Development of enzymatically crosslinked natural deep eutectogels: versatile gels for enhanced drug delivery†
Liane
Meneses
 ,
Dimitra Antonia
Bagaki
,
Ana
Roda
,
Alexandre
Paiva
and
Ana Rita C.
Duarte
*
,
Dimitra Antonia
Bagaki
,
Ana
Roda
,
Alexandre
Paiva
and
Ana Rita C.
Duarte
*
LAQV-REQUIMTE, Departamento de Química, Faculdade de Ciências e Tecnologia, Universidade Nova de Lisboa, 2829-516 Caparica, Portugal. E-mail: ard08968@fct.unl.pt
First published on 30th October 2024
Abstract
Injectable hydrogels have been extensively studied due to their minimally invasive properties, ease of application, and void-filling properties. In this work, we tested the possibility to prepare a new type of gels, so called eutectogels, where water is replaced by a natural deep eutectic system (NADES), conferring it longer stability. Eutectogels based on betaine![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glycerol 1
glycerol 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, were prepared by enzymatic mediated crosslinking, using horseradish peroxidase (HRP) as catalyst and gelatine–phenol conjugated polymer. In comparison to hydrogels, that required higher enzyme concentration (15 U mL−1) to have gelation time under 2 minutes, the eutectogels were obtained using 10 and 5 U mL−1 of HRP, with gelation times of 30 and 50 seconds, respectively. Finally, ketoprofen was loaded into the polymeric matrix, and release studies were conducted. The presence of NADES was essential for the formulation of the drug loaded gel, which was able to release up to 70% of the drug within 10 days, therefore, it was possible to conclude that these eutectogels work as matrix for the controlled delivery of ketoprofen in aqueous medium. The in vitro biological evaluation of the individual components of the eutectogel support no cytotoxic effect, an early indication of potential biocompatibility.
2, were prepared by enzymatic mediated crosslinking, using horseradish peroxidase (HRP) as catalyst and gelatine–phenol conjugated polymer. In comparison to hydrogels, that required higher enzyme concentration (15 U mL−1) to have gelation time under 2 minutes, the eutectogels were obtained using 10 and 5 U mL−1 of HRP, with gelation times of 30 and 50 seconds, respectively. Finally, ketoprofen was loaded into the polymeric matrix, and release studies were conducted. The presence of NADES was essential for the formulation of the drug loaded gel, which was able to release up to 70% of the drug within 10 days, therefore, it was possible to conclude that these eutectogels work as matrix for the controlled delivery of ketoprofen in aqueous medium. The in vitro biological evaluation of the individual components of the eutectogel support no cytotoxic effect, an early indication of potential biocompatibility.
1. Introduction
Hydrogels are polymeric materials reticulated in a three-dimensional form that have water as their dispersion medium, hence they are not complete solids nor liquids. This allows them to have solid behaviour and maintain their shape and volume, while having the thermodynamic properties of a liquid, which facilitates the diffusion of solutes inside the matrix. Along these properties, hydrogels are also characterised by high elasticity and plasticity, decreased friction, biodegradability and biocompatibility.1 Since they were first developed in 1960, hydrogels have been applied in the food industry, materials and biological engineering and biosensors. The use of natural polymers, such as collagen or hyaluronic acid (HA), for hydrogel preparation, raised attention for their application in tissue engineering.1,2Considering their preparation, hydrogels can be divided into chemical and physical hydrogels. Physical hydrogels are formed by physical interactions, such as ionic, hydrogen bonds, van der Waals interactions, etc., which are considered weak, and the result is a heterogenous and unstable hydrogel that can lose its properties when subjected to a different environment. On the other side, in chemical hydrogels the polymeric chains are connected by covalent bonds, hence making them more stable. Polymers can be crosslinked by a variety of agents, such as radiation, chemical crosslinkers or enzymes.3
Enzymatically crosslinked hydrogels offer a wide range of advantages, such as high efficiency and selectivity, fast gelation rate, the use of mild reaction conditions and the fact that no toxic cross-linking agents are used.1,3 Moreover, they show great mechanical properties and can be prepared in situ, in the form of injectable hydrogels. Injectable hydrogels are particularly interesting for tissue engineering since they are able to fill any free space, can be formulated with cells, and do not require surgical implantation or removal, if they are biodegradable.4 The major concern associated with the preparation of injectable hydrogels is the control of the gelation time, which is closely related to the precursor/crosslinker concentration, and should be thoroughly optimised, not only to ensure adequate mechanical properties, but also to guarantee proper growth rate and differentiation of cells inside the polymeric matrix.4,5
Horseradish peroxidase (HRP) is one of the most used enzymes to crosslink phenol-groups due to the ease to tune the final hydrogel's characteristics by adjusting the concentrations of HRP and hydrogen peroxide (H2O2). HRP has been used in combination with numerous polymers, such as HA, gelatine, chitosan or alginate, with the main condition that these polymers must be modified with tyramine or hydroxyphenylpropionic acid to have phenol groups available.3,5 In the presence of phenolic hydroxyl groups and H2O2, HRP promotes the crosslinking through C–C and C–O coupling of the aromatic ring, which ends up forming the hydrogel.6
Over the past few years, several authors combined natural deep eutectic systems (NADES) and polymers to prepare hydrogels. For instance, Bianchi et al. reported the preparation of a gelatine-based hydrogel combined with choline chloride:glycerol, for the prolonged release of drug nanocrystals through mucosal delivery.7 Focusing on the delivery of drugs, the preparation of alginate based hydrogels has also been reported, using ibuprofen as model drug.8 Another example of the incorporation of NADES in gels, include self-polymerization of 2-hydroxyethylmethacrylate with choline chloride:fructose for the delivery of indomethacin9 and with choline chloride:ascorbic acid for the delivery of sunitinib malate.10
The number of publications concerning the use of NADES for the preparation of drug delivery systems using non-steroidal anti-inflammatory drugs is scarce and most focus on the application of NADES as enhancers of solubility in aqueous media.11 Nevertheless, the use of ibuprofen in a deep eutectic system (DES) formulation, allowed to improve its solubility in buffer, while achieving a controlled release profile.12 Recently, the preparation of alginate hydrogels with NADES was also assessed for the delivery of ibuprofen, which was successful.8
In a previous study it was observed that, in the presence of betaine-based NADES, the activity and thermostability of HRP was improved.13,14 In this work we proposed an application in which we took advantage of the improvement of HRP's activity in NADES, as well as of its capacity to crosslink polymers. Hence, the main goal of this proof-of-concept was to prepare enzymatically crosslinked gels using NADES as dispersion medium, in replacement of water, that will be called eutectogels. Gelatine was used as model polymer, since it is easily available. The gels were characterized in terms of their gelation time, stability, and mechanical properties. Their feasibility to be used as delivery system for ketoprofen, was also evaluated. To the extent of our knowledge this is the first report of the preparation of eutectogels through enzymatic crosslinking.
