Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Electrolyte selection toward efficient photoelectrochemical glycerol oxidation on BiVO4†
Heejung
Kong
 a,
Siddharth
Gupta
ab,
Andrés F.
Pérez-Torres
a,
Christian
Höhn
a,
Peter
Bogdanoff
a,
Matthew T.
Mayer
a,
Siddharth
Gupta
ab,
Andrés F.
Pérez-Torres
a,
Christian
Höhn
a,
Peter
Bogdanoff
a,
Matthew T.
Mayer
 ab,
Roel
van de Krol
ab,
Roel
van de Krol
 ac,
Marco
Favaro
ac,
Marco
Favaro
 *a and
Fatwa F.
Abdi
*a and
Fatwa F.
Abdi
 *ad
*ad
aHelmholtz-Zentrum Berlin für Materialien und Energie GmbH, Hahn-Meitner-Platz 1, 14109 Berlin, Germany. E-mail: marco.favaro@helmholtz-berlin.de; fatwa.abdi@helmholtz-berlin.de
bInstitut für Chemie & Biochemie, Freie Universität Berlin, 14195 Berlin, Germany
cInstitut für Chemie, Technische Universität Berlin, Straße des 17. Juni 124, 10623 Berlin, Germany
dSchool of Energy and Environment, City University of Hong Kong, 83 Tat Chee Avenue, Kowloon, Hong Kong S.A.R., China. E-mail: ffabdi@cityu.edu.hk
First published on 31st May 2024
Abstract
Glycerol, a primary by-product of biodiesel production, can be oxidized into various value-added chemicals, significantly enhancing the techno-economic value of photoelectrochemical (PEC) cells. Several studies have explored various photoelectrode materials and co-catalysts, but the influence of electrolytes on PEC glycerol oxidation has remained relatively unexplored despite its significance. Here, we explore the impact of various acidic (pH = 2) electrolytes, namely NaNO3, NaClO4, Na2SO4, K2SO4, and KPi, on PEC glycerol oxidation using nanoporous thin film BiVO4 as a model photoanode. Our experimental findings reveal that the choice of electrolyte anion and cation significantly affects the PEC performance (i.e., photocurrent, onset potential, stability, and selectivity towards value-added products) of BiVO4 for glycerol oxidation. To explain this interesting phenomenon, we correlate the observed performance trend with the ion specificity in the Hofmeister series as well as the buffering capacity of the electrolytes. Notably, NaNO3 is identified as the optimal electrolyte for PEC glycerol oxidation with BiVO4 when considering various factors such as stability and production rates for glycerol oxidation reaction (GOR) products, surpassing the previously favored Na2SO4. Glycolaldehyde emerges as the most dominant product with ∼50% selectivity in NaNO3. The general applicability of our findings is confirmed by similar observation in electrochemical (EC) GOR with a polycrystalline platinum anode. Overall, these results emphasize the critical role of electrolyte selection in enhancing the efficiency of EC/PEC glycerol oxidation.
Introduction
Glycerol, a major by-product of biodiesel production, holds significant potential as a feedstock to produce value-added chemicals.1 A broad palette of high-value products from the glycerol oxidation reaction (GOR), including dihydroxyacetone (DHA), formic acid (FA), glyceraldehyde (GLAD), and glycolaldehyde (GCAD), can be obtained in one or more reaction steps.2 Glycerol can also be oxidized electrochemically, which means it can serve as an anodic reactant in electrochemical (EC) or photoelectrochemical (PEC) devices. The oxidation of glycerol to e.g., DHA in an aqueous environment can be written as| CH2OH − CHOH − CH2OH + 2OH− → CH2OH − CO − CH2OH + 2H2O + 2e− |
and the corresponding reduction reaction as
| 2H2O + 2e− → H2 + 2OH− |
For acidic environments, the reactions are written as
| CH2OH − CHOH − CH2OH → CH2OH − CO − CH2OH + 2H+ + 2e− |
| 2H+ + 2e− → H2 |
The overall reaction is given as
| CH2OH − CHOH − CH2OH → CH2OH − CO − CH2OH + H2 |
The oxidation of glycerol (ΔG = 3.9 kJ mol−1, when coupled with the hydrogen evolution reaction, HER) requires significantly less energy and can be driven at much lower overpotentials compared to hydrogen production via the direct water splitting reaction (ΔG = 237.2 kJ mol−1, requiring an additional overpotential of at least 0.3 V for the oxygen evolution reaction, OER). Moreover, GOR products possess significantly higher economic value than glycerol. For instance, the market price of DHA ranges from approximately 2 to 150 € kg−1, depending on the degree of purity.3 In contrast, refined glycerol (≥99%) is available for ∼0.75 € kg−1, while the worldwide wholesale price for crude glycerol (∼80% purity) was ∼0.1 € kg−1 in 2020.4 Therefore, the techno-economic case for this PEC Power-to-X process is much more attractive than that for PEC water splitting.
In many (photo)electrochemical reactions, the composition of the electrolyte is known to be crucial.5 For example, Ding et al. showed that the choice of cations in alkaline electrolytes affects the water oxidation performance of TiO2 photoanodes. This was attributed to different back-reaction rates (i.e., oxygen reduction reaction) induced by the various cations.6 Similarly, the choice of anions has been shown to influence the performance of WO3 photoanodes, as these anions can be parasitically oxidized alongside water at the electrode surface.5a,7 In the case of PEC glycerol oxidation, too few studies are available to determine the optimal supporting electrolyte for achieving high efficiency and stability. Since Liu et al. demonstrated that glycerol oxidation using BiVO4 photoanodes exhibited the highest efficiency in acidic solutions (pH = 2) based on sodium sulfate (Na2SO4),8 subsequent studies mainly used Na2SO4-based electrolytes and predominantly focused on modifying the photoanode material and/or the deposited co-catalyst to optimize the performance.9 It is still unclear whether altering the electrolyte composition could also optimize the PEC glycerol oxidation performance.
In this paper, we demonstrate that the electrolyte composition substantially impacts the glycerol oxidation performance of our model photoanode material, bismuth vanadate (BiVO4). We investigate acidic electrolytes (pH = 2) with different cations and anions: sodium nitrate (NaNO3), sodium perchlorate (NaClO4), sodium sulfate (Na2SO4), potassium sulfate (K2SO4), and potassium phosphate (KPi), the latter being a pH buffer solution. Our systematic study reveals that, when performance factors such as photocurrent, stability, and production rates towards high-value GOR products are comprehensively considered, BiVO4 photoanodes exhibit the highest glycerol oxidation performance in NaNO3, surpassing the commonly employed Na2SO4. To the best of our knowledge, NaNO3 has not been previously reported as an electrolyte for PEC GOR. The underlying reason behind this observed performance difference is discussed, particularly in relation to the classification of the ions in the Hofmeister series and the pH buffering capacities of the employed solutions. Furthermore, the same GOR performance trend with the electrolyte choice is also observed when the BiVO4 photoanode is replaced with a polycrystalline platinum (Pt) anode, suggesting that our explanations may be broadly applicable, irrespective of the anode material or whether the process is EC- or PEC-driven. Overall, these findings emphasize the critical role of electrolyte selection for achieving high performance and stability in glycerol oxidation and underscore the potential of NaNO3 as a favorable electrolyte for glycerol oxidation.
