maintaining physiologically moist microenvironment to support granulation tissue formation, epithelialization and rapid wound healing | Full-thickness and partial- thickness wounds: venous ulcers, ulcers caused by mixed vascular etiologies, diabetic ulcers, second degree burns, donor sites and other bleeding surface wounds | 58 | |
| Tromboguard® | Alginate, chitosan, polyurethane | Haemostatic activity and control severe bleeding | Used to stop bleeding in traumatic and postoperative wounds, bleeding from accidents | 59 and 60 | |
| Antibacterial activity | |||||
| Hydrogel | ActivHeal® | 85% of water and collection of polymer chains | Rehydrating dry necrotic wounds | Dry and sloughy wounds with nil to low exudate: pressure ulcers, cavity wounds, leg ulcers, graft and donor sites, diabetic ulcers, post op surgical wounds, lacerations and abrasions | 61 |
| Providing a moist wound environment | |||||
| Removing devitalized tissue | |||||
| Encouraging growth and migration of epithelial cells | |||||
| Restore® | Sodium polyacrylate, hyaluronic acid | Providing a moist wound environment | Stages II–IV pressure ulcers, diabetic skin ulcers, venous ulcers, 1st and 2nd degree burns, skin tears, cuts, abrasions, and conditions associated with peristomal care | 62 | |
| Non-greasy, non-staining, and fragrance free | |||||
| Suprasorb® G | CMC polymers, polyethylene and acrylic polymers | Gently removing necrotic tissue | Dry wounds and wounds with low amounts of exudate, acute or chronic superficial wounds (lower leg ulcers, pressure ulcers, first- and second-degree burns, scalds) | 63 | |
| Supplying moisture | |||||
| Relieving wound pain | |||||
| Easy to mould | |||||
| Drying wounds and wounds with low amounts of exudate | |||||
| Aquaderm™ | 2-Acrylamido-2 methyl-1 propanesulfonic acid sodium, poly(ethylene glycol) dimethacrylate, 2-hydroxy-2-methylpropiophenone with 38–55% water | Providing soothing coolness | Pressure ulcers, minor burn, radiation tissue damage | 64 | |
| Non-traumatic, one-piece removal | |||||
| Supporting moist wound healing | |||||
| Bacteria and fluid barrier | |||||
| Absorbing minor exudate | |||||
| Transparent - easy to monitor wound | |||||
| Can be cut to fit wound site | |||||
| Neoheal® | 90% water and polyvinylopyrrolidone, PEG, agar | Maintaining of an optimal moist environment | First degree of burn wounds, second and third degree of burn wounds, ulcerations, bedsores | 65 | |
| Cooling, soothing local pain and discomfort | |||||
| Protecting wounds from external contamination | |||||
| Absorbing wound secretions | |||||
| Permeable to water vapor and oxygen but impermeable to bacteria | |||||
| Allowing the dressing to be removed without trauma | |||||
| Conforms easily to any contour of the body | |||||
| Transparent to enable wound observation and assessment of healing process | |||||
| Biocompatible, non-cytotoxic, non-allergenic | |||||
| Foam | Allevyn life | Hydrophilic polyurethane foam | Providing coverage to the malleolus without the need for secondary retention | Pressure ulcers | 66 |
| Fitting the contours of the human body securely and allowing patients to shower | |||||
| Minimizing the visual impact of strikethrough, providing the patient with confidence that their dressing may not attract the negative attention of others | |||||
| Allowing the dressing to be repositioned | |||||
| Permafoam® | Absorbent foam made of polyurethane | Transporting rapidly wound exudate and ensuring a balanced moist wound environment | Medium to heavily exuding wounds, deeper wounds | 67 | |
| Bounding safely germs and cell debris even under compression | |||||
| Soft and smooth with good padding properties | |||||
| Available in different sizes and shapes | |||||
| HydroTac® | Hydrated polyurethane polymer and propylene glycol | Preventing the dressing from sticking to the wound | Wounds during the granulation and epithelialization phases with low to moderate exudation | 68 | |
| Absorbing wound exudate and releasing moisture | |||||
| Non-border dressing can be cut into the desired size and use a suitable secondary dressing | |||||
| Mepilex Ag | Polyurethane foam containing Ag | Soft and conformable making it easy to use and for many situations | Acute and chronic wounds | 69 | |
| Minimizing pain during dressing changes | |||||
| Minimizing the risk of leakage | |||||
| Inactivate microbial in wound (bacteria and fungi) | |||||
| Tegaderm™ Silicone foam dressing | Polyurethane foam with silicone adhesive | Absorbing and evaporating moisture to help reduce the potential for skin maceration | Pressure injuries, venous leg ulcers, neuropathic ulcers, arterial ulcers, skin tears, and surgical wounds | 70 | |
| Ability to access and assess skin | |||||
| Ability to manage microclimate | |||||
| Ease of application and removal | |||||
| Correcting dressing size for high-risk locations | |||||
| Film | Bioclusive™ Plus | Transparent polyurethane film coated with acrylic adhesive | Providing a barrier to viruses 27 nm in diameter or larger and bacteria | No or light levels of exudate, suitable for secondary cover dressing and securing of catheters; minor burns, donor sites, superficial pressure areas and leg ulcers | 71 |
| Allowing the transmission of oxygen and moisture vapor | |||||
| Mepitel | Two-sided wound contact layer coating with silicone | Preventing maceration by inhibiting the lateral movement of exudate | Skin tears or abrasions, surgical excisions, second-degree burns, blistering conditions such as epidermolysis bullosa, partial and full thickness grafts, and skin damage following radiotherapy or steroid therapy | 72 | |
| Gentle adhesion helping remove with minimum pain and without damaging new tissue | |||||
| Mepore® | Polyurethane coated with polyacrylic adhesive | Bacterial and viral barrier for microbes larger than 25 nm | Secondary dressing for fixation of medical devices such as wound dressings, tubes and cannulas | 73 | |
| Allowing moisture and skin transpiration to be evaporated | |||||
| Transeal® | Polyurethane coated with acrylic, pressure-sensitive adhesive | Maintaining a proven high moisture vapor transmission rate | Vascular access sites, first and second degree burns, superficial wounds, surgical incisions | 74 | |
| Impermeable to water and bacteria |
3.2. Alginate dressings
Alginate is a popular biomaterial known for retaining a structure similar to ECM, exhibiting high biocompatibility. Alginate dressings absorb exudates from the wound, creating a moist environment for wound healing.78 When applied to the wound surface, alginate forms a gel and easily sloughs when removing the dressing or rinsing with sterile saline.79 It can be in the form of porous sheets or fibrous dressings when further processed.80 Alginate performs high absorbent properties but also maintains structural integrity stability.81–83 Additionally, alginate dressings can reduce inflammation, wound odor, act as hemostatic agents, and have good permeability to oxygen, other gases, or liquids.84 Therefore, alginate is suitable for treating acute and chronic wounds such as diabetic foot ulcers, pressure ulcers, burns, and infected surgical wounds.75
Since alginate cannot self-adhesive, a secondary dressing is required.85 In addition, if the alginate does not absorb enough fluid to form the gel, it is possible to leave excess fibers in the wounds due to the fibrous nature of alginates.86 This phenomenon may trigger inflammatory mechanisms against foreign agents. There had been reports of an allergy where there was insufficient moisture in the wound to form a gel87 despite alginate's high biocompatibility.