2. Materials and methods
2.1. Materials
For the preparation of NADES compounds used were D-(+)-xylose (Xyl, ≥ 99%, CAS 58-86-6), glycerol (Gly, ≥ 99.5% CAS 56-81-5), D-sorbitol (Sorb, ≥ 98%, CAS 50-70-4), DL-proline (Pro, 99%, CAS 609-36-9), obtained from Sigma-Aldrich (St. Louis, Missouri, USA), trehalose dihydrate (CAS 6138-23-4) kindly provided by Hayashibara Co., LDA (Okayama, Japan), betaine anhydrous (>97%, CAS 107-43-7) from TCI (Tokyo, Japan) and sucrose (Suc, CAS 57-50-1) obtained from Cmd Chemicals (Funchal, Portugal). Lyophilized powder of peroxidase from horseradish (HRP, type I, 89.63 U mg−1 solid, CAS 9003-99-0, Sigma-Aldrich) was used without further purification. Phenol-4-sulfonic acid sodium salt dihydrate (PSA, 98%, CAS 10580-19-5), 4-aminoantipyrine (4-AAP, ≥ 99%, CAS 83-07-8), hydrogen peroxide 30% solution (H2O2, CAS 7722-84-1), gelatine from bovine skin (∼225 Bloom, type B, CAS 9000-70-8), 2-morpholinoethanesulfonic acid buffer (MES, CAS 145224-94-8), sodium phosphate monobasic (≥99%, CAS 7558-80-7) and ketoprofen (≥98%, CAS 22071-15-4) were obtained from Sigma-Aldrich. N-Hydroxysuccinimide (NHS, ≥98%, CAS 6066-82-6) and 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC, ≥98%, CAS 25952-53-8) were purchased from Thermo Scientific (Kandel, Germany) and finally, tyramine hydrochloride (Tyr, CAS 60-19-5) was obtained from TCI. Phosphate buffered saline (PBS) tablets were obtained from Sigma-Aldrich and prepared as indicated by the supplier (one tablet/200 mL, yielding 10 mM phosphate buffer, pH 7.2–7.6). The chondrogenic cell line ATDC5, derived from mouse teratocarcinoma AT805, was purchased from the European Collection of Authenticated Cell Cultures (ECACC 99072806, UK). Dulbecco's Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F-12) was acquired from Gibco (UK). The trypsin 0.25%, Fetal Bovine Serum (FBS), and the antibiotic–antimycotic solutions (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 IU mL−1 penicillin, 10 mg mL−1 streptomycin and 25 μg mL−1 amphotericin) were acquired from Corning (USA). The MTS reagent (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, <2%) was obtained from Promega (USA).
000 IU mL−1 penicillin, 10 mg mL−1 streptomycin and 25 μg mL−1 amphotericin) were acquired from Corning (USA). The MTS reagent (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, <2%) was obtained from Promega (USA).
2.2. Preparation of NADES
The used natural deep eutectic systems (NADES) composed of betaine and glycerol (BGly) in a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, was prepared using the heating and stirring method.15 The individual components of the NADES were weighed into a flask, that was closed and heated at 40–60 °C, under constant stirring, until a transparent and viscous fluid was obtained. The water content of the NADES was determined by Karl-Fisher titration, performed in an 831 KF Coulometer (Metrohm, Herissau, Switzerland) with generator electrode without diaphragm, using Hydranal Coulomat AG (Honeywell, Charlotte, North Carolina, USA) as a reagent. For each sample, the water content was determined in triplicate. The prepared NADES were kept in closed flasks until used.
2, was prepared using the heating and stirring method.15 The individual components of the NADES were weighed into a flask, that was closed and heated at 40–60 °C, under constant stirring, until a transparent and viscous fluid was obtained. The water content of the NADES was determined by Karl-Fisher titration, performed in an 831 KF Coulometer (Metrohm, Herissau, Switzerland) with generator electrode without diaphragm, using Hydranal Coulomat AG (Honeywell, Charlotte, North Carolina, USA) as a reagent. For each sample, the water content was determined in triplicate. The prepared NADES were kept in closed flasks until used.
2.3. Synthesis of gelatine–phenol conjugates
The synthesis of gelatine–phenol conjugates (Glt–Ph) was adapted from elsewhere.16,17 Briefly, gelatine from bovine skin was dissolved at 2% w/v in MES buffer (50 mM, pH 6) at 60 °C. After complete dissolution of gelatine, the solution was let to cool to room temperature. The following compounds were added to the solution to initiate the reaction: 57.6 mM of Tyr, 38.3 mM of EDC and 19.2 mM of NHS. The pH was corrected to 6 using NaOH (10 M). The reaction proceeded overnight (16–18 h) under constant stirring, at 25 °C. To stop the reaction, 50 mM sodium phosphate was added to the media and stirred for 1 hour. The resultant polymer solution was dialyzed against deionized water, using SnakeSkin Dialysis tubing membranes (MWCO: 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 from Thermo Scientific) for a minimum of 48 h, until no absorbance peak was detected at 275 nm in a UV-vis spectrophotometer (GENESYS™ 50, Thermo Scientific). The dialysed polymer was lyophilized for 48 hours and stored in closed flasks until use.