Results and discussion
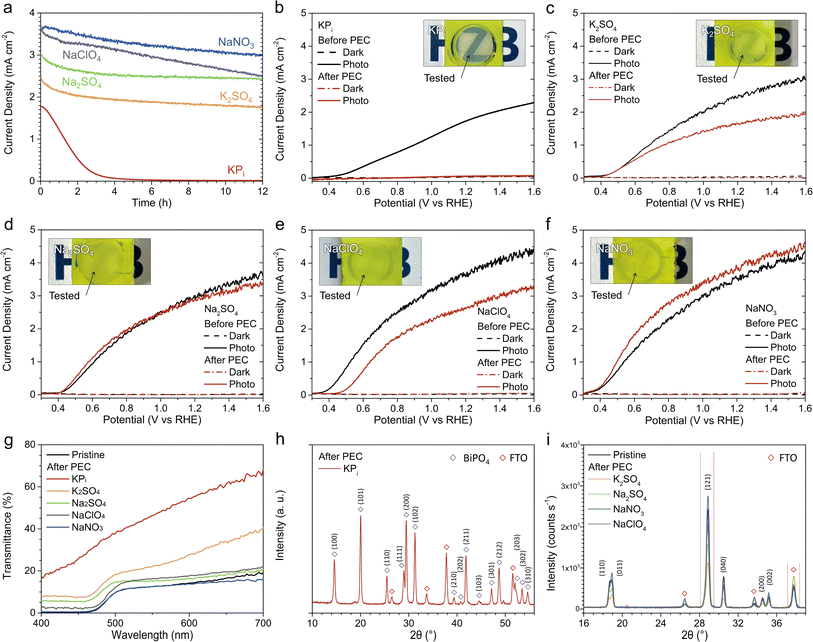
BiVO4 photoanodes were prepared using a previously reported electrodeposition method,10 and the detailed synthesis method is described in the ESI.† The samples exhibit a nanoporous morphology (Fig. S1†), while the X-ray diffractogram shows a monoclinic crystalline phase without any impurities (Fig. S2a†). Tauc analysis of the samples revealed an indirect bandgap of 2.48 eV (Fig. S2b†), which is within the range of typical bandgap values reported for monoclinic BiVO4.11We first compare the PEC performance of BiVO4 toward water oxidation in various acidic electrolytes, in the absence of glycerol. Fig. 1a shows the linear sweep voltammetry (LSV) curves measured under AM 1.5G simulated sunlight (100 mW cm−2) in KPi, K2SO4, Na2SO4, NaClO4, and NaNO3 (see Fig. S3a† for the corresponding dark LSV curves). The concentration of all electrolytes was kept constant at 0.5 M, and the pH value was adjusted to 2. To avoid altering the ionicity of the solutions, the pH of each investigated electrolyte was lowered using mineral acids with the same anion of the electrolyte itself (e.g., in the case of NaNO3, the pH was lowered using HNO3, whereas H2SO4 was used for K2SO4). Our electrodeposited BiVO4 photoanodes showed similar photocurrent (0.5–0.7 mA cm−2) at 1.23 V vs. the reversible hydrogen electrode (VRHE) and onset potential (∼0.6 VRHE) in all electrolytes (see Fig. S4 and Table S1† for the measurements' reproducibility), indicating that the difference in the electrolyte composition does not affect their performance toward water oxidation.
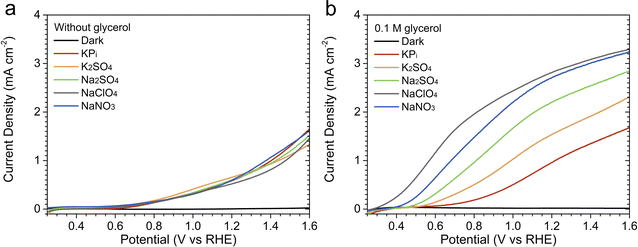 | ||
| Fig. 1 Linear sweep voltammetry (LSV) curves recorded in different acidic solutions (pH = 2), all having a concentration of 0.5 M: (a) in the absence of glycerol and (b) with 0.1 M glycerol. Measurements were performed at a scan rate of 20 mV s−1, sweeping from negative to positive potentials relative to the reversible hydrogen electrode (RHE), under AM 1.5G simulated sunlight illumination. LSV curves recorded in NaNO3 without illumination are also shown as the representative dark curves; dark LSV curves recorded in other electrolytes are shown in Fig. S3.† | ||
To investigate the effect of electrolyte composition on the performance of PEC glycerol oxidation using BiVO4, 0.1 M glycerol was added to each of the five electrolyte solutions. Fig. 1b displays the LSV curves measured under illumination (also see Fig. S5 and Table S2† for reproducibility), while the LSV curves obtained in the absence of illumination are presented in Fig. S3b.† In the presence of 0.1 M glycerol, our BiVO4 photoanodes showed increased photocurrent in all electrolytes. This is attributed to the lower overpotential needed for the GOR as compared to the OER. Interestingly, the photocurrents varied significantly depending on the electrolyte composition. The lowest photocurrent was obtained in the KPi solution, followed by the K2SO4, Na2SO4, NaNO3, and NaClO4 solutions. The photocurrent improvement was rather significant, with 180% increase by changing the electrolyte from KPi to NaClO4 (1.07 ± 0.08 vs. 2.97 ± 0.11 mA cm−2 at 1.23 VRHE).
The same photocurrent trend was also observed when the glycerol concentration was increased. Fig. S6† shows the LSV curves of BiVO4 photoanodes measured with 0.5 M glycerol. As in the case of 0.1 M glycerol, our photoanodes showed the lowest photocurrent in the KPi solution and the highest in the NaClO4 solution. However, the photocurrents in the NaNO3 and NaClO4 solutions were similar, especially at higher potentials (>1.0 VRHE). The photocurrent difference was more pronounced at relatively low potentials. For example, in the NaNO3 and NaClO4 solutions, the photocurrent already reached 1.0 mA cm−2 at approximately 0.5 VRHE, whereas in the KPi solution, a potential of 0.9 VRHE was needed.