With high biocompatibility, to increase the effectiveness of treatment, the current bandage combines alginate with many antibacterial or anti-inflammatory compounds such as ZnO nanoparticles or Edaravone. The results of the in vivo model showed that the above combined materials are safe, capable of closing the wound through keratinocyte adhesion, cell proliferation89 or reducing inflammation.90 Shafei et al. study promotes tissue regeneration by exosome loaded alginate hydrogel.91 This bioactive dressing not only improved wound closure, collagen synthesis, and vessel formation but also was biodegradable and biocompatible.91 In addition, alginate is also combined with many naturally derived extractions such as Malva sylvestris92 or oregano essential oil,93 which shows antimicrobial,93 anti-inflammatory and high biocompatibility properties.92
Clinically, alginate dressing had been shown to be effective in healing processes, causing less pain during dressing changes for patients.11,94 Another randomized study evaluated the clinical therapeutic effect of human granulocyte-macrophage colony-stimulating factor incorporated with alginate dressings in the treatment of refractory chronic skin ulcers. The combination showed many advantages, such as promoting granulation tissue growth, accelerating epithelial regeneration, and effectively alleviating wound pain, thus improving the patient's quality of life.95
3.3. Hydrogel dressings
Hydrogel dressings can provide a moist environment in the wound site and make favorable conditions for tissue regeneration. In addition, this characteristic also gives a comfortable, soothing effect to the wound, especially severe wounds.97,98 However, this fluid accumulation can also lead to skin infections and bacterial growth, giving off foul odors in infected wounds.31,97 Moreover, the hydrogel wound dressing is non-adhesive to the wound or tissues, releasing pain during dressing changes yet not disrupting wound healing.83,96,99 The tuneable mechanical properties of hydrogels enhance their suitability for various wounds.97
Hydrogel dressing products are usually suitable for pressure ulcers, diabetic foot ulcers, skin tears, and surgical wounds and burns, including minor burns, first and second-degree burns.10,99 Hydrogel dressings should be used either with a secondary dressing such as film or foam or without secondary dressing.99
Furthermore, there has been a growing interest in incorporating nanoparticles or nanostructures into hydrogels for improving the properties of hydrogel wound dressings.97,105 Silver is one of the most common nanomaterials combined with hydrogel dressings to enhance the treatment of infected wounds.105,106 Many randomized controlled trials were carried out to evaluate the effectiveness of hydrogel/AgNP dressings and showed the positive results in wound healing.105,106 However, the limitation of hydrogel/AgNP dressing is the high cost.106 Besides AgNP, another study improved the mechanical of hydrogels by incorporating nano-clay with polydopamine and polyacrylamide.107 This hydrogel displayed superior toughness owing to nanoreinforcement by clay and polydopamine-induced cooperative interactions with the hydrogel networks.107 In addition, a new generation of smart hydrogel wound dressings that contain sensors has been developed rapidly for the ability to reveal wound conditions. Some remarkable studies can be mentioned such as flexible pH-sensing hydrogel fibers based on alginate in skin wounds,108 polyvinyl alcohol/xyloglucan (PVA/XG) hydrogel membrane with the ability to absorb exudate and release biological factors.109
3.4. Foam dressings
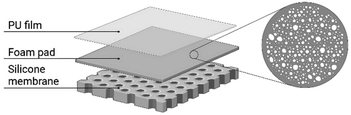
To provide water and microbial resistant barrier to the environment, foam dressings are commonly supplied with a film-backing110 or a silicone membrane111 for keeping the dressing in the right wound site and protecting the wound from trauma when changing the dressings (Fig. 6). Foam dressings can be kept for up to a week, depending on the level of exudate.112 Nevertheless, this advantage is also the disadvantage of foam dressing because not frequent changes may affect the growth of new tissue and damage wounds when removing the dressing.113
Foam dressings are used to treat minimal to moderate wounds, proper for burns, chronic wounds, and deep ulcers.114 However, these dressings are not appreciated for epithelial dry wounds, necrotic wounds, and wounds requiring frequent care.
To meet the demand for treating various wounds, researchers have developed many new foam dressings. Namviriyachote et al. developed an innovative PU foam dressing comprising hydroxypropylmethylcellulose and alginate, containing silver and asiaticoside (AS).117 The findings showed that the foam dressing released AgNP at a 1 mg cm−2 of silver dosage in the formula with 6% alginate and 5% AS for the most optimal antibacterial activity. This dressing improved wound healing both wound closure rate and histological parameters of skin wounds and had no dermatological reactions occurred.117 Another new foam dressing is mentioned to be the povidone-iodine foam dressing, known as Betafoam. To provide evidence about the effectiveness of Betafoam, a clinical trial was performed comparing Betafoam with Medifoam, a commercially available foam dressing for treating diabetic foot ulcers.118 The trial suggests that Betafoam is safe, has a wound healing ability similar to Medifoam, and has no adverse effects.
3.5. Film dressing
4. Polymer materials for wound dressings
Polymers are commonly used in pharmaceutical and biomedical areas due to their biocompatibility and their similarity with ECM. The generated substrate can mimic the biological environment that helps cells participate in proliferation, differentiation,127 and repair damaged tissue.128 In addition, some polymers also have biodegradable and bioresorbable properties to promote the reconstruction of new tissue without inducing the inflammation.129 In this part, a brief introduction of the representative polymers and their recent application was presented.4.1 Cellulose and bacterial cellulose
Cellulose is a major polysaccharide in the cell wall of a plant. Cellulose comprises many glucose units in a linear form which are linked together by β-1,4.130 With the presence of hydrogen bonds that hold the oxygen atoms and hydroxyl groups together, the linear structure of cellulose is well maintained and exhibited the biomechanics property.130,131 As cellulose is formed from the glucose subunits, it is naturally biocompatible with human tissue and can be easily modified without affecting the structural and mechanical properties.131 Due to the semi-crystalline property in an aqueous state, cellulose can present biological effects when modified. However, pure cellulose has a major drawback due to its poor solubility in organic solvents.132 At below 300 °C, cellulose is completely insoluble and degraded when above this temperature.133 To improve the dissolution ability, cellulose derivatives include cellulose esters (cellulose acetate), ethers (carboxymethyl cellulose, methyl cellulose, and ethyl cellulose), and cellulose sulfate represent alternatives to pure cellulose.133In contrast to plant cellulose, some bacteria, algae, and fungi produce cellulose through oxidative fermentation, which is called bacterial cellulose (BC) or microbial cellulose.134 The BC is usually synthesized by Gram-negative bacteria such as Acetobacter, Agrobacterium, Komagataeibacter (formerly Gluconacetobacter), Achromobacter, Azobacter, Rhizobium, Salmonella or Gram-positive bacterium Sarcina ventriculi.135 BC is considered a more biocompatible version of plant cellulose since it is free of lignin, hemicellulose, and pectin.133 Therefore, BC exhibits specific characteristics, high water holding capacity due to being very hydrophilic, a large surface area, high crystallinity, and high mechanical strength.133,135,136 BC absorbs well fluid from the wound due to high water holding capacity, the water molecules bind to the hydroxyl group in the cellulose chain. The high porosity combined with the large surface area suggests that BC can interact with antibacterial drugs or other active ingredients.137 Despite the advanced characteristics, the high cost of BC production hinders industrial implementation. Therefore, the industrial wastes or by-products of fermentation media are utilized to improve the cost-effectiveness such as candied jujube waste water,138 acetone-butanol-ethanol fermentation,139 and pomegranate extract. Fig. 7 presented the semi-interpenetrating network of hydrogels based on fabricating BC and chitosan through glutaraldehyde linkage.140
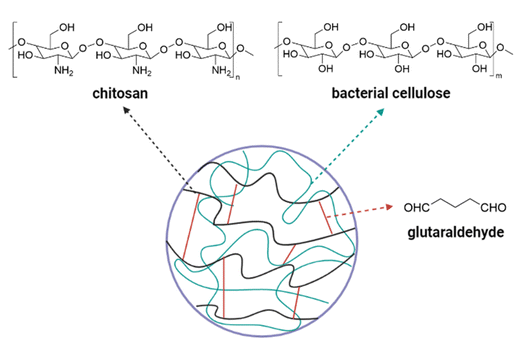 | ||
| Fig. 7 Simple illustrated structure of semi-interpenetrating network.140 | ||
4.2 Collagen
Collagen is an abundant natural polymer found in the ECM, accounting for one-third of the protein in the body. Collagen exists mainly in epithelial and connective tissues such as bone, cartilage, ligaments, tendon, skin and is an essential component in cell interactions, regulation of cell anchoring, and cell migration.141,142 Cooperative with glycosaminoglycan, collagen has been an essential factor for cell attachment, proliferation, and differentiation.143,144 The high flexibility of collagen dressings is due to the three-dimensional structure, which can absorb liquids many times their weight thanks to the accessible surface and high capillarity.145 Collagen dressings reduce the risk of secondary bacterial invasion by acting as chemoattractant for neutrophils,146 and affect other healing processes such as decreasing protease activity.147 However, the disadvantages of collagen based-biomaterials are their rapid degradation rate and low stability, resulting in a significant loss of mechanical properties.1484.3 Chitosan
Chitosan (CS) is obtained by chitin deacetylation and extracted from the extracellular matrix of marine crustaceans, shrimp, crabs, shellfish, and some fungi.149,150 CS is a linear polysaccharide made up of D-glucosamine and N-acetyl-D-glucosamine units. CS is a popular and renewable natural biomaterial for regenerative medicine with many valuable properties such as compatibility and biodegradability, inertness, allergenic, antibacterial, and hemostatic effects.151,152 It is also applied to treat wounds, especially chronic wounds, infections, or ulcers.151,1534.4 Hyaluronic acid
Hyaluronic acid (HA) is a natural polymer found in skin, lens, or synovial fluids, composed of D-glucuronic acid and N-acetyl-D-glucosamine disaccharide. HA is characterized by high viscosity and fast degradation rate.HA is widely used in medical fields such as orthopedics and plastic surgery because its characteristics involve several structural properties of tissues, cell signaling, and critical elements of ECM.154,155 Additionally, HA plays a role in healing acute and chronic wounds by promoting early inflammation, increasing cellular infiltration, enhancing granulation tissue formation, and facilitating cell migration.156–158 HA also performs good swelling behavior due to the presence of the carboxyl group in the side chains and has a thick wall structure to support the strength of the material.159 The swelling–reducing properties and stable structure may prove that HA can be a potential material for dressings. However, HA is sensitive to the molecular weight factor. According to Campo et al., only medium molecular weight HA could enhance wound repair, while low molecular weights contributed to further inflammation, and high molecular weights may exert inflammatory pathologies such as rheumatoid arthritis.160
4.5 Fibrinogen and fibrin
Fibrin and fibrinogen participate in various physiological functions such as fibrinolysis, cellular and matrix interactions, and the inflammatory response.161 In the wound healing process, fibrin and fibrinogen roles are expressed through mediating both hemostasis and homeostasis.162 Fibrinogen molecules are made up of two sets of three different peptide chains including Aα, Bβ and γ chains, and connected by disulfide bridges.163 The formation of fibrin releases small peptides and is catalyzed by thrombin.161,164 Fibrin is highly extensible and has elasticity properties.165 The scaffolds made of fibrin or fibrinogen can attain a high cell seeding efficiency and then proliferate, migrate, and differentiate into specific tissues/organs by secreting ECM.166 Fibrinogen provides a surface for cellular attachment and proliferation and consists of a fibrous network for cell signaling and cell-matrix167 which is a major advantage for healing. However, these proteins are easily degraded and have poor mechanical properties.1684.6 Polylysine
Polylysine is a cationic polymer synthesized through condensation polymerization or fermentation of amino acid lysine. Lysine is available in two chiral forms which are L-lysine and D-lysine resulting in α-polylysine or ε-polylysine.169 ε-Poly-L-lysine is a naturally occurring polymer and presents various characteristics: water solubility,170 biodegradability, non-toxic for drug delivery systems, biological adhesives,171 antibacterial ability.169,172 Because of its the biodegradability and biocompatibility characteristics, polylysine has raised the attention of medical applications.169,173 However, due to the limitation in mechanical properties, it should be modified or blended with other polymers to improve the mechanical strength.169All studies on dressings which are natural-based polymer for wound healing applications are summarized in Table 2.