000 from Thermo Scientific) for a minimum of 48 h, until no absorbance peak was detected at 275 nm in a UV-vis spectrophotometer (GENESYS™ 50, Thermo Scientific). The dialysed polymer was lyophilized for 48 hours and stored in closed flasks until use.
2.4. Quantification of gelatine modification extent
The conjugation reaction was confirmed by attenuated total reflection Fourier transform infrared spectroscopy (ATR-FTIR) performed in a PerkinElmer spectrum two spectrometer. The samples – gelatine, Tyr or Glt–Ph – were placed directly in the ATR-FTIR cell, and spectra were recorded from 4000 to 400 cm−1 resolution with 16 scans, at room temperature.To quantify the extent of the conjugation, 0.1% w/v Glt–Ph was dissolved in water and the absorbance at 275 nm was determined using a UV-vis spectrophotometer (GENESYS™ 50, Thermo Scientific). The percentage of tyramine in the sample was calculated through a calibration curve prepared with tyramine hydrochloride (0.000625 to 0.2% w/v). The total mass of tyramine in Glt–Ph was calculated through eqn (1). The yield of modification was calculated using eqn (2).
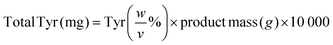 | (1) |
 | (2) |
2.5. Preparation of hydrogels
To prepare standard hydrogels, a solution of 6.25 w/v% Glt–Ph in PBS was prepared, by heating at high temperature and low stirring. After complete dissolution of the polymer, the hydrogels were produced by gently mixing Glt–Ph and HRP solution, and later adding the H2O2 solution to start the reaction. Two concentrations of HRP solution were tested and the concentration of each component in the hydrogel was 5% w/v Glt–Ph, using 5 or 10 U mL−1 HRP and 3 mM H2O2.162.6. Preparation of eutectogels
For the preparation of eutectogels, the NADES BGly (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio) was used. Due to the high viscosity of the mixture, water was added to the NADES, at a ratio of 90 wt% BGly and 10 wt% water (90BGly). Using 90BGly, a 6.25% w/v Glt–Ph solution was prepared, by heating at high temperatures and low stirring and let to rest overnight, before using to prepare gels. The gels were prepared in the same rational as before, freshly prepared solution of HRP was added to Glt–Ph in NADES solution and gently mixed, after which H2O2 was added for the crosslinking reaction to start. The final concentration of the components in the eutectogel were, 5% w/v Glt–Ph, 5 or 10 U mL−1 HRP and 3 mM H2O2.
2 molar ratio) was used. Due to the high viscosity of the mixture, water was added to the NADES, at a ratio of 90 wt% BGly and 10 wt% water (90BGly). Using 90BGly, a 6.25% w/v Glt–Ph solution was prepared, by heating at high temperatures and low stirring and let to rest overnight, before using to prepare gels. The gels were prepared in the same rational as before, freshly prepared solution of HRP was added to Glt–Ph in NADES solution and gently mixed, after which H2O2 was added for the crosslinking reaction to start. The final concentration of the components in the eutectogel were, 5% w/v Glt–Ph, 5 or 10 U mL−1 HRP and 3 mM H2O2.
2.7. Characterization of eutectogels
The rheological properties of the prepared hydrogels and eutectogels were determined using a MCR102 rheometer (Anton Paar) fitted with an 8 mm parallel plate geometry with sandblasted surface (PP08/S, Anton Paar) in oscillatory method. The measurements were performed in the linear viscoelastic region (LVR) of the materials, determined by amplitude sweep at a frequency of 1 Hz, 37 °C and within a shear strain (γ) ranging from 10−6 to 10 (LVR from 10−6 to 0.5).18 Frequency sweep analysis was performed under controlled deformation (using a shear strain of 0.2, in the LVR). The gels were equilibrated at 37 °C for 10 min and the storage (G′) and loss (G′′) modulus were determined in a frequency range from 10 to 0.1 rad s−1. The properties of the eutectogels were analysed after preparation and at 1 month, 3 months and 1 year. All measurements were performed in triplicate.
2.8. Preparation of drug delivery systems
The amount of ketoprofen released from the samples was determined using a calibration curve of ketoprofen in PBS with concentrations ranging from 1 to 10 mg L−1 (Fig. S1, ESI†). The absorbance of the samples at 262 nm was determined in a microplate reader (VICTOR Nivo™, PerkinElmer, USA). The solubility of ketoprofen in PBS was determined as the maximum amount of ketoprofen released to the PBS, once equilibrium was reached.
The release profile of ketoprofen in PBS was obtained by calculating Mt/Minf, where Mt represents the mass of ketoprofen released at time t and Minf represents the total mass of ketoprofen released at equilibrium.
2.9. Cytotoxicity
The cytotoxicity evaluation was performed in a chondrogenic cell line (ATDC5). Cells were cultured in media composed of DMEM/F-12 with phenol red, supplemented with 10% FBS and 1% PS, and incubated in a humidified incubator, with 5% CO2, at 37 °C. Media was changed every 2–3 days, until cells were confluent (70–80%).The samples for testing were prepared as follows: the initial components of the gel were individually prepared by solubilization in the culture media at 37 °C for 24 hours, at concentrations of Glt–Ph 1.04% w/v, HRP 2.44 U mL−1, H2O2 0.75 mM and 90BGly 125 mg mL−1. Cytotoxicity was assessed through the MTS colorimetric assay, according to ISO 10993-5.20 Suspensions of 1.5 × 105 cells mL−1 were seeded in a 96-well plate for 24 hours, after which the media was removed and the cells exposed to 100 μL of each sample, in triplicate, for 24 hours. The cells were then washed with PBS and MTS was added (1.6% in DMEM/F-12 + 0.5% FBS). After an incubation period of 3 hours, the absorbance was measured at 490 nm (VICTOR Nivo™ microplate reader, PerkinElmer, USA), and the results expressed as cell viability (%), determined as previously described.11
3. Results and discussion
3.1. Synthesis of gelatine–phenol conjugates
The gelatine–phenol (Glt–Ph) conjugates were obtained using bovine skin gelatine and tyramine hydrochloride. The synthesis was performed through the carbodiimide-mediated condensation of the amino groups of tyramine and the carboxyl groups of gelatine, which allows for the binding of phenol groups to a carboxyl group, as shown in Fig. 1.17 The cross-linking of protein chains occurs through tyrosine residues; however they can be inaccessible or be present in small amounts, hence the incorporation of more phenol groups is essential for hydrogel production.16 | ||
| Fig. 1 Schematic representation of the conjugation reaction used for the synthesis of gelatine–phenol conjugates. | ||
The synthesis of Glt–Ph conjugates was performed as described by Chuang et al., using fixed concentrations of gelatine, Tyr, EDC and NHS.17 The reaction was carried out over-night at 25 °C, as indicated in Table 1.