Chronoamperometry (CA) was performed at 1.23 VRHE for a duration of 12 hours to assess the stability of our photoanodes in the five acidic electrolytes containing 0.5 M glycerol, as reported in Fig. 2a. In the KPi solution, the photocurrent decreased rapidly to 1/e (36.7%) of its initial value within only 1.8 hours, ultimately resulting in a complete loss of photocurrent within 8 hours. In contrast, the photoanodes exhibited photocurrent retention of approximately 70–80% over the 12 hours period in K2SO4, Na2SO4, NaClO4, and NaNO3. The corresponding LSV curves obtained before and after the CA are shown in Fig. 2b–f. After the CA measurement in KPi, no photoactivity remained (see Fig. 2b), consistent with the complete photocurrent loss during the CA experiment. The samples tested in K2SO4 and NaClO4 also showed a consistent photocurrent decrease after the CA (see Fig. 2c and e). Interestingly, the samples tested in Na2SO4 and NaNO3 maintained similar photocurrent levels in their LSV measurements compared to those observed before the 12 hours CA, indicating some degree of recovery (see Fig. 2d and f). Note that we limit our study to pH 2 electrolyte solutions, as the use of more acidic pH (e.g., pH 1) renders the BiVO4 to be unstable even in the NaNO3 solution (Fig. S7†).
The photographs of the sample taken after the 12 hours CA in each electrolyte solution are presented in the insets of Fig. 2b–f. In the case of the samples tested in KPi, the exposed area became white and transparent. This is not the case for samples tested in K2SO4, Na2SO4, NaClO4, and NaNO3; no significant difference was observed between the appearance of the tested and untested regions. We quantified our observation by measuring the UV-vis transmittance of these regions, as shown in Fig. 2g. Notably, the transmittance of the sample tested in NaNO3 closely matched that of the pristine sample across the entire wavelength range. The samples tested in K2SO4, Na2SO4, and NaClO4 exhibited a notable increase in transmittance, albeit with varying amount, suggesting the possible loss of BiVO4 material due to photocorrosion and consequent dissolution. The sample tested in KPi displayed significantly higher transmittance across the entire wavelength range, consistent with the appearance of the sample after electrolysis (see inset of Fig. 2b).
We then compared the crystallinity of the BiVO4 samples before and after the 12 hours CA measurements. Fig. 2h displays the X-ray diffractogram for the sample tested in KPi. Diffraction peaks corresponding to BiPO4 (as well as the FTO substrate) were detected, while peaks characteristic of monoclinic BiVO4 were absent. This suggests that during the CA measurement, vanadium atoms in BiVO4 leached into the electrolyte and were substituted by phosphorus atoms from the KPi solution, leading to a crystal structure transformation and eventually a complete conversion to BiPO4. Indeed, BiPO4 has a wide bandgap (∼3.85 eV),12 which explains the white and transparent appearance of the film after CA. Such a conversion could also account for the low photocurrent observed with KPi (Fig. 1b). Note that the formation of a BiPO4 thin film on the surface of BiVO4 upon exposure to KPi buffer solution at pH 7 has been previously reported.13 A full conversion to BiPO4 has not been reported, which we attribute to the use of lower pH.
Fig. 2i shows the X-ray diffractograms of the BiVO4 samples measured after the 12 hours CA measurements in K2SO4, Na2SO4, NaClO4, and NaNO3. Unlike the sample measured in KPi, diffraction peaks characteristic of monoclinic BiVO4 can be detected in these samples. For samples measured in K2SO4, Na2SO4, and NaClO4, the intensities of the (121) diffraction peak, which is the most prominent reflection for electrodeposited monoclinic BiVO410, decreased, while the intensities of the FTO peak at 2θ = 37.6° increased. In the case of the sample measured in NaNO3, the X-ray diffraction (XRD) pattern closely resembled that of the pristine sample. We quantified the area under the (121) peak and the FTO peak at 2θ = 37.6° and then calculated their ratio. For the sample tested in NaNO3, this ratio was 4.8, which was higher than that of the pristine sample (3.9). In contrast, the ratios for the samples tested in NaClO4, Na2SO4, and K2SO4 were considerably lower; 2.8 for NaClO4, 1.8 for Na2SO4, and 1.4 for K2SO4, respectively.
The XRD and UV-vis transmittance results above indicate that BiVO4 substantially degraded during GOR in acidic NaClO4, Na2SO4 and K2SO4, likely due to photocorrosion, but not in NaNO3. These findings are interesting because, according to the Pourbaix diagram,14 BiVO4 is expected to decompose into BiO+ and VO43− at pH 2 and 1.23 VRHE. This suggests that NO3− (either by itself or in conjunction with glycerol) plays a role in stabilizing BiVO4 under these conditions. The exact mechanism requires further investigation beyond the scope of this study.
The surface chemical characteristics of the pristine sample and samples subjected to 12 hours CA in glycerol-containing K2SO4, Na2SO4, NaClO4, and NaNO3 electrolytes were investigated using X-ray photoelectron spectroscopy (XPS). The sample subjected to the same treatment in KPi was not tested since XRD already showed a clear transformation to BiPO4. The Bi 4f core-level spectra are displayed in Fig. S8,† and the O 1s and V 2p core-level spectra are presented in Fig. S9.† While the binding energy difference (ΔBE) between the Bi 4f5/2 and 4f7/2 peaks remained constant, the tested samples exhibited a slight shift towards higher binding energies. The magnitude of this peak shift was 0.1 eV in the NaNO3 solution and increased to 0.2 eV in the K2SO4, Na2SO4, and NaClO4 solutions. A notable difference among the samples was observed in the O 1s core-level spectra. The samples tested in the K2SO4, Na2SO4, and NaClO4 solutions showed distinct peaks at higher binding energies of approximately 531–533 eV. These peaks could originate from adsorbed hydroxyl ions (at ∼532 eV),15 likely due to glycerol adsorption during GOR. Their significant emergence may imply a somewhat reduced surface chemical stability of BiVO4 during PEC oxidation of glycerol in the K2SO4, Na2SO4, and NaClO4 solutions at pH 2. Indeed, if the O2− ions in BiVO4 were substituted by hydroxyl (OH−) ions, the resulting decrease in the number of valence electrons could potentially lead to a shift to higher binding energies in the Bi 4f and V 2p peaks. In contrast, the sample tested in NaNO3 solution exhibited only a slight increase in signal intensity near 532 eV, indicating the relatively superior chemical stability of BiVO4 in pH 2 NaNO3 solution during PEC glycerol oxidation. Overall, these XPS findings are in agreement with the XRD and UV-vis results.