| Material | Name of dressing | Trial | Effectiveness | References |
|---|---|---|---|---|
| a AgNPs: silver nanoparticles, RPC: pH responsive cellulose, PB: poly(vinyl alcohol)/borax, CMC: carboxymethyl cellulose, BC: bacterial cellulose, CS: chitosan, PVA: poly(vinyl alcohol), PTE: Poria cocos triterpenes extract, NEs: nanoemulsions, HA: hyaluronic acid, BNN6: N,N′-disecbutyl-N,N′-dinitroso-p-phenylenediamine, εPL: ε-polylysine. | ||||
| Cellulose | Cellulose nanocrystals and AgNPs | In vivo, in vivo | No toxic effects of the combination of cellulose and AgNPs | 174 |
| Promoting rapid wound healing compared to control groups | ||||
| RPC/PB hydrogel | In vitro, in vivo | Exhibiting excellent antibacterial, skin tissue regeneration and wound closure capabilities | 175 | |
| Na CMC with merremia mammosa gel | In vivo | Not irritable, accelerating healing process through increasing collagen synthesis and angiogenesis | 176 | |
| Oxidized regenerated cellulose membrane | Clinical trials | Stop bleeding in patients with uncontrollable bleeading | 177 | |
| Bacterial cellulose | BC reinforced chitosan-based hydrogel | In vitro | Showing good biocompability and excellent antibacterial activity against E. coli and S. aureus | 178 |
| Dialdehyde carboxymethyl BC/CS composites | In vivo | Accelerating the wound healing rate and inhibit bacterial proliferation | 179 | |
| BC membrane | In vivo | Good biocompability and prevent fibrosis in trabeculectomy | 180 | |
| BC gel and associated film | Clinical trials | Decreasing significantly in the size of wound, lower dressing change frequency compared to group using Rayon® | 181 | |
| BC dressing | Clinical trials | Shorter healing time in managing second-degree burn wounds and skin graft donor sites compared to vaseline gauzes | 182 | |
| Collagen | Modified collagen gel | In vitro | Enhancing macrophage attraction to the wound site, reducing proinflammatory virulence, promoting anti-inflammatory macrophage polarization, addressing wound inflammation, and improving angiogenesis | 183 |
| Collagen-based composite dressing | Clinical trial | Forming granulation tissue, enhancing epithelialization, and having faster wound healing time | 184 and 185 | |
| Chitosan | CS-based opticell dressing | In vivo | The total bleeding significantly decreased in excisional wounds mimicking debridement | 186 |
| HemCon® dental dressing | Clinical trial | Pain values and post-extraction socket healing were lower after suture removal on treating anti-platelet patients | 187 | |
| Chitosan dressing | Clinical trial | Reducing wound size and wound depth on chronic, difficult-to-heal wounds such as diabetic ulcers, leg vein ulcers | 188 | |
| Hyaluronic acid | Incorporation of PVA/HA/cellulose nanocrystals as nanofiber | In vitro | Loading with L-arginine exhibited excellent proliferative and adhesive potential, high wound gap-closure, and showed antibacterial activity against Klebsiella pneumonia | 189 |
| 0.2% HA | In vivo | Healing skin abrasions in rat's model | 190 | |
| PTE-NEs fabricated HA hydrogel | In vitro, in vivo | No toxicity, improve the wound healing through reducing inflamation, enhancing collagen synthesis, accelerating M2 macrophage polarization, and angiogenesis | 191 | |
| 0.2% and 0.8% HA gel | Clinical trial | Complete epithelization. Pain and burning sensation scales were also lower. Color match scores were higher | 192 | |
| Healoderm | Clinical trial | The diabetic foot ulcer group had a higher complete healing rate, faster ulcer healing velocity, and shorter mean duration for achieving a 50% ulcer size reduction | 193 | |
| Fibrinogen and fibrin | Fibrin combined with Na carboxymethylcellulose | In vitro | In the form of a mesh, supporting the fibroblast adhesion and proliferation, accelerating the wound healing | 194 |
| Fibrin-based hydrogel load BNN6 mesoporous polydopamine nanoparticles | In vitro, in vivo | Clearing the infection of methicillin-resistant S. aureus through cell membrane and genetic metabolism damage under 808 nm laser irridation. Accelerating wound healing through collagen deposition and the proliferation of hair follicles | 195 | |
| 3D salmon fibrinogen and chitosan scaffold | In vitro, in vivo | The cell proliferate in the scaffold and the wound healing is more effective than the untreated group | 196 | |
| Alginate-fibrinogen-nisin hydrogel | In vitro, in vivo | Inhibiting the bacteria growth, accelerate the formation of blood clot, show the higher rates of wound healing, re-epithelialization, and collagen deposition | 197 | |
| Heterologous fibrin sealant | Clinical trial | Heterologous fibrin sealant is safe and non-immunogenic, showing good preliminary efficacy in chronic venous ulcers treatment | 198 | |
| Polylysine | Gelatin nanofiber dressing contains εPL | In vitro, in vivo | Eliciting bactericidal activity in burn wounds for fibroblasts migration and re-epithelialization. In partial thickness burns of porcine model, promoting wound closure and reduce hypertrophic scarring | 199 |
| Carbon dots and εPL hydrogel | In vitro, in vivo | Having broad spectrum in antibacterial activity. Enhancing angiogenesis and epithelization that accelerate the wound healing rate | 200 | |
| Modified HA/εPL hydrogel | In vitro, in vivo | Killing bacteria in infected wound and improving the wound status in rat model | 201 | |
| εPL modified natural silk fiber membrane | In vivo | Exhibiting thicker granulation tissue, higher collagen composition, help accelerate wound healing rate | 202 | |
5. Discussion
Using wound dressing for wound care and treatment is an effective method, proven by the positive results from numerous experiments. Modern dressings are prioritized for research and development.31,203 Modern dressings ensure the essential factor of creating a moist environment for wound healing, pain reduction, and antibacterial. However, the effectiveness of these popular modern dressings is still limited to only a few clinical studies. Wound dressing efficacy trials are usually performed on flat wounds, with less complexity, creating an oversimplification of the actual clinical features.204On the other hand, a wound is considered a complex clinical problem, and the effectiveness of treatment depends on several factors: diagnosis, patient comorbidities, anatomical location, physiological status, and wound size.203,204 Thus, the general recommendation is that dressing selection should be tailored to the wound and patient, under the guidance and consideration of physicians with expertise in wound treatment.112,205,206 That would help the patients receive the proper treatment with the right purpose, achieve optimal efficiency and limit unnecessary risks during treatment.