| Experiment | Glt–Ph1 | Glt–Ph2 | Glt–Ph3 |
|---|---|---|---|
| pH | 6.0 | 6.0 | 6.0 |
| Temperature (°C) | 25 | 25 | 25 |
| Tyr conjugation yield (%) | 8.47 | 7.10 | 5.60 |
The success of the conjugation reaction was determined through UV-vis and through ATR-FTIR analysis. Performing a UV-Vis scan, using a 0.1 w/v% Glt–Ph solution, allowed to confirm that the conjugation reaction occurred, since a peak at 275 nm was only visible in the presence of tyramine. To further confirm the conjugation, ATR-FTIR analysis was performed. The spectra obtained are visible in Fig. 2 and are in accordance with others found in literature.21,22 Briefly, in gelatine's spectrum, it is possible to see the stretching of amine bonds, at 3300 cm−1 and bending at 1590 cm−1. The carbonyl groups of gelatine are visible at wavenumbers 1640 cm−1.22 As for tyramine hydrochloride, it was also possible to identify the bands corresponding to amine I vibration, at ∼3070 cm−1 and from 800 to 600 cm−1, along with band at 1500 cm−1, corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C of the phenol group.21 In the spectrum of the conjugated polymer (Glt–Ph), it is possible to identify bands that correlate to the ones found both in gelatine and in tyramine, as well as the disappearance of the bands from 800 to 600 cm−1, associated to the amine group of tyramine, indicating the formation of the bond between tyramine and gelatine. There was also an increase in intensity of the bands around 1300 and 1100 cm−1, associated to the stretching of C–N bonds, which are formed during the condensation reaction.21
C of the phenol group.21 In the spectrum of the conjugated polymer (Glt–Ph), it is possible to identify bands that correlate to the ones found both in gelatine and in tyramine, as well as the disappearance of the bands from 800 to 600 cm−1, associated to the amine group of tyramine, indicating the formation of the bond between tyramine and gelatine. There was also an increase in intensity of the bands around 1300 and 1100 cm−1, associated to the stretching of C–N bonds, which are formed during the condensation reaction.21
 | ||
| Fig. 2 ATR-FTIR spectra of gelatine (Glt, full black line), tyramine hydrochloride (Tyr, dashed black line) and the conjugate Glt–Ph (dashed grey line). The spectra were obtained between 4000 and 400 cm−1, in transmittance mode (Individual spectrum can be seen in Fig. S2, ESI†). | ||
With the confirmation that the reaction was successful, the yield of tyramine incorporation was quantified, through UV-vis, using a calibration curve prepared with tyramine hydrochloride. Although the reaction conditions were kept the same, the yield of Tyr conjugation was not stable, varying from 5.60 to 8.47% (Table 1).
3.2. Preparation of gels and gelation time
To access the viability of obtaining gels with the produced Glt–Ph conjugates, hydrogels were prepared using 5% w/v Glt–Ph in PBS, and concentrations of HRP ranging from 0.15 to 15 U mL−1. Under all conditions tested, hydrogels were formed and the relation between HRP concentration and gelation time was assessed. Higher HRP concentrations yielded lower gelation times, for instance 0.15 and 15 U mL−1, lead to 13 and 2 minutes of gelation time, respectively.For the preparation of eutectogels, betaine![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glycerol (BGly) in a molar ratio of 1
glycerol (BGly) in a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was used. The full characterization of this NADES has been published elsewhere.13,14 After several tests, it was concluded that Glt–Ph was not soluble in NADES, therefore, a mixture of 90 wt% BGly and 10 wt% water (BGly90) was used for the preparation of the eutectogels. Adding water to NADES is commonly used to tune the viscosity of the systems and, as described by other authors, such low amount of water is not expected to disrupt the hydrogen bond network of the NADES.23
2 was used. The full characterization of this NADES has been published elsewhere.13,14 After several tests, it was concluded that Glt–Ph was not soluble in NADES, therefore, a mixture of 90 wt% BGly and 10 wt% water (BGly90) was used for the preparation of the eutectogels. Adding water to NADES is commonly used to tune the viscosity of the systems and, as described by other authors, such low amount of water is not expected to disrupt the hydrogen bond network of the NADES.23
Eutectogels were then successfully prepared using the same conditions described for hydrogels, 5% w/v Glt–Ph dissolved in BGly90, using only two concentrations of HRP, namely 5 and 10 U mL−1. As it was observed during the preparation of hydrogels, the gelation time was closely related to the HRP concentration. The same trend was observed for the eutectogels, with gelation times of 52 and 30 seconds for 5 and 10 U mL−1 of HRP, respectively (Fig. 3). The concentrations of HRP used for the eutectogels preparation were chosen considering that the enzymatic activity of HRP is improved in the presence of NADES.14 In this case, using 10 U mL−1 of HRP allowed to achieve the lowest gelation time of the study. The use of a higher concentration of enzyme to further reduce the gelation time was not considered since it could impair the homogenization of the compounds of the eutectogel, resulting in an uneven material.
 | ||
| Fig. 3 Gelation time of eutectogels (mean ± SD) prepared with 5% w/v Glt–Ph in a BGly90 matrix (full data is reported in Table S1, ESI†). | ||
The relation between gelation time and HRP concentration had already been described by other authors that observed the same behaviour: increasing concentrations of HRP lead to shorter gelation times.16 The gelation times obtained by these authors are not comparable to the ones obtained in this work, since the concentration of H2O2 used was much higher (50 mM against 3 mM).16 In a more recent study, similar results were obtained, in a time range closer to the one obtained in our work. Mubarok et al. determined that for HRP concentrations of 1, 5 and 15 U mL−1, the gelation was 57, 35 and 15 seconds, respectively.24 Nevertheless, the most important aspects when considering in vivo applications of hydrogels, are that hydrogels should be successfully obtained in less than 1 minute, along with the fact that they can be prepared using low amounts of hydrogen peroxide, since it is a strong oxidant which can have toxic effects on cells and live tissues.16,24 The eutectogels herein developed, comply with both critical features.