The selectivity and production rates towards value-added GOR products during the 12 hours CA tests in various acidic electrolyte solutions were analyzed using high-performance liquid chromatography (HPLC). Detailed experimental methods for the HPLC analyses are provided in the ESI† (also refer to Fig. S10–S12† for calibration information). KPi was excluded from our analysis due to the severe instability of BiVO4 during the GOR in the pH 2 KPi solution (vide supra). Fig. 3a displays the selectivity of BiVO4 towards various GOR products and the total faradaic efficiency, calculated in terms of charges used for GOR, in K2SO4, Na2SO4, NaClO4, and NaNO3 solutions (the corresponding chromatograms are shown in Fig. S13–S16†). In our study, GCAD (C2H4O2), GLAD (C3H6O3), DHA (C3H6O3), and FA (CH2O2) were detected as the GOR products. Notably, GCAD emerged as the most dominant product in all electrolytes, which is consistent with a recent report.16 However, the selectivity varied depending on the electrolyte. In K2SO4, the selectivity for GLAD (28%) a C3 molecule, was the highest among the electrolytes tested. Furthermore, the selectivity for FA, the only C1 product in our case, was as low as 10%. Interestingly, in the Na2SO4, NaClO4, and NaNO3 solutions, the selectivities for GCAD (C2) and FA (C1) increased, while the selectivity for GLAD (C3) decreased (the selectivity for DHA remained relatively constant at ∼20% in all electrolytes). In NaNO3, the selectivity for GCAD exceeded 50% and the selectivity for GLAD was as low as 14%. These results suggest that the rate of C–C cleavage in GLAD, which was proposed as a pathway to the formation of GCAD and FA,16 might differ among various electrolyte solutions, consequently influencing the overall GOR performance. One possible explanation is that GLAD adsorbs more favorably onto the BiVO4 surface in the NaNO3 solution compared to others. Further computational and/or experimental studies beyond the scope of our work are needed to reveal the actual mechanism. Fig. 3b presents the production rates of the four GOR products. In the NaNO3 solution, the production rate of GCAD was calculated to be 318 mmol m−2 h−1, which is, to the best of our knowledge, the highest reported value for undoped BiVO4 without any co-catalysts.
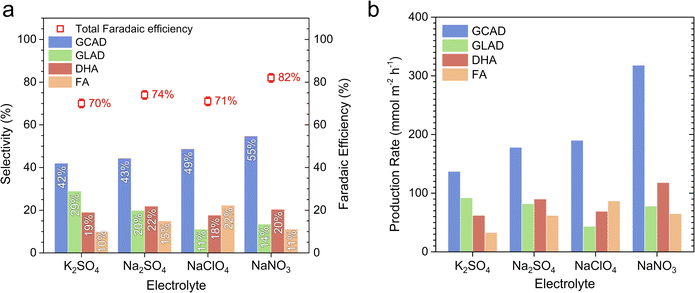 | ||
| Fig. 3 Product analysis using high-performance liquid chromatography (HPLC). (a) Selectivity and (b) production rate towards glycerol oxidation reaction (GOR) products in various acidic electrolytes (pH = 2). Total faradaic efficiency, calculated in terms of charges used for GOR, is presented on the right y-axis in (a). The methods for calculating selectivity, production rate, and faradaic efficiency are described in the ESI.† Liquid samples were collected after 12 hours photoelectrolysis at a constant potential of 1.23 VRHE, corresponding to the measurements shown in Fig. 2a. GCAD: glycolaldehyde, GLAD: glyceraldehyde, DHA: dihydroxyacetone, FA: formic acid. | ||
The total faradaic efficiency was highest in the NaNO3 solution, as shown in Fig. 3a, where an estimated 82% of the photo-generated holes were used for oxidizing glycerol. Mass spectroscopy (MS) results (see Fig. S17†) obtained from the NaNO3 solution indicate that no O2 was generated above our detection limit. This suggests that the OER was effectively suppressed in the NaNO3 solution, likely owing to the relatively low energy requirement of the GOR. A slight increase in the mass signal corresponding to CO2 was observed, suggesting the potential for complete oxidation of glycerol to CO2. We tentatively attribute the remaining photo-generated holes (∼18%) to the generation of other glycerol oxidation products including CO2 (undetected with HPLC and MS) or to photocorrosion. The lower faradaic efficiencies observed in Na2SO4 (74%), NaClO4 (71%), and K2SO4 (72%) support the hypothesis of partial hole consumption due to photocorrosion, especially given the poorer photostability of BiVO4 in these solutions. We also confirm that nitrate reduction reaction (NRR), which could potentially occur at the counter electrode when NaNO3 is used as the electrolyte, was not detected in our experiments (see Fig. S18†).
In terms of photocurrent, stability, and selectivity to high-value GCAD, NaNO3 was identified as the most suitable electrolyte for PEC glycerol oxidation using BiVO4. To elucidate how the choice of electrolyte influences the GOR performance of BiVO4, it is first necessary to examine the electrical conductivities of the electrolyte solutions. The electrical conductivities of 0.5 M NaNO3, NaClO4, Na2SO4, K2SO4, and KPi solutions (all at pH 2) were measured as 75.5 mS cm−1, 98.2 mS cm−1, 102.6 mS cm−1, 141.2 mS cm−1, and 100.0 mS cm−1, respectively, which do not correlate with the trends observed in GOR performance. Moreover, if the differing GOR performance observed in BiVO4 was due to differences in electrical conductivity among the electrolytes, then the water-splitting performance of BiVO4 should have varied accordingly.
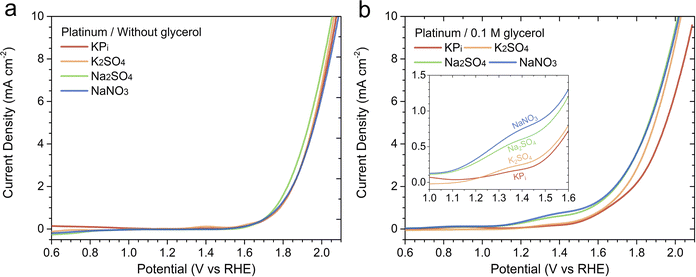
Moreover, to determine whether the GOR performance trend in different acidic electrolytes is specific to BiVO4 and/or the PEC process, we examined the GOR performance of a polycrystalline Pt anode. Fig. 4a displays the LSV curves of the Pt anode measured without glycerol in various acidic electrolytes. Note that NaClO4 solution is excluded from this particular study, the formation of platinum oxide that degrades the activity has been reported when Pt is exposed to perchloric acid.17 The Pt anode exhibited similar LSV curves across all electrolytes. For example, the onset potential for OER was observed at approximately 1.6 VRHE, and an overpotential of ∼0.8 V was required to achieve 10 mA cm−2 (at ∼2.0 VRHE) in all solutions. Fig. 4b presents the LSV curves of the Pt anode measured in the presence of 0.1 M glycerol. The current density for the Pt anode began to increase earlier than in its absence, similar to the case with BiVO4, reflecting the lower energy requirement of GOR compared to OER. Interestingly, the Pt anode exhibited the same GOR performance trend as BiVO4 in terms of current density. At 1.4 VRHE, a potential more negative than the onset for OER, the current densities at 1.4 VRHE ranked as follows: NaNO3 > Na2SO4 > K2SO4 > KPi (see Fig. 4b). These results indicate that the influence of the electrolyte on GOR performance, particularly in terms of current density, is not unique to BiVO4 and may be generally applicable to other anode materials and whether the reaction is EC- or PEC-driven.