Table 3 shows the specific advantages, disadvantages, and application range of modern dressings for different wounds, which provide a clinical guideline for selecting suitable wound dressings for effective wound healing. In addition, it is still necessary to have more research and clinical trials to demonstrate the effectiveness and safety of various types of modern wound dressings in the future.
| Dressing type | Advantages | Disadvantages | Application |
|---|---|---|---|
| Hydrocolloids | Self-adhesive, no need for extra tape | The gel formed can be thick, yellow, smelly, and easy to mistake for an infection | Low to moderate exudate wounds. Scratch, post-surgery wounds, pressure ulcers, shallow leg ulcers |
| Creating a light layer of padding | Not suitable for exudative wounds | ||
| Moisturizing | |||
| Painless during removing the dressing | |||
| Easy to use | |||
| Alginates | High absorbency | Not suitable for dry wounds | Pressure ulcers, fluid lower-extremity ulcers, infected wounds |
| Hemostasis | Sticking to granulation tissue easily if not changed frequently | ||
| Highly biocompatible | |||
| Hydrogels | Cools, soothe wounds, and relieve pain | Not suitable for fluid oozing wounds | Burns, especially partial burns, foot ulcers |
| Changing the physicochemical properties, forming reactive materials, responding to changes in temperature, pH, and drug release | Requiring tape for fixation | ||
| Foams | Absorbing and transmitting moisture | Effecting on new tissue growth ability and causing injury when removing the dressing if kept for too long | Burns, chronic wounds, deep ulcers, wounds in exudate cavities |
| Wound cushion | |||
| Not necessary to change as often, depending on the amount of discharge | |||
| Films | Thin and elastic, easy to pull, shape to the wound | Easy to fold and stick | Burns, wounds of joints, skin grafts with small thickness, superficial lacerations |
| Transparent, easy to monitor wound condition | Poor absorbing, only suitable for wounds with little secretion | ||
| Impervious to microorganisms | No antibacterial properties |
Developing new material platforms for modern dressings with positive results demonstrated through various tests has led to an innovation in improving the dressing products on the market. The current trend of creating ideal dressing products has been to combine materials with outstanding advantages needed for the wound healing process such as intrinsic antibacterial properties (AgNPs, ZnO), high biological compatibility, being environmentally friendly, and being easy to handle, particularly natural polymer materials. Novel materials have been developed to respond to the different conditions and stages of wound healing. Moreover, wound dressings are loaded with bioactive components including antibiotics, growth factors, herbal extracts, essential oil, antioxidants, anti-inflammatory agents, and vitamins, to improve therapeutic outcomes or to overcome the limitation of the dressing. Simultaneously, wound dressings components play an essential role as drug delivery systems.207 So far, incorporating bioactive agents into the structure of the dressing may considerably enhance its biocompatibility and qualities, resulting in a significant healing process. However, this modification might have a detrimental impact on the dressing's ability to absorb exudate and its mechanical characteristics.208 Thus, there is still a necessity for research to minimize negative effects that may result from modifying materials with bioactive components, for instance loss of mechanical properties or decreased cell proliferation.
Nevertheless, a variety of new materials for modern wound dressings not only creates synergistic effects from the strengths of each material but also causes difficulties in complex synthesis processes, requiring many advanced techniques, limiting the popular, cost-effective, if scaled-up production. Therefore, these new materials platforms have been not yet widely applied in clinical practice. Thus, the future tasks are to focus on developing new materials and to find solutions that simplify the material synthesis process, aiming to develop potential materials into commercial products serving social needs. In addition, future research on wound dressing production should not only stop enhancing healing treatment but also ensure aesthetic requirements after treatment and limit scar tissue formation.
6. Conclusions
Modern dressing has become a priority selection for wound care and treatment due to its advancements in promoting healing wound. With the rapid development of modern technology, more and more new potential biomaterials have been created and have been intensely exploited for wound healing applications. In this context, recent advances in the development of natural-based materials as well as the applications of modern wound dressings are presented. However, the limitations of new modern dressings are the complicated production process, the lack of quality assurance for biological materials, as well as the effectiveness of the component materials for widespread use. Thus, material scientists should have more trials and experiment to determine the actual effectiveness of new modern dressings in wound healing.Conflicts of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.Acknowledgements
We acknowledge the support of time and facilities from Ho Chi Minh City University of Technology (HCMUT) for this study.Notes and references
- K. Jung, S. Covington, C. K. Sen, M. Januszyk, R. S. Kirsner, G. C. Gurtner and N. H. Shah, Wound Repair Regen., 2016, 24, 181–188 CrossRef PubMed.
- E. M. Tottoli, R. Dorati, I. Genta, E. Chiesa, S. Pisani and B. Conti, Pharmaceutics, 2020, 12, 735 CrossRef CAS PubMed.
- J. F. Guest, G. W. Fuller and P. Vowden, BMJ Open, 2020, 10, e045253 CrossRef PubMed.
- Y. Hu, H. Li, X. Lv, Y. Xu, Y. Xie, L. Yuwen, Y. Song, S. Li, J. Shao and D. Yang, Nanoscale, 2022, 14, 12967–12983 RSC.
- C. E. Fife and M. J. Carter, Wounds, 2012, 24, 10–17 Search PubMed.
- S. R. Nussbaum, M. J. Carter, C. E. Fife, J. DaVanzo, R. Haught, M. Nusgart and D. Cartwright, Value Health, 2018, 21, 27–32 CrossRef PubMed.
- L. Gould, P. Abadir, H. Brem, M. Carter, T. Conner-Kerr, J. Davidson, L. DiPietro, V. Falanga, C. Fife, S. Gardner, E. Grice, J. Harmon, W. R. Hazzard, K. P. High, P. Houghton, N. Jacobson, R. S. Kirsner, E. J. Kovacs, D. Margolis, F. McFarland Horne, M. J. Reed, D. H. Sullivan, S. Thom, M. Tomic-Canic, J. Walston, J. Whitney, J. Williams, S. Zieman and K. Schmader, J. Am. Geriatr. Soc., 2015, 23, 1–13 Search PubMed.
- GlobeNewswire, Advanced Wound Care Market worth over USD 13 billion by 2024, Global Market Insights, Inc., https://www.globenewswire.com/fr/news-release/2018/09/17/1571505/0/en/Advanced-Wound-Care-Market-worth-over-USD-13-billion-by-2024-Global-Market-Insights-Inc.html, accessed 23 Nov 2022 Search PubMed.
- K. Heyer, M. Augustin, K. Protz, K. Herberger, C. Spehr and S. J. Rustenbach, Dermatology, 2013, 226, 172–184 CrossRef CAS PubMed.
- S. H. Aswathy, U. Narendrakumar and I. Manjubala, Heliyon, 2020, 6, e03719 CrossRef CAS PubMed.
- L. Sadati, R. Froozesh, A. Beyrami, Z. N. Khaneghah, S. A. Elahi, M. F. Asl and T. Salehi, Adv. Skin Wound Care, 2019, 32, 1–5 CrossRef PubMed.
- H.-R. Metelmann, T. v. Woedtke and K.-D. Weltmann, Comprehensive Clinical Plasma Medicine: Cold Physical Plasma for Medical Application, Springer Cham, 1st edn, 2018 Search PubMed.
- T. Velnar, T. Bailey and V. Smrkolj, J. Int. Med. Res., 2009, 37, 1528–1542 CrossRef CAS PubMed.
- M. Phillipson and P. Kubes, Trends Immunol., 2019, 40, 635–647 CrossRef CAS PubMed.
- M. Rodrigues, N. Kosaric, C. A. Bonham and G. C. Gurtner, Physiol. Rev., 2018, 99, 665–706 CrossRef PubMed.
- G. Younan, F. Suber, W. Xing, T. Shi, Y. Kunori, M. Åbrink, G. Pejler, S. M. Schlenner, H.-R. Rodewald, F. D. Moore Jr, R. L. Stevens, R. Adachi, K. F. Austen and M. F. Gurish, J. Immunol., 2010, 185, 7681–7690 CrossRef CAS PubMed.
- S. Werner, T. Krieg and H. Smola, J. Invest. Dermatol., 2007, 127, 998–1008 CrossRef CAS PubMed.
- J. J. Tomasek, G. Gabbiani, B. Hinz, C. Chaponnier and R. A. Brown, Nat. Rev. Mol. Cell Biol., 2002, 3, 349–363 CrossRef CAS PubMed.
- J. M. Reinke and H. Sorg, Eur. Surg. Res., 2012, 49, 35–43 CrossRef CAS PubMed.
- K. R. Jones, K. Fennie and A. Lenihan, Wounds, 2007, 19, 51–63 Search PubMed.
- D. T. Robles and D. Berg, Clin. Dermatol., 2007, 25, 26–32 CrossRef PubMed.
- R. G. Frykberg and J. Banks, Adv. Wound Care, 2015, 4, 560–582 CrossRef PubMed.
- S. Guo and L. A. DiPietro, J. Dent. Res., 2010, 89, 219–229 CrossRef CAS PubMed.
- W. Y. J. Chen and A. A. Rogers, Wound Repair Regen., 2007, 15, 434–449 CrossRef PubMed.
- S. Khanna, S. Biswas, Y. Shang, E. Collard, A. Azad, C. Kauh, V. Bhasker, G. M. Gordillo, C. K. Sen and S. Roy, Plos One, 2010, 5, e9539 CrossRef PubMed.
- C. Wetzler, H. Kämpfer, B. Stallmeyer, J. Pfeilschifter and S. Frank, J. Invest. Dermatol., 2000, 115, 245–253 CrossRef CAS PubMed.
- G. D. Winter, Nature, 1962, 193, 293–294 CrossRef CAS PubMed.
- C. D. Hinman and H. Maibach, Nature, 1963, 200, 377–378 CrossRef CAS PubMed.
- A. Flett, F. Russell, S. Stringfellow, P. J. Cooper, D. G. Gray and S. Lawton, Nurs. Resid. Care, 2002, 4, 328–344 CrossRef.
- L. Demarre, S. Verhaeghe, A. Van Hecke, E. Clays, M. Grypdonck and D. Beeckman, J. Adv. Nurs., 2015, 71, 391–403 CrossRef PubMed.
- J. S. Boateng, K. H. Matthews, H. N. E. Stevens and G. M. Eccleston, J. Pharm. Sci., 2008, 97, 2892–2923 CrossRef CAS PubMed.