3.3. Characterization of the produced gels
These results indicate that the structure created in the eutectogels is more robust than the one obtained in the conventional hydrogels. This can be linked to the presence of BGly, since the dense hydrogen bond network of NADES improves not only the link between its components, but also the protein-NADES bonds, creating a more stable 3D structure.8
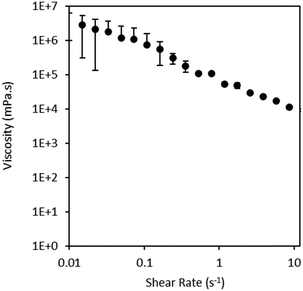 | ||
| Fig. 4 Viscosity vs. shear rate obtained for the mixture of 5% w/v Glt–Ph in 90BGly (full data is reported in Table S2, ESI†). | ||
Besides the viscosity of the injectable material, when designing a hydrogel it is also important to assess storage (G′) and loss (G′′) moduli, which provide information about the elastic and viscous behaviour of the material, correlating to its ability to absorb or dissipate energy.25
The frequency sweep study for the PBS-based hydrogels (Fig. 5) shows a typical hydrogel profile, with G′ consistently above G′′, which is representative of elastic behaviour and also of crosslinked hydrogels.26,27 It can also be observed that the different concentrations of HRP (5 and 10 U mL−1) have no impact on the rheological properties of the hydrogels, since the same resistance to deformation was obtained in both samples.
 | ||
| Fig. 5 Frequency sweep of PBS-based hydrogels prepared with different HRP concentrations: 5 U mL−1 (black open symbols) and 10 U mL−1 (grey filled symbols). Storage (G′) and loss (G′′) moduli as a function of frequency (rad s−1) (full data is reported in Tables S3 and S4, ESI†). | ||
To determine if the storage time had any influence on the properties, the rheological properties of the eutectogels were determined after preparation and periodically, after 1 month, 3 months and again after 1 year, since the gels were still intact at this point. As it can be seen in Fig. 6, after preparation (blue symbols), the eutectogels produced with both concentrations of HRP, show G′ consistently above G′′, indicating that they behave as elastic materials, and there is no cross of G′ and G′′, as expected for a crosslinked gel.27
 | ||
| Fig. 6 Frequency sweep of eutectogels prepared with (A) 10 U mL−1 HRP and (B) 5 U mL−1 HRP. Storage (G′) and loss (G′′) moduli as a function of frequency (Hz). Blue represents the samples after preparation (t = 0), orange represents the samples after 1 month, green after 3 months and grey after 1 year of storage at 37 °C. The full data is reported in Tables S5 and S6 (ESI†). | ||
Moreover, in all conditions tested, G′ shows independence of frequency up to 5 rad s−1, with a slight decrease for higher frequencies. It is also possible to observe that, especially for the longer time point (1 year), the absolute value of both moduli becomes higher, indicating higher resistance to deformability. This increase could mean that there is an increase of the gels’ stiffness over time, which could be related to the evaporation of water during storage or even to a higher degree of crosslinking, caused by residual HRP activity over time. In comparison to PBS-based hydrogels, eutectogels have lower G′ and G′′, indicating that these gels are softer, which can also represent more flexibility.26 This could be explained by the presence of glycerol in the formulation, since it is a well-known plasticizer for biopolymers, particularly hydrophilic polymers such as gelatine. Its presence can have impact on the polymers’ properties, for instance increasing their flexibility and elasticity and decreasing its Young's modulus.28
3.4. Incorporation and release profile of ketoprofen from eutectogels
Injectable hydrogels offer a wide range of advantages, including their application as drug release matrices. Herein, we evaluated the incorporation of ketoprofen, and its subsequential release from the eutectogels, in order to access the feasibility of their application for biomedical ends.Polarized optical microscopy (POM), was used to confirm the complete dissolution of ketoprofen in NADES, by using a cross polarizer that allows to identify if there are crystals present in the sample, which would mean that the mixtures were not homogeneous.29 The images collected can be seen in Fig. 7. The uniform black images represent amorphous and homogenous liquids, confirming the total dissolution of up to 10 mgKet gcarrier−1 in BGly (Fig. 7(A)), as well as in the gel precursor mixture, Glt–Ph in 90BGly (Fig. 7(B)).