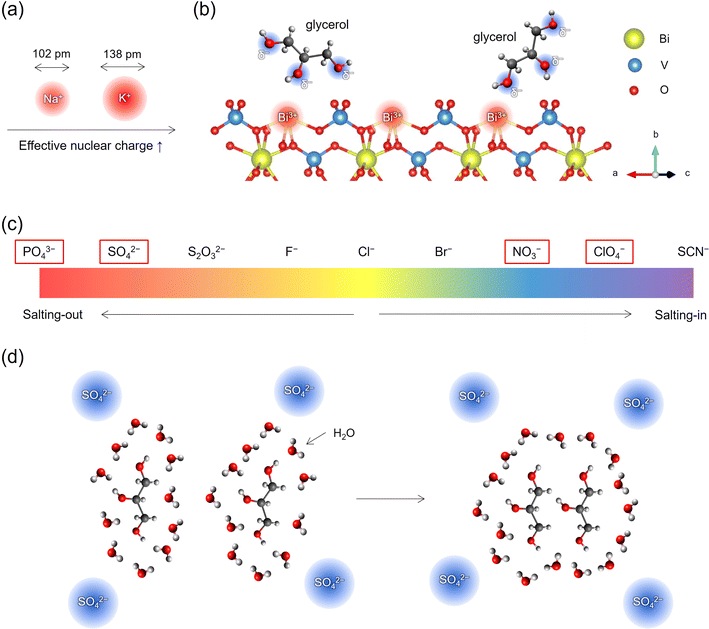
These findings lead us to investigate the impact of specific ions in the electrolytes on PEC glycerol oxidation performance, extending beyond mere electrolyte conductivity or anode material. Specifically, the photocurrent trend in the different electrolytes (for BiVO4: NaClO4 > NaNO3 > Na2SO4 > K2SO4 > KPi) suggests that the presence of specific ions in the electrolyte is beneficial/detrimental to the PEC glycerol oxidation performance. First, the trend indicates that Na+ is more favorable than K+ for GOR on BiVO4. We attribute this observation to the prominent difference between the cation size. As illustrated in Fig. 5a, K+ has an approximately 30% larger effective ionic radius of 138 pm compared to Na+ (102 pm),18 resulting in a larger effective nuclear charge than Na+.19 According to density functional theory (DFT) calculations performed by Liu et al., the hydroxyl groups of glycerol spontaneously adsorb at the exposed Bi3+ sites of BiVO4 through electrostatic interactions between Bi3+ and oxygen in the hydroxyl groups, as depicted in Fig. 5b.8 Similarly, previous DFT calculations on glycerol adsorption to Pt surfaces also showed that the hydroxyl groups of glycerol adsorb to the surface Pt atoms. Furthermore, previous liquid chromatography-mass spectrometry results showed that glycerol molecules in Na2SO4 solutions (pH 2) were found in the form of C3H8O3Na (glycerol = C3H8O3).8 This observation indicates a strong interaction between the hydroxyl groups of glycerol, with their negative partial charge δ−, and the cations in the electrolytes. Given that K+ has a larger effective nuclear charge (defined as Zeff = Z − S, where Z is the number of protons (atomic number) and S is the shielding constant),20 the glycerol hydroxyl groups are expected to interact more strongly with K+ than with Na+. Consequently, this can lead to stronger electrical neutralization of the hydroxyls of glycerol, which may weaken the electrostatically induced adsorption of glycerol molecules to the Bi3+ sites. As a result, GOR at BiVO4 in K+-containing electrolytes would be hindered.
In addition, the superior performance of BiVO4 in NaNO3 and NaClO4 than in Na2SO4 may be explained considering the different types of anions, NO3−, ClO4−, and SO42−. The Pt anode also exhibited a higher GOR current in NaNO3 than in Na2SO4. Interestingly, we note that SO42− stands out as one of the anions exhibiting potent specific ion effects, positioned towards the left end of the Hofmeister series,21 as shown in Fig. 5c. This indicates a strong salting-out effect of SO42−. In contrast, NO3− and ClO4− are positioned closer to the right end of the Hofmeister series, indicating that these anions exhibit comparatively weak specific ion effects and limited salting-out effects. Anions with relatively small sizes and high charge densities, such as SO42− and PO43−, trigger the salting-out phenomenon in aqueous solutions by inducing electronic repulsion and enhancing the hydrophobic nature of the molecules involved.22 The stronger hydrophobicity prompts the molecules to aggregate, thereby minimizing the entropic cost related to a highly ordered structure at the interface between the solute and water, as illustrated in Fig. 5d. This aggregation of glycerol molecules could reduce the probability of glycerol adsorbing onto Bi3+ sites, thereby hindering GOR on BiVO4. It is noted that a similar anion effect following the trend of the Hofmeister series has also been recently discussed to explain different H2 microbubble coalescence efficiencies.23
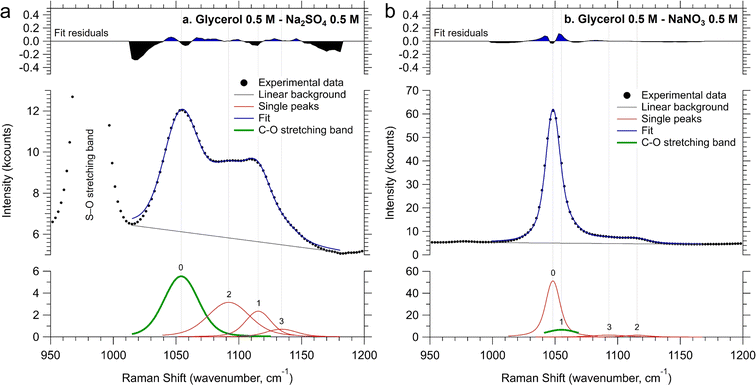
In order to experimentally investigate the aforementioned molecular aggregation, we performed Raman scattering spectroscopy on electrolyte solutions containing 0.5 M glycerol (the detailed method is described in the ESI†). We focused on the C–O stretching band, centered at around 1050 cm−1, as indicated by the grey area in the full range spectra reported in Fig. S19.†24 The band was scanned between 950 cm−1 and 1200 cm−1 and fitted with multiple symmetric Voigt functions after subtracting a linear background. Fig. S20a and S20b† report the fitting results for the spectral references, namely 0.5 M glycerol in water and 0.5 M of aqueous NaNO3. The latter was taken since a spectral overlap exists between the C–O and the N–O stretching bands. The line shape of the Voigt functions retrieved by fitting the spectral references was then kept constant during the fitting of the pristine electrolytes, with their spectral position and full-width at half-maximum (FWHM) allowed to adjust in a range of 5 cm−1 with respect to their values in the spectral references during the fitting procedure. Fig. 6a and b show the fitting procedure obtained on the 0.5 M Na2SO4 and NaNO3 solutions (pH 2) containing 0.5 M glycerol, respectively. Table 1 summarizes the Raman shift values of the C–O stretching band retrieved during the fitting procedure of the samples above and the corresponding difference with respect to the glycerol in water without any supporting electrolyte. It is evident that there is a red shift of the C–O stretching frequency, indicating that the reduced mass of the C–O stretching mode of glycerol increases upon the addition of salts. This phenomenon can be attributed to the formation of glycerol “aggregates” in the solution, driven by the coulombic interaction between the partial negative charge present on the oxygen of glycerol's OH groups and the ions introduced into the solution. Notably, the observed downward shift of the C–O stretching frequency is more pronounced with Na2SO4 than with NaNO3. These observations support our hypothesis that sulfate groups prompt the formation of slightly more glycerol aggregates in the solution.25
| C–O stretching Raman shift (cm−1) | C–O stretching ΔRaman shift (cm−1) | |
|---|---|---|
| 0.5 M glycerol | 1055.8 | — |
| 0.5 M glycerol + 0.5 M NaNO3 | 1055.0 | 0.8 |
| 0.5 M glycerol + 0.5 M Na2SO4 | 1054.1 | 1.7 |