- L. J. Borda, F. E. Macquhae and R. S. Kirsner, Curr. Dermatol. Rep., 2016, 5, 287–297 CrossRef.
- G. D. Mogoşanu and A. M. Grumezescu, Int. J. Pharm., 2014, 463, 127–136 CrossRef PubMed.
- S. Dhivya, V. V. Padma and E. Santhini, BioMedicine, 2015, 5, 22 CrossRef PubMed.
- K. L. Andrews, K. M. Derby, T. M. Jacobson, B. A. Sievers and L. J. Kiemele, in Braddom's Physical Medicine and Rehabilitation, ed. D. X. Cifu, Elsevier, Philadelphia, 6th edn, 2021, ch. 24, pp. 469–484 Search PubMed.
- S. Rahmani and D. J. Mooney, in The Diabetic Foot: Medical and Surgical Management, ed. A. Veves, J. M. Giurini and R. J. Guzman, Humana Press, Totowa, New Jersey, 4th edn, 2018, ch. 15, pp. 247–256 Search PubMed.
- C. M. Wietlisbach, in Cooper's Fundamentals of Hand Therapy, ed. C. M. Wietlisbach, Mosby, St. Louis (MO), 3rd edn, 2020, ch. 17, pp. 154–166 Search PubMed.
- A. Agarwal, J. F. McAnulty, M. J. Schurr, C. J. Murphy and N. L. Abbott, in Advanced Wound Repair Therapies, ed. D. Farrar, Woodhead Publishing, 2011, pp. 186–208 Search PubMed.
- F. L. Bowling, S. T. Rashid and A. J. M. Boulton, Nat. Rev. Endocrinol., 2015, 11, 606–616 CrossRef PubMed.
- N. M. Aruan, I. Sriyanti, D. Edikresnha, T. Suciati, M. M. Munir and K. Khairurrijal, Procedia Eng., 2017, 170, 31–35 CrossRef CAS.
- A. Janowska, M. Macchia and B. Paggi, in Science and Practice of Pressure Ulcer Management, ed. M. Romanelli, M. Clark, A. Gefen and G. Ciprandi, Spinger London, London, 2nd edn, 2018, ch. 12, pp. 159–173 Search PubMed.
- S. Thomas, Int. Wound J., 2008, 5, 602–613 CrossRef PubMed.
- J. Z. M. Lim, N. S. L. Ng and C. Thomas, J. R. Soc. Med., 2017, 110, 104–109 CrossRef PubMed.
- S. G. Jin, A. M. Yousaf, S. W. Jang, M.-W. Son, K. S. Kim, D.-W. Kim, D. X. Li, J. O. Kim, C. S. Yong and H.-G. Choi, Drug Dev. Res., 2015, 76, 157–165 CrossRef CAS PubMed.
- T. Takeuchi, M. Ito, S. Yamaguchi, S. Watanabe, M. Honda, T. Imahashi, T. Yamada and T. Kokubo, Nagoya J. Med. Sci., 2020, 82, 487–498 CAS.
- K.-Y. Sung and S.-Y. Lee, Wounds, 2016, 28, 145–151 Search PubMed.
- J. Chen, J. Chen, J. Yang, Y. Chen, Y. Liang and Y. Lin, Pediatr. Crit. Care Med., 2020, 21, e752–e758 CrossRef PubMed.
- N. Shinohara, N. Nonomura, M. Eto, G. Kimura, H. Minami, S. Tokunaga and S. Naito, Ann. Oncol., 2014, 25, 472–476 CrossRef CAS PubMed.
- C. Sabando, W. Ide, M. Rodríguez-Díaz, G. Cabrera-Barjas, J. Castaño, R. Bouza, N. Müller, C. Gutiérrez, L. Barral, J. Rojas, F. Martínez and S. Rodríguez-Llamazares, Curr. Top. Med. Chem., 2020, 20, 280–292 CrossRef CAS PubMed.
- Convatec, DuoDERM® Dressings, https://www.convatec.com/advanced-wound-care/duoderm-dressings/, accessed Nov 23, 2022 Search PubMed.
- Coloplast, Comfeel®Plus, https://www.coloplast.com/products/wound/comfeel-plus/, accessed Nov 23, 2022 Search PubMed.
- Smith-Nephew, Cutinova®Hydro, https://www.smith-nephew.com/professional/products/advanced-wound-management/other-wound-care-products/cutinova-hydro/, accessed Nov 23, 2022 Search PubMed.
- Smith-Nephew, Replicare®, https://www.smith-nephew.com/professional/products/advanced-wound-management/replicare/replicare/, accessed Nov 23, 2022 Search PubMed.
- Convatec, Kaltostat®, https://www.convatec.com/en-au/products/pc-wound-diabetic-foot-ulcers/kaltostat-alginate-calcium-sodium-dressing, accessed Nov 23, 2022 Search PubMed.
- DermaSciences, Algicell®Ag, https://www.integralife.com/algicell-ag-calcium-alginate-dressing-with-antimicrobial-silver/product/wound-reconstruction-care-outpatient-clinic-private-office-supportive-therapies-algicell-ag-calcium-alginate-dressing-with-antimicrobial-silver, accessed Nov 23, 2022 Search PubMed.
- S. G. Kim, K. Y. Song, H. H. Lee, E. Y. Kim, J. H. Lee, H. M. Jeon, K. H. Jeon, H. M. Jin, D. J. Kim, W. Kim, H. M. Yoo, J. G. Kim and C. H. Park, Medicine, 2019, 98, e15141 CrossRef CAS PubMed.
- A. Medical, Activon® – Manuka Honey, https://uk.advancismedical.com/products/activon-manuka-honey/algivon, accessed Nov 23, 2022 Search PubMed.
- 3M™, Fibracol™ Plus Collagen Wound Dressing with Alginate, https://www.3m.com/3M/en_US/p/d/b5005265077/, accessed Nov 23, 2022 Search PubMed.
- M. K. Kucharska, M. H. Struszczyk, A. Niekraszewicz, D. Ciechańska, E. Witczak, S. Tarkowska, K. Fortuniak, A. Gulbas-Diaz, A. Rogaczewska, I. Płoszaj, A. Pluta and T. Gąsiorowski, Prog. Chem. Appl. Chitin Deriv., 2011, 16, 121–130 Search PubMed.
- Tricomed, Tromboguard®, https://tricomed.com/products/tromboguard/, accessed Jun 26, 2022 Search PubMed.
- Activheal, Activheal® hydrogel is an effective method for hydrating dry necrotic and sloughy wounds, https://activheal.com/wound-care-dressing-range/hydrogel-dressing/, accessed Nov 23, 2022 Search PubMed.
- H. Woundcare, Restore Hydrogel Dressings, https://www.hollister.com/-/media/files/pdfs-for-download/wound-care/restore-hydrogel-techsheet-911140-1110.ashx, accessed Nov 23, 2022 Search PubMed.
- L. R. Globa, Suprasorb® G Gel Wound Dressing, https://www.lohmann-rauscher.com/au-en/produkte-alt/wound-care/modern-wound-care/suprasorb-g-gel-dressing/, accessed Nov 23, 2022 Search PubMed.
- DermaRite, AquaDerm™ Hydrogel Sheet Dressing, https://dermarite.com/product/aquaderm/, accessed Nov 23, 2022 Search PubMed.
- Kikgel, Neoheal®Hydrogel dressing for wound management, https://kikgel.com.pl/en/products/neoheal/, accessed Nov 23, 2022 Search PubMed.
- Smith-Nephew, Allevyn Life, https://www.smith-nephew.com/professional/products/advanced-wound-management/allevyn/allevyn-life1/, accessed Nov 23, 2022 Search PubMed.
- Hartmann, PermaFoam® Classic, https://www.hartmann.info/en-dx/products/wound-management/hydroactive-wound-dressings/foam-wound-dressings/permafoam%C2%AE-classic, accessed Nov 23, 2022 Search PubMed.
- Hartmann, HydroTac®, https://www.hartmann.info/en-dx/products/wound-management/hydroactive-wound-dressings/foam-wound-dressings/hydrotac%C2%AE#products, accessed Nov 23, 2022 Search PubMed.
- Mölnlycke, Mepilex Ag, https://www.molnlycke.ae/products-solutions/mepilex-ag/, accessed Feb 2, 2023 Search PubMed.
- 3M™, Tegaderm™ Silicone Foam Dressings, https://www.3m.com/3M/en_US/medical-us/tegaderm-silicone-foam-dressings/, accessed Nov 23, 2022 Search PubMed.
- 3M™, Bioclusive™ Plus Transparent Film Dressing, https://www.acelity.com/healthcare-professionals/global-product-catalog/catalog/bioclusive-plus-dressing, accessed Nov 23, 2022 Search PubMed.
- Mölnlycke, Mepitel, https://www.molnlycke.com/products-solutions/mepitel/, accessed Nov 23, 2022 Search PubMed.
- Mölnlycke, Mepore® Film, https://www.molnlycke.com/products-solutions/mepore-film/, accessed Nov 23, 2022 Search PubMed.
- DeRoyal, Transeal®, https://www.deroyal.com/products/search-catalog-item/catalog-item-preview/wc-burn-transeal, accessed Nov 23, 2022 Search PubMed.