 | ||
| Fig. 7 Polarized optical microscopy images of (A) ketoprofen dissolved in pure BGly and (B) ketoprofen dissolved in gel precursor solution (Glt–Ph in 90BGly). | ||
Hence, eutectogels were successfully produced with the incorporation of ketoprofen, at the final composition of 5 wt% Glt–Ph in 90BGly, 10 U mL−1 of HRP, and 3 mM H2O2, with 10 mg g−1 of drug. Despite our attempts, it was not possible to obtain hydrogels with the incorporation of ketoprofen in the dispersion medium. Hence, the release of ketoprofen was evaluated from two samples, the eutectogel and BGly alone, followed through UV-vis until a plateau was achieved (full data is reported in Tables S7 and S8, ESI†). For both samples, the concentration of ketoprofen determined upon stabilization was assumed as the solubility of ketoprofen in PBS in the studied conditions, reported in Table 2. The solubilities determined are close to the expected since ketoprofen solubility in PBS has been described to be 500 μg mL−1.30
| Solubility in PBS (μg mL−1) | |
|---|---|
| Ketoprofen | 50030 |
| Eutectogel | 657.5 ± 89.5 |
| BGly | 447.6 ± 26.2 |
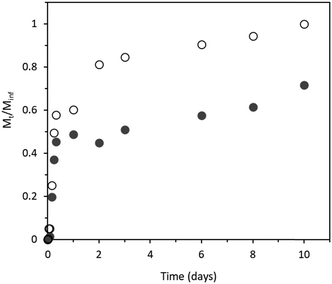
The release profile of ketoprofen from BGly and from the eutectogel is shown in Fig. 8. For BGly, 3 phases of release can be identified. First, there was a fast release over the first 8 h, during which 60% of the total ketoprofen was released to the medium. The second phase is comprised between 8 h and 2 days, with the release reaching 80%. The third, and last release phase, occurred from 2 days until 10 days, with a release of the remaining 20% over 8 days, at a much slower rate. In the case of the eutectogel, only 2 phases were observed. The first phase, corresponding to a fast release occurred over the first 24 h however, with only 50% of the total ketoprofen being released. From there until day 10 the second phase was observed, and ketoprofen was released at a slower rate, reaching 70% of the total ketoprofen entrapped in the eutectogel. This indicates that the release could be prolonged for more than 10 days, conferring a more sustained release profile. In order to avoid losses or toxic effects, the amount of drug in the eutectogel should be optimised to guarantee that 100% of the drug is released from the matrix.
The observed profile corresponds to a type II release profile,31 characterized by a fast release in an initial short time period that tends to stabilize over a longer period. In real conditions, i.e. administration to the blood stream, for instance, these results could be different, since the concentration of drug in plasma is affected by a variety of factors such as stress, physical activity or sleeping. Moreover, physiological conditions, such as presence of enzymes, may interfere with the process and rate of degradation of the eutectogel, which will consequently affect the drug release rate and profile. Nevertheless, as a proof-of-concept work, these results seem very promising for the design of injectable drug delivery systems. To optimize the design of the drug delivery system, two main conditions should be considered: the amount of ketoprofen loaded, and the percentage of gelatine used, since it has been reported that it can influence the release profile of the drug.
The ketoprofen release profile herein obtained was comparable with the one reported by other authors in carriers containing gelatine.32,33 When ketoprofen was loaded in hybrid aerogels of silica and gelatine, a sustained release profile was obtained. However, the authors observed that the profile was dependent on the amount of gelatine in the aerogel, with the higher percentages yielding a more controlled profile, without an initial stage of fast release observed for lower gelatine contents.32 On a different study, ketoprofen was loaded into polyvinyl alcohol and gelatine scaffolds reinforced with bioglass to evaluate its potential as drug carrier. It was concluded that the scaffolds could successfully create a controlled delivery of ketoprofen after an initial fast release.33
The release profile of ketoprofen from the eutectogel can be modelled through the application of a simple mathematical model derived from Fick's law of diffusion. Despite being simple to apply, the model relies on multiple assumptions that must be taken into consideration. For instance, the assumptions include that the polymer has a concentration of drug above its solubility limit, or that the equilibrium is not altered during the release experiment.34 Assuming these conditions, Eqn 3 can be applied to determine the kinetic constant (K) and release exponent (n), that are characteristic of the drug release mechanism. The linearity of the equation can only be verified up to 60% of total amount of drug released, M(inf).
 | (3) |
The values of the calculated parameters were n = 2.02 and K = 0.0078 (R2 = 0.9597). Values of n > 1 reflect that ketoprofen is released from the matrix following a super case II transport, which is common in hydrogels due to the swelling of the matrix.35 Typically, in hydrogels, the drug is released in multiple steps. An initial release happens when the polymer is immersed in aqueous medium, due to a high concentration gradient. Then, as the hydrogel starts to retain water (or swelling) two diffusion forces appear, one being in the hydrated surface and the other being between the polymer surface and the environment. Once the swelling is complete, the system reaches equilibrium, and the concentration of drug becomes constant in the medium. The final stage of release is only observed over long time periods, as the polymer starts to degrade, releasing the entrapped drug to the media.35
3.5. In vitro cytotoxicity of eutectogel components
The in vitro cytotoxicity was evaluated for the individual components of the eutectogel. As shown in Fig. 9, up to the concentrations herein studied, all components presented a viability ≥70%, which is considered non-cytotoxic, according to ISO 10993-5. This is a good indicator of biocompatibility, however, further tests would be needed to evaluate the biocompatibility of the eutectogel.20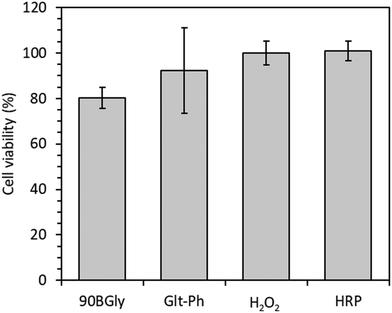 | ||
| Fig. 9 Cell viability (mean ± SD, %) in the presence of 90BGly 125 mg mL−1, Glt–Ph 1.04% w/v, H2O2 0.75 mM, and HRP 2.44 U mL−1. (The raw data used to obtain the presented cell viability can be found on Table S9, ESI†). | ||
4. Conclusions
Throughout this work the preparation of eutectogels using enzymatic crosslinking was discussed. The main goal of this work was to develop a formulation for an injectable gel, to be crosslinked in vivo, that could also be used for drug delivery and presented enhanced properties over conventional hydrogels. Using Glt–Ph with an average of 6% tyramine, two types of gels were prepared, hydrogels, using PBS as solvent, and eutectogels, in which the aqueous solvent was substituted by the BGly. The preparation of PBS-based hydrogels was optimised, leading to the optimal conditions of 5 wt% polymer, 5/10 U mL−1 HRP and 3 mM H2O2. The same conditions were later used in the preparation of the eutectogels. From the results obtained it was concluded that the concentration of HRP had high influence on the gelation time of the gel, reducing it from 52 to 30 seconds, when using 5 or 10 U mL−1 of HRP, respectively. Moreover, the eutectogels were more stable than regular hydrogels, both in solvent and in the dry form, which they kept for one year at 37 °C. Concerning the rheological properties, the injectability of the mixture of 90BGly and Glt–Ph was assessed, and it was concluded that despite its high viscosity it presented shear-thinning behaviour, which is a good indicator of injectability. The prepared eutectogels showed a typical rheological behaviour for gels, with a storage modulus higher than the loss modulus, at frequencies up to 10 rad s−1. It was also observed that storage at 37 °C had impact in the absolute values of storage and loss moduli, which increased for all the samples tested, independently of the enzyme concentration. Finally, a proof-of-concept test of the application of the eutectogels as drug delivery systems was conducted using ketoprofen. The release study showed that after an initial period (24 h) during which the concentration of drug increased rapidly in the medium, a stable release could be maintained up to 10 days. Moreover, the pure components of the eutectogel were not cytotoxic under the studied conditions, which can be an early indicator of biocompatibility, even though further studies would have to be conducted to confirm it.Overall, the goals proposed for this work were achieved, since it was possible to obtain eutectogels that had better properties that the PBS-based hydrogels. The evidence of injectability of the precursor mixture and short gelation times are promising for their application as in vivo injectable gels, with the possibility to use them as drug release systems. The future steps of this work include biocompatibility assays of the gels, which are crucial for the development of this system for biomedical applications.
Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
The authors have no conflict of interests to declare.Acknowledgements
This project has received funding from the European Union's Horizon 2020 - European Research Council (ERC) under grant agreement ERC-2016-CoG 725034. This work received support through the Associate Laboratory for Green Chemistry (LAQV) and help from FCT/MCTES (LA/P/0008/2020 DOI 10.54499/LA/P/0008/2020, UIDP/50006/2020 DOI 10.54499/UIDP/50006/2020 and UIDB/50006/2020 DOI 10.54499/UIDB/50006/2020), through national funds. This work also received funding from FCT/MCTES through the PhD scholarship DOI 10.54499/SFRH/BD/148510/2019 and through the contract 2022.04001.CEECIND.References
- Z. Li, F. Lu and Y. Liu, A Review of the Mechanism, Properties, and Applications of Hydrogels Prepared by Enzymatic Cross-linking, J. Agric. Food Chem., 2023, 71(27), 10238–10249 CrossRef CAS PubMed.
- A. S. Hoffman, Hydrogels for biomedical applications, Adv. Drug Delivery Rev., 2012, 64(Supp), 18–23 CrossRef.
- S. Trombino, C. Servidio, F. Curcio and R. Cassano, Strategies for hyaluronic acid-based hydrogel design in drug delivery, Pharmaceutics, 2019, 11(8), 1–17 CrossRef PubMed.
- L. S. Wang, J. E. Chung, P. Pui-Yik Chan and M. Kurisawa, Injectable biodegradable hydrogels with tunable mechanical properties for the stimulation of neurogenesic differentiation of human mesenchymal stem cells in 3D culture, Biomaterials, 2010, 31(6), 1148–1157 CrossRef CAS PubMed.
- F. Lee, K. H. Bae and M. Kurisawa, Injectable hydrogel systems crosslinked by horseradish peroxidase, Biomed. Mater., 2015, 11(1), 14101 CrossRef PubMed.
- J. J. Roberts, P. Naudiyal, K. S. Lim, L. A. Poole-warren and P. J. Martens, A comparative study of enzyme initiators for crosslinking phenol-functionalized hydrogels for cell encapsulation, Biomater. Res., 2016, 1–12 CAS.
- M. B. Bianchi, C. Zhang, E. Catlin, G. Sandri, M. Calderón and E. Larrañeta, et al., Bioadhesive eutectogels supporting drug nanocrystals for long-acting delivery to mucosal tissues, Mater Today Bio., 2022, 17(December), 100471 CrossRef CAS PubMed.
- S. N. Pedro, M. S. M. Mendes, B. M. Neves, I. F. Almeida, P. Costa and I. Correia-Sá, et al., Deep Eutectic Solvent Formulations and Alginate-Based Hydrogels as a New Partnership for the Transdermal Administration of Anti-Inflammatory Drugs, Pharmaceutics, 2022, 14(4), 827 CrossRef CAS PubMed.
- C. Mukesh, K. K. Upadhyay, R. V. Devkar, N. A. Chudasama, G. G. Raol and K. Prasad, Preparation of a Noncytotoxic Hemocompatible Ion Gel by Self-Polymerization of HEMA in a Green Deep Eutectic Solvent, Macromol. Chem. Phys., 2016, 217(17), 1899–1906 CrossRef CAS.
- M. Mokhtarpour, H. Shekaari and A. Shayanfar, Design and characterization of ascorbic acid based therapeutic deep eutectic solvent as a new ion-gel for delivery of sunitinib malate, J. Drug Delivery Sci. Technol., 2020, 56, 101512, DOI:10.1016/j.jddst.2020.101512.
- A. Roda, A. Paiva and A. R. C. Duarte, Therapeutic liquid formulations based on low transition temperature mixtures for the incorporation of anti-inflammatory drugs, Pharmaceutics, 2021, 13(10), 1620 CrossRef CAS PubMed.
- I. M. Aroso, R. Craveiro, Â. Rocha, M. Dionísio, S. Barreiros and R. L. Reis, et al., Design of controlled release systems for THEDES - Therapeutic deep eutectic solvents, using supercritical fluid technology, Int. J. Pharm., 2015, 492(1–2), 73–79 CrossRef CAS PubMed.
- L. Meneses, N. F. Gajardo-Parra, E. Cea-Klapp, J. M. Garrido, C. Held and A. R. Duarte, et al., Improving the activity of horseradish peroxidase in betaine-based natural deep eutectic systems, RSC Sustainainability, 2023, 1(4), 886–897 RSC.