We also note that the photocurrent trend may also be explained by the ability of the different electrolytes in suppressing (or allowing) pH changes. When glycerol undergoes oxidation at the anode, protons are generated. These protons subsequently migrate to the cathode and participate in the HER. Since acidic environment is preferred for the GOR,8 the overall glycerol oxidation performance may be affected if protons produced near the BiVO4/electrolyte interface are removed from the reaction environment through buffering. In other words, differently from what has been previously observed for PEC water oxidation,26 a lower buffering capacity within the electrolyte solution might correlate with an improved GOR performance. We therefore evaluated the buffering capacity of the solutions by introducing equimolar amounts of protons to the glycerol-containing solutions. For instance, 25 mmol of H2SO4 was added to 1 L of a pH 2 solution of either K2SO4 or Na2SO4 containing 0.1 M glycerol, each with an electrolyte concentration of 0.5 M. Subsequently, the pH of the resulting mixtures was measured. In a separate instance, 50 mmol of HNO3 or HClO4 was added to 1 L of a pH 2 NaNO3 or NaClO4 solution containing 0.1 M glycerol (electrolyte concentration = 0.5 M). The resultant changes in pH (ΔpH) induced by the addition of 50 mM protons are presented in Table 2. Interestingly, the NaNO3 and NaClO4 solutions exhibited considerably higher ΔpH (indicative of the lowest buffering capacity) compared to the other three solutions, whereas the KPi solution demonstrated the lowest ΔpH (indicative of the highest buffering capacity). The estimated buffering capacities were ranked as follows: NaNO3 < NaClO4 < Na2SO4 < K2SO4 < KPi. This trend agrees well with the photocurrent trend, except for the ranking between NaClO4 and NaNO3, thus implying a potential impact of buffering action in the electrolyte on the GOR process.
| KPi | K2SO4 | Na2SO4 | NaClO4 | NaNO3 | |
|---|---|---|---|---|---|
| Initial pH | 2.00 ± 0.01 | 1.99 ± 0.01 | 1.98 ± 0.01 | 2.00 ± 0.01 | 1.99 ± 0.02 |
| Final pH | 1.93 ± 0.01 | 1.80 ± 0.01 | 1.72 ± 0.02 | 1.38 ± 0.03 | 1.19 ± 0.07 |
| ΔpH | 0.07 ± 0.01 | 0.19 ± 0.01 | 0.27 ± 0.01 | 0.62 ± 0.05 | 0.80 ± 0.09 |
Despite the evidence provided above, the speculative nature of these explanations is acknowledged. Further experiments beyond the scope of this study would be needed to fully reveal the mechanism behind the clearly observed performance trend. Another factor that needs to be considered is the effect of anions on the reorganization energy of glycerol in the electrolyte. As outlined in the Marcus-Gerischer theory,27 the charge transfer rate at a semiconductor/liquid interface depends on the alignment between the energy levels in the semiconductor (i.e., conduction and valence bands) and the donor/acceptor states of the redox species in the electrolyte (in our case, glycerol). Any change in the reorganization energy, possibly induced by the presence of different anions, would change the energy gap between the donor/acceptor states of the solute. Hence, this would change the relative energy alignment at the interface and consequently modify the electron transfer kinetics. The reorganization energy of glycerol in these acidic electrolytes, which is to the best of our knowledge not yet available, needs therefore to be evaluated.
Conclusion
In summary, the influence of various acidic electrolytes (KPi, K2SO4, Na2SO4, NaClO4, and NaNO3; pH = 2) on the PEC glycerol oxidation over BiVO4 was systematically investigated. Interestingly, we observed that BiVO4 exhibited the following GOR performance trend: NaClO4, NaNO3 > Na2SO4 > K2SO4 > KPi, with the photocurrent in NaClO4 ∼3-fold of that in KPi. Although our BiVO4 photoanodes exhibited the highest photocurrent in NaClO4, the low production rate of GOR products, due to the poor stability of BiVO4 in this electrolyte solution, made it less promising than NaNO3. NaNO3 emerged as the preferred electrolyte for PEC glycerol oxidation on BiVO4, offering superior performance in terms of photocurrent, stability, and selectivity towards value-added GOR products. Glycolaldehyde (GCAD) was identified as the most dominant GOR product in our study, achieving a selectivity higher than 50% in NaNO3. We attributed the observed GOR performance trend among the employed electrolytes to the various effects induced by both cations and anions on the GOR performance of BiVO4. First, the larger effective nuclear charge of K+vs. Na+ may result in reduced electrostatic adsorption of glycerol onto the BiVO4 surface, consistent with the lower performance in K+-containing electrolytes. In addition, the specific ion effects were considered as they may impact the aggregation of glycerol in the electrolyte. Our Raman spectroscopy results suggest more extensive glycerol aggregation in the presence of SO42− compared to NO3−, consistent with the weaker salting-out effect of NO3− ions indicated by the Hofmeister series. Finally, we found an inverse relationship between the buffering capacity of the electrolyte and the GOR performance of BiVO4. The underlying mechanism is likely a combination of the factors above, and future studies should be directed towards understanding this complex process through e.g., operando investigation of the BiVO4/liquid electrolyte interface during PEC GOR and an assessment of the reorganization energy of glycerol in diverse electrolytes. The general applicability of our results to other anode materials and/or whether the reaction is EC- or PEC-driven was suggested as dark EC measurements with polycrystalline Pt anodes also showed a similar GOR current density trend. Overall, our findings suggest the critical role of electrolyte selection in optimizing EC/PEC glycerol oxidation, with potential implications for other (photo)electrocatalytic reactions.Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.Author contributions
H. K.: investigation (sample preparation, EC, PEC, XRD, XPS, UV-vis, HPLC, solution conductivity measurements, buffering capacity measurements), data curation, visualisation, conceptualisation, writing – original draft; S. G.: investigation (HPLC), writing – review & editing; A. F. P.-T.: investigation (HPLC), writing – review & editing; C. H.: investigation (XPS), writing – review & editing; P. B.: investigation (MS), data curation, visualisation, writing – review & editing; M. T. M.: resources, writing – review & editing; R. v. d. K.: supervision, writing – review & editing; M. F.: supervision, investigation (Raman spectroscopy), data curation, visualisation, conceptualisation, writing – review & editing; F. F. A.: supervision, funding acquisition, conceptualisation, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the European Innovation Council (EIC) via OHPERA project (grant agreement 101071010). The authors acknowledge the Helmholtz Energy Materials Foundry (HEMF, GZ 714-48172-21/1) and the CatLab project (Förderkennzeichen 03EW0015A) funded by the German Federal Ministry of Education and Research (BMBF). The measurements for product analysis via in-line mass spectroscopy were carried out in the “Solar Fuels Testing Facility” laboratory of the Helmholtz Association's HEMF initiative. The authors thank Dr David E. Starr for valuable discussions about the Hofmeister series, and Prof. Dr Sixto Gimenez Julia, Dr Camilo Arturo Mesa Zamora, and Eva Jie Yun Ng Leon for their assistance in the HPLC data acquisition and analysis.References
- G. Dodekatos, S. Schünemann and H. Tüysüz, Recent Advances in Thermo-, Photo-, and Electrocatalytic Glycerol Oxidation, ACS Catal., 2018, 8(7), 6301–6333 CrossRef CAS.