- E. A. Kamoun, E.-R. S. Kenawy and X. Chen, J. Adv. Res., 2017, 8, 217–233 CrossRef CAS PubMed.
- M. A. Taemeh, A. Shiravandi, M. A. Korayem and H. Daemi, Carbohydr. Polym., 2020, 228, 115419 CrossRef CAS PubMed.
- P. N. Sudha, S. Aisverya, R. Nithya and K. Vijayalakshmi, in Advances in Food and Nutrition Research, ed. S.-K. Kim, Academic Press, 2014, ch. 8, vol. 73, pp. 145–181 Search PubMed.
- R. Mahsood and M. Miraftab, J. Wound Care, 2014, 23, 153–159 CrossRef PubMed.
- J. C. Dumville, S. J. Keogh, Z. Liu, N. Stubbs, R. M. Walker and M. Fortnam, Cochrane Database Syst. Rev., 2015, CD011277 Search PubMed.
- B. A. Aderibigbe and B. Buyana, Pharmaceutics, 2018, 10, 42 CrossRef PubMed.
- Helmiyati and M. Aprilliza, Presented in part at the IOP Conference Series: Materials Science and Engineering, Bali, Indonesia, Jul 26–27, 2016 Search PubMed.
- M. Matyash, F. Despang, C. Ikonomidou and M. Gelinsky, Tissue Eng., Part C, 2013, 20, 401–411 CrossRef PubMed.
- S. K. Bajpai and N. Kirar, Des. Monomers Polym., 2016, 19, 89–98 CrossRef CAS.
- I. R. Sweeney, M. Miraftab and G. Collyer, Int. Wound J., 2012, 9, 601–612 CrossRef PubMed.
- A. Sood, M. S. Granick and N. L. Tomaselli, Adv. Wound Care, 2013, 3, 511–529 CrossRef PubMed.
- C. Weller, C. Weller and V. Team, in Advanced Textiles for Wound Care ed. S. Rajendran, Woodhead Publishing, 2nd edn, 2019, ch. 4, pp. 105–134 Search PubMed.
- S. McCarthy, V. Dvorakova, P. O'Sullivan and J. F. Bourke, Contact Dermatitis, 2018, 79, 396–397 CrossRef PubMed.
- M. Fischer, F. Gebhard, T. Hammer, C. Zurek, G. Meurer, C. Marquardt and D. Hoefer, J. Biomater. Appl., 2017, 31, 1267–1276 CrossRef CAS PubMed.
- A. Dodero, S. Scarfi, M. Pozzolini, S. Vicini, M. Alloisio and M. Castellano, ACS Appl. Mater. Interfaces, 2020, 12, 3371–3381 CrossRef CAS PubMed.
- Y. Fan, W. Wu, Y. Lei, C. Gaucher, S. Pei, J. Zhang and X. Xia, Mar. Drugs, 2019, 17, 285 CrossRef CAS PubMed.
- S. Shafei, M. Khanmohammadi, R. Heidari, H. Ghanbari, V. Taghdiri Nooshabadi, S. Farzamfar, M. Akbariqomi, N. S. Sanikhani, M. Absalan and G. Tavoosidana, J. Biomed. Mater. Res. A, 2020, 108, 545–556 CrossRef CAS PubMed.
- M. Contardi, A. M. d. M. d. Ayyoub, M. Summa, D. Kossyvaki, M. Fadda, N. Liessi, A. Armirotti, D. Fragouli, R. Bertorelli and A. Athanassiou, ACS Appl. Bio Mater., 2022, 5, 2880–2893 CrossRef CAS PubMed.
- H. Lu, J. A. Butler, N. S. Britten, P. D. Venkatraman and S. S. Rahatekar, Nanomaterials, 2021, 11, 2062 CrossRef CAS PubMed.
- S. O'Meara, M. Martyn-St James and U. J. Adderley, Cochrane Database Syst. Rev., 2015, CD010182 Search PubMed.
- G. Huang, T. Sun, L. Zhang, Q. Wu, K. Zhang, Q. Tian and R. Huo, Exp. Ther. Med., 2014, 7, 1772–1776 CrossRef CAS PubMed.
- L. Devi and P. Gaba, J. Crit. Rev., 2019, 6, 1–10 Search PubMed.
- D. Zeng, S. Shen and D. Fan, Chin. J. Chem. Eng., 2021, 30, 308–320 CrossRef CAS.
- Y. Liang, J. He and B. Guo, ACS Nano, 2021, 15, 12687–12722 CrossRef CAS PubMed.
- I. Firlar, M. Altunbek, C. McCarthy, M. Ramalingam and G. Camci-Unal, Gels, 2022, 8, 127 CrossRef CAS PubMed.
- H. Chen, R. Cheng, X. Zhao, Y. Zhang, A. Tam, Y. Yan, H. Shen, Y. S. Zhang, J. Qi, Y. Feng, L. Liu, G. Pan, W. Cui and L. Deng, NPG Asia Mater., 2019, 11, 3 CrossRef CAS.
- L. L. M. Jose, R. M. Leocadio, L. Olga, B. P. Miriam, C. Dios-Guerra and L. M. Silvia, J. Wound Care, 2020, 29, 202 Search PubMed.
- M. D. Holbert, B. R. Griffin, S. M. McPhail, R. S. Ware, K. Foster, D. C. Bertoni and R. M. Kimble, Trials, 2019, 20, 13 CrossRef PubMed.
- S. Niansheng, C. Binghuan, W. Cheng, G. Hua, B. Xu, Z. Ran and H. Ran, Zhonghua Shao Shang Za Zhi, 2021, 37, 1085–1089 Search PubMed.
- L. Zhang, H. Yin, X. Lei, J. N. Y. Lau, M. Yuan, X. Wang, F. Zhang, F. Zhou, S. Qi, B. Shu and J. Wu, Front. Bioeng. Biotechnol., 2019, 7, 342 CrossRef PubMed.
- K. A. Deo, G. Lokhande and A. K. Gaharwar, in Encyclopedia of Tissue Engineering and Regenerative Medicine, ed. R. L. Reis, Academic Press, Oxford, 2019, pp. 21–32 Search PubMed.
- E. Yahia, A. El-Sharkawey and M. Bayoumi, Pakistan J. Medical Health Sci., 2021, 15, 1571–1574 CrossRef.
- L. Han, X. Lu, K. Liu, K. Wang, L. Fang, L.-T. Weng, H. Zhang, Y. Tang, F. Ren, C. Zhao, G. Sun, R. Liang and Z. Li, ACS Nano, 2017, 11, 2561–2574 CrossRef CAS PubMed.
- A. Tamayol, M. Akbari, Y. Zilberman, M. Comotto, E. Lesha, L. Serex, S. Bagherifard, Y. Chen, G. Fu, S. K. Ameri, W. Ruan, E. L. Miller, M. R. Dokmeci, S. Sonkusale and A. Khademhosseini, Adv. Healthcare Mater., 2016, 5, 711–719 CrossRef CAS PubMed.
- C. Occhiuzzi, A. Ajovalasit, M. A. Sabatino, C. Dispenza and G. Marrocco, 2015.
- J. Nielsen and K. Fogh, Chronic Wound Care Manag. Res., 2015, 2, 31–38 Search PubMed.
- A.-G. Niculescu and A. M. Grumezescu, Polymers, 2022, 14, 421 CrossRef CAS PubMed.
- C. D. Weller, V. Team and G. Sussman, Front. Pharmacol., 2020, 11, 155 CrossRef CAS PubMed.
- S. M. Lee, I. K. Park, Y. S. Kim, H. J. Kim, H. Moon, S. Mueller and Y.-I. L. Jeong, Biomater. Res., 2016, 20, 15 CrossRef PubMed.
- J.-A. Jung, K.-H. Yoo, S.-K. Han, E.-S. Dhong and W.-K. Kim, Adv. Skin Wound Care, 2016, 29, 546–555 CrossRef PubMed.
- O. M. Alvarez, M. S. Granick, A. Reyzelman and T. Serena, J. Comp. Eff. Res., 2021, 10, 481–493 CrossRef PubMed.
- P. Davies, S. McCarty and K. Hamberg, J. Wound Care, 2017, 26, S1–S32 CrossRef PubMed.
- N. Namviriyachote, V. Lipipun, Y. Akkhawattanangkul, P. Charoonrut and G. C. Ritthidej, Asian J. Pharm. Sci., 2019, 14, 63–77 CrossRef PubMed.
- H. C. Gwak, S. H. Han, J. Lee, S. Park, K.-S. Sung, H.-J. Kim, D. Chun, K. Lee, J.-H. Ahn, K. Kwak and H.-J. Chung, Int. Wound J., 2020, 17, 91–99 CrossRef PubMed.
- K. Vowden and P. Vowden, Surgery, 2017, 35, 489–494 Search PubMed.
- R. A. A. Dutra, G. M. Salomé, J. R. Alves, V. O. S. Pereira, F. D. Miranda, V. B. Vallim, M. J. A. de Brito and L. M. Ferreira, J. Wound Care, 2015, 24, 268–275 CrossRef CAS PubMed.