- N. F. Gajardo-Parra, L. Meneses, A. R. C. Duarte, A. Paiva and C. Held, Assessing the Influence of Betaine-Based Natural Deep Eutectic Systems on Horseradish Peroxidase, ACS Sustainable Chem Eng., 2022, 10(38), 12873–12881 Search PubMed.
- L. Meneses, F. Santos, A. R. Gameiro, A. Paiva and A. R. C. Duarte, Preparation of binary and ternary deep eutectic systems, J. Visualized Exp., 2019, 152, e60326 Search PubMed.
- S. Sakai, K. Hirose, K. Taguchi, Y. Ogushi and K. Kawakami, An injectable, in situ enzymatically gellable, gelatin derivative for drug delivery and tissue engineering, Biomaterials, 2009, 30(20), 3371–3377, DOI:10.1016/j.biomaterials.2009.03.030.
- C.-H. Chuang, R.-Z. Lin, H.-W. Tien, Y.-C. Chu, Y.-C. Li and J. M. Melero-Martin, et al., Enzymatic regulation of functional vascular networks using gelatin hydrogels, Acta Biomater., 2015, 19(1), 85–99 CrossRef CAS PubMed.
- M. Nicholls, A. Manjoo, P. Shaw, F. Niazi and J. Rosen, Rheological properties of commercially available hyaluronic acid products in the United States for the treatment of osteoarthritis knee pain, Clin. Med. Insights: Arthritis Musculoskeletal Disord., 2018, 11, 1–5 Search PubMed.
- A. Roda, A. Paiva, C. Rita and A. Duarte, A Low Transition Temperature Mixture-based viscosupplementation complemented with celecoxib for osteoarthritis treatment, Int. J. Pharm., 2024, 656, 124088 CrossRef CAS PubMed.
- International Organization for Standardization ISO 10993-5, Biological evaluation of medical devices – Part 5: Tests for in vitro cytotoxicity, 2009.
- N. H. Nguyen, Q. A. Bui, L. H. Dang, D. T. Nguyen and N. Q. Tran, Preparation of injectable free-HRP-mediated and self-catalyzed gelatin-tyramine hydrogel for biomedical applications, Vietnam J Chem., 2022, 60(5), 606–614 CrossRef CAS.
- P. H. L. Tran, T. T. D. Tran, Van Vo T, C. L. N. Vo and B. J. Lee, Novel multifunctional biocompatible gelatin-oleic acid conjugate: Self-assembled nanoparticles for drug delivery, J. Biomed. Nanotechnol., 2013, 9(8), 1416–1431 CrossRef CAS PubMed.
- Y. Dai, G.-J. Witkamp, R. Verpoorte and Y. H. Choi, Tailoring properties of natural deep eutectic solvents with water to facilitate their applications, Food Chem., 2015, 187, 14–19 CrossRef CAS PubMed.
- W. Mubarok, Y. Qu and S. Sakai, Influence of Hydrogen Peroxide-Mediated Cross-Linking and Degradation on Cell-Adhesive Gelatin Hydrogels, ACS Appl. Bio Mater., 2021, 4(5), 4184–4190 CrossRef CAS PubMed.
- M. H. Chen, L. L. Wang, J. J. Chung, Y. H. Kim, P. Atluri and J. A. Burdick, Methods to Assess Shear-Thinning Hydrogels for Application As Injectable Biomaterials, ACS Biomater. Sci. Eng., 2017, 3(12), 3146–3160 CrossRef CAS PubMed.
- H. Alizadeh Sardroud, S. Nemati, A. Baradar Khoshfetrat, M. Nabavinia and Y. Beygi Khosrowshahi, Barium-cross-linked alginate-gelatine microcapsule as a potential platform for stem cell production and modular tissue formation, J. Microencapsulation, 2017, 34(5), 488–497 CrossRef CAS PubMed.
- G. Stojkov, Z. Niyazov, F. Picchioni and R. K. Bose, Relationship between structure and rheology of hydrogels for various applications, Gels, 2021, 7(4), 255 CrossRef CAS.
- N. Suderman, M. I. N. Isa and N. M. Sarbon, The effect of plasticizers on the functional properties of biodegradable gelatin-based film: A review, Food Biosci., 2018, 24, 111–119 CrossRef CAS.
- F. Santos, M. I. P. S. Leitão and A. R. C. Duarte, Properties of therapeutic deep eutectic solvents of L-arginine and ethambutol for tuberculosis treatment, Molecules, 2019, 24, 1 Search PubMed.
- Cayman. Product Information (±)-Ketoprofen [Internet]. 2022 [cited 2024 Mar 1]. Available from: https://www.caymanchem.com/product/10006661/(±)-ketoprofen.
- A. K. Bajpai, S. K. Shukla, S. Bhanu and S. Kankane, Responsive polymers in controlled drug delivery, Prog. Polym. Sci., 2008, 33, 1088–1118 CrossRef CAS.
- M. Kéri, A. Forgács, V. Papp, I. Bányai, P. Veres and A. Len, et al., Gelatin content governs hydration induced structural changes in silica-gelatin hybrid aerogels – Implications in drug delivery, Acta Biomater., 2020, 105(March 2020), 131–145 CrossRef PubMed.
- A. I. Raafat and W. M. Abd-Allah, In vitro apatite forming ability and ketoprofen release of radiation synthesized (gelatin-polyvinyl alcohol)/bioglass composite scaffolds for bone tissue regeneration, Polym. Compos., 2018, 39(3), 606–615 CrossRef CAS.
- J. Siepmann and N. A. Peppas, Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC), Adv. Drug Delivery Rev., 2001, 48, 139–157 CrossRef CAS PubMed.
- E. S. Bacaita, B. C. Ciobanu, M. Popa, M. Agop and J. Desbrieres, Phases in the temporal multiscale evolution of the drug release mechanism in IPN-type chitosan based hydrogels, Phys. Chem. Chem. Phys., 2014, 16(47), 25896–25905 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4tb01672f |
| This journal is © The Royal Society of Chemistry 2024 |