- M. Simões, S. Baranton and C. Coutanceau, Electrochemical Valorisation of Glycerol, ChemSusChem, 2012, 5(11), 2106–2124 CrossRef PubMed.
- (a) B. Katryniok, H. Kimura, E. Skrzyńska, J.-S. Girardon, P. Fongarland, M. Capron, R. Ducoulombier, N. Mimura, S. Paul and F. Dumeignil, Selective catalytic oxidation of glycerol: perspectives for high value chemicals, Green Chem., 2011, 13(8), 1960–1979 RSC; (b) X. Zhou, Q. Sun, H. Yan, C. Yang and N. Yan, Integrating Time, Region, and Society Dimensions into a Multi-Dimensional Life-Cycle Analysis for Sustainable Dihydroxyacetone Production, ACS Sustain. Chem. Eng., 2023, 11(29), 10795–10811 CrossRef CAS; (c) Á. Vass, B. Endrődi and C. Janáky, Coupling electrochemical carbon dioxide conversion with value-added anode processes: An emerging paradigm, Curr. Opin. Electrochem., 2021, 25, 100621 CrossRef.
- C. A. G. Quispe, C. J. R. Coronado and J. A. Carvalho Jr, Glycerol: Production, consumption, prices, characterization and new trends in combustion, Renewable Sustainable Energy Rev., 2013, 27, 475–493 CrossRef CAS.
- (a) J. C. Hill and K.-S. Choi, Effect of Electrolytes on the Selectivity and Stability of n-type WO3 Photoelectrodes for Use in Solar Water Oxidation, J. Phys. Chem. C, 2012, 116(14), 7612–7620 CrossRef CAS; (b) C. Ding, J. Shi, D. Wang, Z. Wang, N. Wang, G. Liu, F. Xiong and C. Li, Visible light driven overall water splitting using cocatalyst/BiVO4 photoanode with minimized bias, Phys. Chem. Chem. Phys., 2013, 15(13), 4589–4595 RSC; (c) C. Ding, J. Shi, Z. Wang and C. Li, Photoelectrocatalytic Water Splitting: Significance of Cocatalysts, Electrolyte, and Interfaces, ACS Catal., 2017, 7(1), 675–688 CrossRef CAS; (d) S. Zhang, I. Ahmet, S.-H. Kim, O. Kasian, A. M. Mingers, P. Schnell, M. Kölbach, J. Lim, A. Fischer and K. J. J. Mayrhofer, et al., Different Photostability of BiVO4 in Near-pH-Neutral Electrolytes, ACS Appl. Energy Mater., 2020, 3(10), 9523–9527 CrossRef CAS PubMed.
- C. Ding, X. Zhou, J. Shi, P. Yan, Z. Wang, G. Liu and C. Li, Abnormal Effects of Cations (Li+, Na+, and K+) on Photoelectrochemical and Electrocatalytic Water Splitting, J. Phys. Chem. B, 2015, 119(8), 3560–3566 CrossRef CAS PubMed.
- Q. Mi, R. H. Coridan, B. S. Brunschwig, H. B. Gray and N. S. Lewis, Photoelectrochemical oxidation of anions by WO3 in aqueous and nonaqueous electrolytes, Energy Environ. Sci., 2013, 6(9), 2646–2653 RSC.
- D. Liu, J.-C. Liu, W. Cai, J. Ma, H. B. Yang, H. Xiao, J. Li, Y. Xiong, Y. Huang and B. Liu, Selective photoelectrochemical oxidation of glycerol to high value-added dihydroxyacetone, Nat. Commun., 2019, 10(1), 1779 CrossRef PubMed.
- (a) R. Tang, L. Wang, Z. Zhang, W. Yang, H. Xu, A. Kheradmand, Y. Jiang, R. Zheng and J. Huang, Fabrication of MOFs' derivatives assisted perovskite nanocrystal on TiO2 photoanode for photoelectrochemical glycerol oxidation with simultaneous hydrogen production, Appl. Catal., B, 2021, 296, 120382 CrossRef CAS; (b) Y.-H. Wu, D. A. Kuznetsov, N. C. Pflug, A. Fedorov and C. R. Müller, Solar-driven valorisation of glycerol on BiVO4 photoanodes: effect of co-catalyst and reaction media on reaction selectivity, J. Mater. Chem. A, 2021, 9(10), 6252–6260 RSC; (c) L. Luo, W. Chen, S.-M. Xu, J. Yang, M. Li, H. Zhou, M. Xu, M. Shao, X. Kong and Z. Li, et al., Selective Photoelectrocatalytic Glycerol Oxidation to Dihydroxyacetone via Enhanced Middle Hydroxyl Adsorption over a Bi2O3-Incorporated Catalyst, J. Am. Chem. Soc., 2022, 144(17), 7720–7730 CrossRef CAS PubMed; (d) J. Ouyang, X. Liu, B.-H. Wang, J.-B. Pan, S. Shen, L. Chen, C.-T. Au and S.-F. Yin, WO3 Photoanode with Predominant Exposure of {202} Facets for Enhanced Selective Oxidation of Glycerol to Glyceraldehyde, ACS Appl. Mater. Interfaces, 2022, 14(20), 23536–23545 CrossRef CAS PubMed; (e) H. Tateno, S.-Y. Chen, Y. Miseki, T. Nakajima, T. Mochizuki and K. Sayama, Photoelectrochemical Oxidation of Glycerol to Dihydroxyacetone Over an Acid-Resistant Ta:BiVO4 Photoanode, ACS Sustain. Chem. Eng., 2022, 10(23), 7586–7594 CrossRef CAS; (f) Y. Miao, Z. Li, Y. Song, K. Fan, J. Guo, R. Li and M. Shao, Surface active oxygen engineering of photoanodes to boost photoelectrochemical water and alcohol oxidation coupled with hydrogen production, Appl. Catal., B, 2023, 323, 122147 CrossRef CAS; (g) Y. Han, M. Chang, Z. Zhao, F. Niu, Z. Zhang, Z. Sun, L. Zhang and K. Hu, Selective Valorization of Glycerol to Formic Acid on a BiVO4 Photoanode through NiFe Phenolic Networks, ACS Appl. Mater. Interfaces, 2023, 15(9), 11678–11690 CrossRef CAS PubMed.