- A. Jafari, S. Hassanajili, M. B. Karimi, A. Emami, F. Ghaffari and N. Azarpira, J. Mech. Behav. Biomed. Mater., 2018, 88, 395–405 CrossRef CAS PubMed.
- M. Li, J. Chen, M. Shi, H. Zhang, P. X. Ma and B. Guo, Chem. Eng. J., 2019, 375, 121999 CrossRef CAS.
- F. Gulmez, A. Yercan, B. Kocaaga and F. S. Guner, J. Drug Deliv. Sci. Technol., 2021, 61, 102160 CrossRef CAS.
- N. Tang, R. Zhang, Y. Zheng, J. Wang, M. Khatib, X. Jiang, C. Zhou, R. Omar, W. Saliba, W. Wu, M. Yuan, D. Cui and H. Haick, Adv. Mater., 2022, 34, 2106842 CrossRef CAS PubMed.
- M. Kazanavičius, A. Cepas, V. Kolaityte, R. Simoliuniene and R. Rimdeika, J. Wound Care, 2017, 26, 281–291 CrossRef PubMed.
- L. C. Schmeel, D. Koch, F. C. Schmeel, B. Bücheler, C. Leitzen, B. Mahlmann, D. Kunze, M. Heimann, D. Brüser, A.-V. Abramian, F. Schoroth, T. Müdder, F. Röhner, S. Garbe, B. G. Baumert, H. H. Schild and T. M. Wilhelm-Buchstab, Polymers, 2019, 11, 2112 CrossRef CAS PubMed.
- S. Hajebi, S. Mohammadi Nasr, N. Rabiee, M. Bagherzadeh, S. Ahmadi, M. Rabiee, M. Tahriri, L. Tayebi and M. R. Hamblin, Int. J. Polym. Mater. Polym. Biomater., 2021, 70, 926–940 CrossRef CAS.
- M. Filippi, G. Born, M. Chaaban and A. Scherberich, Front. Bioeng. Biotechnol., 2020, 8, 474 CrossRef PubMed.
- S. Huang and X. Fu, J. Controlled Release, 2010, 142, 149–159 CrossRef CAS PubMed.
- G. Praveen Kumar, R. Shreeya Sai, P. Deepali Venkatesh, V. Priyadharsini, S. Vidhya, C. Chandrananthi, C. Shreya, S. Krithika and G. Keerthana, in Cellulose, ed. P. Alejandro Rodríguez and E. E. M. María, IntechOpen, Rijeka, 2019, ch. 4 Search PubMed.
- R. Naomi, R. Bt Hj Idrus and M. B. Fauzi, Int. J. Environ. Res. Public Health, 2020, 17, 6803 CrossRef CAS PubMed.
- N. Mohd, S. F. S. Draman, M. S. N. Salleh and N. B. Yusof, AIP Conf. Proc., 2017, 1809, 020035 CrossRef.
- M. Oprea and S. I. Voicu, Carbohydr. Polym., 2020, 247, 116683 CrossRef CAS PubMed.
- S. M. Choi, K. M. Rao, S. M. Zo, E. J. Shin and S. S. Han, Polymers, 2022, 14, 1080 CrossRef CAS PubMed.
- J. D. de Amorim, C. J. da Silva Junior, A. D. de Medeiros, H. A. do Nascimento, M. Sarubbo, T. P. de Medeiros, A. F. Costa and L. A. Sarubbo, Molecules, 2022, 27, 5580 CrossRef CAS PubMed.
- E. Tsouko, C. Kourmentza, D. Ladakis, N. Kopsahelis, I. Mandala, S. Papanikolaou, F. Paloukis, V. Alves and A. Koutinas, Int. J. Mol. Sci., 2015, 16, 14832–14849 CrossRef CAS PubMed.
- N. Shah, M. Ul-Islam, W. A. Khattak and J. K. Park, Carbohydr. Polym., 2013, 98, 1585–1598 CrossRef CAS PubMed.
- Z. Li, L. Wang, J. Hua, S. Jia, J. Zhang and H. Liu, Carbohydr. Polym., 2015, 120, 115–119 CrossRef CAS PubMed.
- C. Huang, X. Y. Yang, L. Xiong, H. J. Guo, J. Luo, B. Wang, H. R. Zhang, X. Q. Lin and X. D. Chen, Lett. Appl. Microbiol., 2015, 60, 491–496 CrossRef CAS PubMed.
- M. Ul-Islam, W. Alhajaim, A. Fatima, S. Yasir, T. Kamal, Y. Abbas, S. Khan, A. H. Khan, S. Manan, M. W. Ullah and G. Yang, Int. J. Biol. Macromol., 2023, 231, 123269 CrossRef CAS PubMed.
- F. J. O'Brien, Mater. Today, 2011, 14, 88–95 CrossRef.
- R. Parenteau-Bareil, R. Gauvin and F. Berthod, Materials, 2010, 3, 1863–1887 CrossRef CAS.
- M. Abas, M. El Masry and H. Elgharably, in Wound Healing, Tissue Repair, and Regeneration in Diabetes, ed. D. Bagchi, A. Das and S. Roy, Academic Press, 2020, ch. 19, pp. 393–401 Search PubMed.
- I. N. Amirrah, M. F. Mohd Razip Wee, Y. Tabata, R. Bt Hj Idrus, A. Nordin and M. B. Fauzi, Polymers, 2020, 12, 2168 CrossRef CAS PubMed.
- A. Gaspar-Pintiliescu, A.-M. Stanciuc and O. Craciunescu, Int. J. Biol. Macromol., 2019, 138, 854–865 CrossRef CAS PubMed.
- S. S. Mathew-Steiner, S. Roy and C. K. Sen, Bioengineering, 2021, 8, 63 CrossRef CAS PubMed.
- C. Holmes, J. S. Wrobel, M. P. MacEachern and B. R. Boles, Diabetes, Metab. Syndr. Obes.: Targets Ther., 2013, 6, 17–29 CrossRef PubMed.
- G. A. Rico-Llanos, S. Borrego-González, M. Moncayo-Donoso, J. Becerra and R. Visser, Polymers, 2021, 13, 599 CrossRef CAS PubMed.
- N. Gull, S. M. Khan, M. T. Zahid Butt, S. Khalid, M. Shafiq, A. Islam, S. Asim, S. Hafeez and R. U. Khan, RSC Adv., 2019, 9, 31078–31091 RSC.
- N. Gull, S. M. Khan, A. Islam and M. T. Z. Butt, in Bio Monomers for Green Polymeric Composite Materials, eds. P. M. Visakh, O. Bayraktar and G. Menon, Wiley, New York, United States, 2019, ch. 9, pp. 175–199 Search PubMed.
- A. M. Abdel-Mohsen, J. Frankova, R. M. Abdel-Rahman, A. A. Salem, N. M. Sahffie, I. Kubena and J. Jancar, Int. J. Pharm., 2020, 582, 119349 CrossRef CAS PubMed.
- R. A. Pérez, J.-E. Won, J. C. Knowles and H.-W. Kim, Adv. Drug Delivery Rev., 2013, 65, 471–496 CrossRef PubMed.
- T. Phaechamud, K. Yodkhum, J. Charoenteeraboon and Y. Tabata, Mater. Sci. Eng., C, 2015, 50, 210–225 CrossRef CAS PubMed.
- C. Chircov, A. M. Grumezescu and L. E. Bejenaru, Rom. J. Morphol. Embryol., 2018, 59, 71–76 Search PubMed.
- A. Fakhari and C. Berkland, Acta Biomater., 2013, 9, 7081–7092 CrossRef CAS PubMed.
- J. Voigt and V. R. Driver, Wound Repair Regen., 2012, 20, 317–331 CrossRef PubMed.
- J. S. Frenkel, Int. Wound J., 2014, 11, 159–163 CrossRef PubMed.
- M. Dovedytis, Z. J. Liu and S. Bartlett, Eng. Regener., 2020, 1, 102–113 Search PubMed.
- A. Ström, A. Larsson and O. Okay, J. Appl. Polym. Sci., 2015, 132, 42194 CrossRef.
- G. M. Campo, A. Avenoso, A. D'Ascola, V. Prestipino, M. Scuruchi, G. Nastasi, A. Calatroni and S. Campo, BioFactors, 2012, 38, 69–76 CrossRef CAS PubMed.
- P. A. Janmey, J. P. Winer and J. W. Weisel, J. R. Soc., Interface, 2008, 6, 1–10 CrossRef PubMed.
- P. Heher, S. Mühleder, R. Mittermayr, H. Redl and P. Slezak, Adv. Drug Delivery Rev., 2018, 129, 134–147 CrossRef CAS PubMed.
- N. Laurens, P. Koolwijk and M. P. M. De Maat, J. Thromb. Haemostasis, 2006, 4, 932–939 CrossRef CAS PubMed.
- I. S. Bayer, Molecules, 2022, 27, 4504 CrossRef CAS PubMed.
- N. Y. Becerra, L. M. Restrepo, Y. Galeano, A. C. Tobón, L. F. Turizo and M. Mesa, Int. J. Biomater., 2021, 2021, 9933331 Search PubMed.
- W. Bensaid, J. T. Triffitt, C. Blanchat, K. Oudina, L. Sedel and H. Petite, Biomaterials, 2003, 24, 2497–2502 CrossRef CAS PubMed.
- S. A. Sell, P. S. Wolfe, K. Garg, J. M. McCool, I. A. Rodriguez and G. L. Bowlin, Polymers, 2010, 2, 522–553 CrossRef CAS.
- C. Schneider-Barthold, S. Baganz, M. Wilhelmi, T. Scheper and I. Pepelanova, BioNanoMaterials, 2016, 17, 3–12 Search PubMed.
- P. Zarrintaj, S. Ghorbani, M. Barani, N. P. Singh Chauhan, M. Khodadadi Yazdi, M. R. Saeb, J. D. Ramsey, M. R. Hamblin, M. Mozafari and E. Mostafavi, Bioeng. Transl. Med., 2022, 7, e10261 CAS.
- S. C. Shukla, A. Singh, A. K. Pandey and A. Mishra, Biochem. Eng. J., 2012, 65, 70–81 CrossRef CAS.
- C. Shi, Y. He, X. Feng and D. Fu, J. Biomater. Sci., Polym. Ed., 2015, 26, 1343–1356 CrossRef CAS PubMed.
- T. Bo, P.-P. Han, Q.-Z. Su, P. Fu, F.-Z. Guo, Z.-X. Zheng, Z.-L. Tan, C. Zhong and S.-R. Jia, Food Control, 2016, 61, 123–134 CrossRef CAS.
- N. A. Patil and B. Kandasubramanian, Eur. Polym. J., 2021, 146, 110248 CrossRef CAS.
- R. Singla, S. Soni, V. Patial, P. M. Kulurkar, A. Kumari, S. Mahesh, Y. S. Padwad and S. K. Yadav, Sci. Rep., 2017, 7, 10457 CrossRef PubMed.
- G. Yang, Z. Zhang, K. Liu, X. Ji, P. Fatehi and J. Chen, J. Nanobiotechnol., 2022, 20, 312 CrossRef CAS PubMed.
- E.-E. Tudoroiu, C.-E. Dinu-Pîrvu, M. G. Albu Kaya, L. Popa, V. Anuta, R. M. Prisada and M. V. Ghica, Pharmaceuticals, 2021, 14, 1215 CrossRef CAS PubMed.
- E. Masci, G. Faillace and M. Longoni, BMC Res. Notes, 2018, 11, 239 CrossRef PubMed.
- L. Deng, B. Wang, W. Li, Z. Han, S. Chen and H. Wang, Int. J. Biol. Macromol., 2022, 217, 77–87 CrossRef CAS PubMed.
- Y. Xie, K. Qiao, L. Yue, T. Tang, Y. Zheng, S. Zhu, H. Yang and Z. Fang, Bioact. Mater., 2022, 17, 248–260 CrossRef CAS PubMed.
- S. Yazgan, I. O. Tekin, N. Akpolat and O. Koc, J. Glaucoma, 2021, 30, 1001–1010 CrossRef PubMed.
- L. G. Silva, A. V. Albuquerque, F. C. M. Pinto, R. S. Ferraz-Carvalho, J. L. A. Aguiar and E. M. Lins, J. Mater. Sci.: Mater. Med., 2021, 32, 79 CrossRef CAS PubMed.
- X. Pan, C. Han, G. Chen and Y. Fan, Evidence-Based Complementary Altern. Med., 2022, 2022, 5217617 Search PubMed.
- A. Das, M. Abas, N. Biswas, P. Banerjee, N. Ghosh, A. Rawat, S. Khanna, S. Roy and C. K. Sen, Sci. Rep., 2019, 9, 14293 CrossRef PubMed.
- B. Colak, S. Yormaz, I. Ece, A. Çalişir, K. Körez, M. Çınar and M. Sahin, Int. J. Lower Extremity Wounds, 2020, 21, 279–289 CrossRef PubMed.
- H.-C. Tsai, H.-C. Shu, L.-C. Huang and C.-M. Chen, Formos. J. Surg., 2019, 52, 52–56 CrossRef.
- A.-H. Stricker-Krongrad, Z. Alikhassy, N. Matsangos, R. Sebastian, G. Marti, F. Lay and J. W. Harmon, ePlasty, 2018, 18, e14 Search PubMed.
- R. Pippi, M. Santoro and A. Cafolla, J. Oral Maxillofac. Surg., 2017, 75, 1118–1123 CrossRef PubMed.
- X. Mo, J. Cen, E. Gibson, R. Wang and S. L. Percival, Wound Repair Regen., 2015, 23, 518–524 CrossRef PubMed.
- Y. Hussein, E. M. El-Fakharany, E. A. Kamoun, S. A. Loutfy, R. Amin, T. H. Taha, S. A. Salim and M. Amer, Int. J. Biol. Macromol., 2020, 164, 667–676 CrossRef CAS PubMed.
- M. N. Leite and M. A. C. Frade, Heliyon, 2021, 7, e07572 CrossRef CAS PubMed.
- X. Ding, S. Li, M. Tian, P. Yang, Y. Ding, Y. Wang, G. Duan, D. Zhang, B. Chen and Q. Tan, Int. J. Biol. Macromol., 2023, 226, 1490–1499 CrossRef CAS PubMed.
- S. Yıldırım, H. Ö. Özener, B. Doğan and B. Kuru, J. Periodontol., 2018, 89, 36–45 Search PubMed.
- M. Lee, S. H. Han, W. J. Choi, K. H. Chung and J. W. Lee, Wound Repair Regen., 2016, 24, 581–588 CrossRef PubMed.
- M. Bacakova, J. Pajorova, T. Sopuch and L. Bacakova, Materials, 2018, 11, 2314 CrossRef PubMed.
- X. Lv, Y. Xu, X. Ruan, D. Yang, J. Shao, Y. Hu, W. Wang, Y. Cai, Y. Tu and X. Dong, Acta Biomater., 2022, 146, 107–118 CrossRef CAS PubMed.
- I. Laidmäe, K. Ērglis, A. Cēbers, P. A. Janmey and R. Uibo, J. Mater. Sci.: Mater. Med., 2018, 29, 182 CrossRef PubMed.
- M. Soleimanpour, S. S. Mirhaji, S. Jafari, H. Derakhshankhah, F. Mamashli, H. Nedaei, M. R. Karimi, H. Motasadizadeh, Y. Fatahi, A. Ghasemi, M. S. Nezamtaheri, M. Khajezade, M. Teimouri, B. Goliaei, C. Delattre and A. A. Saboury, Sci. Rep., 2022, 12, 7213 CrossRef CAS PubMed.
- L. P. F. Abbade, S. R. C. S. Barraviera, M. R. C. Silvares, A. B. B. d. C. O. Lima, G. R. Haddad, M. A. N. Gatti, N. B. Medolago, M. T. Rigotto Carneiro, L. D. dos Santos, R. S. Ferreira and B. Barraviera, Front. Immunol., 2021, 12, 627541 CrossRef CAS PubMed.
- V. Mayandi, A. C. Wen Choong, C. Dhand, F. P. Lim, T. T. Aung, H. Sriram, N. Dwivedi, M. H. Periayah, S. Sridhar, M. Fazil, E. T. L. Goh, G. Orive, R. W. Beuerman, T. M. S. Barkham, X. J. Loh, Z. X. Liang, V. A. Barathi, S. Ramakrishna, S. J. Chong, N. K. Verma and R. Lakshminarayanan, ACS Appl. Mater. Interfaces, 2020, 12, 15989–16005 CrossRef CAS PubMed.
- C. Mou, X. Wang, J. Teng, Z. Xie and M. Zheng, J. Nanobiotechnol., 2022, 20, 368 CrossRef CAS PubMed.
- S. Liu, X. Liu, Y. Ren, P. Wang, Y. Pu, R. Yang, X. Wang, X. Tan, Z. Ye, V. Maurizot and B. Chi, ACS Appl. Mater. Interfaces, 2020, 12, 39936 CrossRef CAS PubMed.
- C. Li, Q. Zhang, D. Lan, M. Cai, Z. Liu, F. Dai and L. Cheng, Int. J. Biol. Macromol., 2022, 220, 1049–1059 CrossRef CAS PubMed.
- C. Shi, C. Wang, H. Liu, Q. Li, R. Li, Y. Zhang, Y. Liu, Y. Shao and J. Wang, Front. Bioeng. Biotechnol., 2020, 8, 182 CrossRef PubMed.
- K. Harding, P. Chadwick, S. L. A. Jeffery, D. Gray, E. Lindsay, I. Younis, A. Sharpe, K. Cutting and M. Butcher, J. Wound Care, 2019, 28, 497 CrossRef PubMed.
- K. C. Broussard and J. G. Powers, Am. J. Clin. Dermatol, 2013, 14, 449–459 CrossRef PubMed.
- M. Saco, N. Howe, R. Nathoo and B. Cherpelis, J. Am. Acad. Dermatol., 2016, 74, AB293 Search PubMed.
- S. Alven, S. Peter, Z. Mbese and B. A. Aderibigbe, Polymers, 2022, 14, 724 CrossRef CAS PubMed.
- Y. P. Afsharian and M. Rahimnejad, Polym. Test., 2021, 93, 106952 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2023 |