- T. W. Kim and K.-S. Choi, Nanoporous BiVO4 Photoanodes with Dual-Layer Oxygen Evolution Catalysts for Solar Water Splitting, Science, 2014, 343(6174), 990 CrossRef CAS PubMed.
- (a) J. K. Cooper, S. Gul, F. M. Toma, L. Chen, P.-A. Glans, J. Guo, J. W. Ager, J. Yano and I. D. Sharp, Electronic Structure of Monoclinic BiVO4, Chem. Mater., 2014, 26(18), 5365–5373 CrossRef CAS; (b) M. Lamers, W. Li, M. Favaro, D. E. Starr, D. Friedrich, S. Lardhi, L. Cavallo, M. Harb, R. van de Krol and L. H. Wong, et al., Enhanced Carrier Transport and Bandgap Reduction in Sulfur-Modified BiVO4 Photoanodes, Chem. Mater., 2018, 30(23), 8630–8638 CrossRef CAS.
- (a) Y. Zhang, H. Fan, M. Li and H. Tian, Ag/BiPO4 heterostructures: synthesis, characterization and their enhanced photocatalytic properties, Dalton Trans., 2013, 42(36), 13172–13178 RSC; (b) J. Cao, B. Xu, H. Lin and S. Chen, Highly improved visible light photocatalytic activity of BiPO4 through fabricating a novel p–n heterojunction BiOI/BiPO4 nanocomposite, Chem. Eng. J., 2013, 228, 482–488 CrossRef CAS.
- M. Favaro, F. F. Abdi, M. Lamers, E. J. Crumlin, Z. Liu, R. van de Krol and D. E. Starr, Light-Induced Surface Reactions at the Bismuth Vanadate/Potassium Phosphate Interface, J. Phys. Chem. B, 2018, 122(2), 801–809 CrossRef CAS PubMed.
- F. M. Toma, J. K. Cooper, V. Kunzelmann, M. T. McDowell, J. Yu, D. M. Larson, N. J. Borys, C. Abelyan, J. W. Beeman and K. M. Yu, et al., Mechanistic insights into chemical and photochemical transformations of bismuth vanadate photoanodes, Nat. Commun., 2016, 7(1), 12012 CrossRef.
- (a) J. Stoch and J. Gablankowska-Kukucz, The effect of carbonate contaminations on the XPS O 1s band structure in metal oxides, Surf. Interface Anal., 1991, 17(3), 165–167 CrossRef CAS; (b) J.-C. Dupin, D. Gonbeau, P. Vinatier and A. Levasseur, Systematic XPS studies of metal oxides, hydroxides and peroxides, Phys. Chem. Chem. Phys., 2000, 2(6), 1319–1324 RSC.
- A. M. Hilbrands, M. K. Goetz and K.-S. Choi, C–C Bond Formation Coupled with C–C Bond Cleavage during Oxidative Upgrading of Glycerol on a Nanoporous BiVO4 Photoanode, J. Am. Chem. Soc., 2023, 145(46), 25382–25391 CrossRef CAS PubMed.
- A. M. Gómez-Marín and J. M. Feliu, Pt(111) surface disorder kinetics in perchloric acid solutions and the influence of specific anion adsorption, Electrochim. Acta, 2012, 82, 558–569 CrossRef.
- R. Shannon, Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, 32(5), 751–767 CrossRef.
- E. Clementi and D. L. Raimondi, Atomic Screening Constants from SCF Functions, J. Chem. Phys., 2004, 38(11), 2686–2689 CrossRef.
- A. Stokłosa, J. Zajęcki and S. S. Kurek, Effective nuclear charge of an ion, Mater. Sci.-Pol., 2004, 22(1), 35–45 Search PubMed.
- B. Kang, H. Tang, Z. Zhao and S. Song, Hofmeister Series: Insights of Ion Specificity from Amphiphilic Assembly and Interface Property, ACS Omega, 2020, 5(12), 6229–6239 CrossRef CAS PubMed.
- (a) R. Zangi, M. Hagen and B. J. Berne, Effect of Ions on the Hydrophobic Interaction between Two Plates, J. Am. Chem. Soc., 2007, 129(15), 4678–4686 CrossRef CAS PubMed; (b) G. Graziano, Hydrophobic interaction of two large plates: An analysis of salting-in/salting-out effects, Chem. Phys. Lett., 2010, 491(1), 54–58 CrossRef CAS; (c) N. O. Johnson, T. P. Light, G. MacDonald and Y. Zhang, Anion–Caffeine Interactions Studied by 13C and 1H NMR and ATR–FTIR Spectroscopy, J. Phys. Chem. B, 2017, 121(7), 1649–1659 CrossRef CAS PubMed; (d) A. M. Hyde, S. L. Zultanski, J. H. Waldman, Y.-L. Zhong, M. Shevlin and F. Peng, General Principles and Strategies for Salting-Out Informed by the Hofmeister Series, Org. Process Res. Dev., 2017, 21(9), 1355–1370 CrossRef CAS.
- S. Park, L. Liu, Ç. Demirkır, O. van der Heijden, D. Lohse, D. Krug and M. T. M. Koper, Solutal Marangoni effect determines bubble dynamics during electrocatalytic hydrogen evolution, Nat. Chem., 2023, 15, 1532–1540 CrossRef CAS PubMed.
- M. Favaro, H. Kong and R. Gottesman, In situ and operando Raman spectroscopy of semiconducting photoelectrodes and devices for photoelectrochemistry, J. Phys. D: Appl. Phys., 2024, 57(10), 103002 CrossRef.
- E. Mendelovici, R. L. Frost and T. Kloprogge, Cryogenic Raman spectroscopy of glycerol, J. Raman Spectrosc., 2000, 31(12), 1121–1126 CrossRef CAS.
- K. Obata, R. van de Krol, M. Schwarze, R. Schomäcker and F. F. Abdi, In situ observation of pH change during water splitting in neutral pH conditions: impact of natural convection driven by buoyancy effects, Energy Environ. Sci., 2020, 13(12), 5104–5116 RSC.
- (a) R. A. Marcus, On the Theory of Oxidation-Reduction Reactions Involving Electron Transfer. I, J. Chem. Phys., 2004, 24(5), 966–978 CrossRef; (b) R. A. Marcus, Electron Transfer Reactions in Chemistry: Theory and Experiment (Nobel Lecture), Angew. Chem., Int. Ed., 1993, 32(8), 1111–1121 CrossRef; (c) H. Gerischer, The impact of semiconductors on the concepts of electrochemistry, Electrochim. Acta, 1990, 35(11), 1677–1699 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4sc01651c |
| This journal is © The Royal Society of Chemistry 2024 |